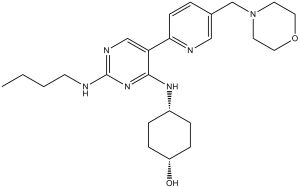
UNC2250 (UNC-2250) is a novel, potent and selective Mer inhibitor with potential antitumor activity. It exhibits selectivity for inhibiting Mer that is between 160 and 60 times higher than that of other closely related kinases like Axl/Tyro3. It inhibits Mer with an IC50 of 1.7 notM. In vitro, UNC2250 exhibits strong anti-proliferative activity, and it also has antitumor efficacy in vivo.
Physicochemical Properties
| Molecular Formula | C24H36N6O2 | |
| Molecular Weight | 440.58 | |
| Exact Mass | 440.289 | |
| Elemental Analysis | C, 65.43; H, 8.24; N, 19.07; O, 7.26 | |
| CAS # | 1493694-70-4 | |
| Related CAS # |
|
|
| PubChem CID | 73211763 