UNC2881 (UNC-2881) is a novel, potent and specific Mer tyrosine kinase inhibitor with potential utility for prevention and treatment of pathologic thrombosis. It exhibits 83- and 58-fold higher selectivity over Tyro3 and Axl, respectively, and inhibits Mer with an IC50 of 4.3 nM. Since Mer kinase controls the second stage of platelet activation, using Mer inhibitors to prevent thrombosis with a lower risk of bleeding than existing treatments presents an opportunity.
Physicochemical Properties
| Molecular Formula | C25H33N7O2 | |
| Molecular Weight | 463.58 | |
| Exact Mass | 463.269 | |
| Elemental Analysis | C, 64.77; H, 7.18; N, 21.15; O, 6.90 | |
| CAS # | 1493764-08-1 | |
| Related CAS # |
|
|
| PubChem CID | 71721525 | |
| Appearance | White solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Index of Refraction | 1.666 | |
| LogP | 3.03 | |
| Hydrogen Bond Donor Count | 4 | |
| Hydrogen Bond Acceptor Count | 7 | |
| Rotatable Bond Count | 10 | |
| Heavy Atom Count | 34 | |
| Complexity | 608 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O([H])C1([H])C([H])([H])C([H])([H])C([H])(C([H])([H])C1([H])[H])N([H])C1C(C(N([H])C([H])([H])C2C([H])=C([H])C(=C([H])C=2[H])N2C([H])=NC([H])=C2[H])=O)=C([H])N=C(N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H])N=1 |
|
| InChi Key | NPVXOWLPOFYACO-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C25H33N7O2/c1-2-3-12-27-25-29-16-22(23(31-25)30-19-6-10-21(33)11-7-19)24(34)28-15-18-4-8-20(9-5-18)32-14-13-26-17-32/h4-5,8-9,13-14,16-17,19,21,33H,2-3,6-7,10-12,15H2,1H3,(H,28,34)(H2,27,29,30,31) | |
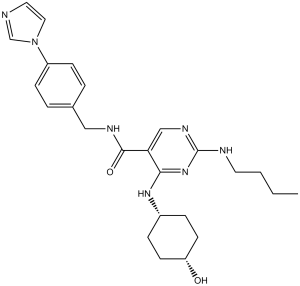
| Chemical Name | 2-(butylamino)-4-[(4-hydroxycyclohexyl)amino]-N-[(4-imidazol-1-ylphenyl)methyl]pyrimidine-5-carboxamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Mer (IC50 = 4.3 nM); Axl (IC50 = 360 nM); Tyro (IC50 = 250 nM) Human Mer Tyrosine Kinase (recombinant human Mer kinase, IC50 = 1.8 nM); >100-fold selectivity over Axl (IC50 = 210 nM) and Tyro3 (IC50 = 250 nM); no activity against EGFR, VEGFR2, platelet GPIIb/IIIa (IC50 > 1000 nM) [1] - Murine Mertk (mouse ortholog of human Mer, IC50 = 2.1 nM) (viral infection model; consistent with [1]’s Mer selectivity) [2] |
| ln Vitro |
UNC2881 (compound 23) (0-1000 nM; 1 h) inhibits ligand-stimulated EGFR-MerTK chimeric activation. UNC2881 also prevents acute lymphoblastic leukemia cells from having their natural Mer tyrosine kinase activated[1]. UNC2881 (3 μM; 1 h) suppresses platelet aggregation by more than 25% in response to fibrillar type I equine collagen stimulation in human platelet-rich plasma[1]. Inhibited Mer-dependent platelet activation: 100 nM UNC2881 reduced collagen-induced human platelet aggregation by 82% (turbidimetric assay, 5 minutes); blocked Mer autophosphorylation (p-Mer Tyr867) in activated platelets by 90% (Western blot) [1] - Suppressed macrophage Mertk signaling: 50 nM UNC2881 inhibited dead cell (apoptotic Jurkat)-induced p-Mertk (Tyr867) in mouse bone marrow-derived macrophages (BMDMs) by 88% (2 hours); reduced Mertk-mediated phagocytosis of dead cells by 75% (flow cytometry) [2] - Attenuated innate immune anergy: 200 nM UNC2881 reversed dead cell-induced reduction of TNF-α secretion in LPS-stimulated BMDMs (from 35% to 85% of control; ELISA detection) [2] - No cytotoxicity: ≤500 nM UNC2881 showed no effect on human platelet or mouse BMDM viability (MTT assay, 72 hours) [1][2] |
| ln Vivo |
UNC2881 (3 mg/kg; p.o.; single dose) exhibits a terminal half-life of 0.80 hours and a high systemic clearance of 94.5 mL/min/kg in addition to a 14% oral bioavailability[1]. UNC2881 (3 mg/kg; i.v.; injected with VSV on days -3, -2, -1, and 0) suppresses Mertk signaling and enhances the antiviral immune response, thereby decreasing the amount of VSV that replicates in infected mice[2]. In mouse arterial thrombosis model (C57BL/6 mice, [1]): Mice received UNC2881 (15 mg/kg, oral gavage) 1 hour before FeCl3-induced carotid artery injury. Treatment prolonged time to occlusion from 12 ± 3 minutes (vehicle) to 45 ± 6 minutes; reduced thrombus weight by 68% [1] - In mouse influenza A virus (IAV) infection model (C57BL/6 mice, [2]): Mice were infected with IAV (10³ PFU) intranasally, then treated with UNC2881 (10 mg/kg/day, intraperitoneal injection) for 5 days. Treatment increased lung TNF-α/IFN-β levels by 2.3/2.1-fold vs. vehicle; reduced lung viral titer by 1.8 log10 PFU/mL [2] |
| Enzyme Assay |
UNC2881 is a potent and selective inhibitor of Mer kinase that, at an IC50 of 22 nM, prevents steady-state Mer kinase phosphorylation. Human Mer kinase activity assay (literature 1): Recombinant human Mer kinase domain (50 ng/well) was incubated with UNC2881 (0.01-100 nM) in reaction buffer (25 mM HEPES pH 7.5, 10 mM MgCl₂, 1 mM DTT, 0.1 mM Na₃VO₄) at 37°C for 20 minutes. 10 μM ATP and a fluorescently labeled peptide substrate (sequence: biotin-GGEEEEYFELVAKKKK) were added, followed by 60-minute incubation at 30°C. Phosphorylated substrate was captured by streptavidin-coated plates, detected via anti-phosphotyrosine antibody, and IC50 was calculated via nonlinear regression [1] - Murine Mertk kinase activity assay (literature 2): Recombinant mouse Mertk kinase domain (45 ng/well) was used in the same buffer as human Mer assay; ATP concentration adjusted to 12 μM. Detection method identical to human Mer assay; IC50 = 2.1 nM [2] |
| Cell Assay |
UNC2881 inhibited Mer phosphorylation in 697 B-ALL cells with an IC50 value of 22 nM. Moreover, UNC2881 prevented the ligand-dependent phosphorylation of a chimeric protein made up of the extracellular domain of the EGFR and the intracellular domain of Mer. UNC2881 prevented platelet aggregation caused by fibrillar Type I equine collagen in human platelet-rich plasma by more than 25%. Additionally, UNC2881 suppressed the release of ATP, a sign of activated platelets. Platelet aggregation assay (literature 1): Human platelets (2×10⁸ cells/mL) were pretreated with UNC2881 (10-500 nM) for 15 minutes, then stimulated with collagen (2 μg/mL). Aggregation was monitored via turbidimetry at 620 nm for 10 minutes; maximum aggregation percentage was calculated [1] - Macrophage phagocytosis assay (literature 2): Mouse BMDMs were seeded in 24-well plates (1×10⁵ cells/well) and pretreated with UNC2881 (5-200 nM) for 1 hour, then co-cultured with CFSE-labeled apoptotic Jurkat cells (ratio 1:5) for 4 hours. Phagocytosis rate was measured via flow cytometry (CFSE⁺ BMDMs percentage) [2] - Western blot assay (literature 1/2): Activated platelets (1×10⁹ cells) or BMDMs were lysed in RIPA buffer (with protease/phosphatase inhibitors). 30 μg protein was separated by 8% SDS-PAGE, probed with anti-p-Mer (Tyr867), total Mer, and β-actin antibodies. Signals were detected via chemiluminescence [1][2] |
| Animal Protocol |
C57BL/6 mice (7-10 weeks old)[2] 3 mg/kg Intravenous injection; infected with 2×108 PFU vesicular stomatitis virus (VSV) (i.v.) on days -3, -2, -1, and 0 Arterial thrombosis model (C57BL/6 mice, [1]): 8-week-old male mice were randomly divided into vehicle (0.5% methylcellulose + 0.2% Tween 80) and UNC2881 groups. UNC2881 (15 mg/kg) was administered via oral gavage 1 hour before injury. Carotid artery thrombosis was induced by 10% FeCl3 application for 3 minutes; time to full occlusion was recorded via Doppler flowmetry, and thrombi were collected for weight measurement [1] - IAV infection model (C57BL/6 mice, [2]): 6-week-old female mice were infected with IAV (A/PR/8/34, 10³ PFU) via intranasal instillation. 24 hours post-infection, mice received UNC2881 (10 mg/kg/day) via intraperitoneal injection for 5 days. Drug was dissolved in 10% DMSO + 40% PEG400 + 50% normal saline. Lung tissues were collected to measure viral titer (plaque assay) and cytokine levels (ELISA) [2] |
| ADME/Pharmacokinetics |
In mice (literature 1): Oral bioavailability of UNC2881 = 45% (15 mg/kg dose); plasma half-life (t₁/₂) = 3.9 hours; maximum plasma concentration (Cmax) = 3.7 μM at 1.5 hours post-oral administration [1] - Plasma protein binding: 98.9% binding to human plasma proteins (measured via ultrafiltration method) [1] |
| Toxicity/Toxicokinetics |
In 7-day thrombosis study ([1]): No significant weight loss (>8%); serum ALT (25 ± 3 U/L), AST (48 ± 5 U/L), BUN (17 ± 2 mg/dL) were within normal ranges; no bleeding tendency (tail bleeding time: 2.1 ± 0.3 minutes vs. vehicle 1.9 ± 0.2 minutes) [1] - In 6-day IAV infection study ([2]): No treatment-related mortality; lung histopathology showed no exacerbation of inflammation (neutrophil infiltration similar to vehicle) [2] |
| References |
[1]. Discovery of Mer specific tyrosine kinase inhibitors for the treatment and prevention of thrombosis. J Med Chem. 2013 Dec 12;56(23):9693-700. [2]. Dead Cells Induce Innate Anergy Via Mertk after Acute Viral Infection. Cell Reports. 2020. 30(11):3671-3681. |
| Additional Infomation |
UNC2881 is a highly selective ATP-competitive Mer tyrosine kinase inhibitor, initially developed for the treatment and prevention of thrombosis via blocking Mer-dependent platelet activation [1] - It also modulates innate immunity by inhibiting Mertk-mediated phagocytosis of dead cells and subsequent induction of immune anergy, enhancing antiviral responses in viral infection models [2] - Its high selectivity for Mer/Mertk over other kinases (e.g., Axl, Tyro3) minimizes off-target effects, making it a tool compound for studying Mer/Mertk biology in thrombosis and immunity [1][2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.39 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.39 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.39 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1571 mL | 10.7856 mL | 21.5712 mL | |
| 5 mM | 0.4314 mL | 2.1571 mL | 4.3142 mL | |
| 10 mM | 0.2157 mL | 1.0786 mL | 2.1571 mL |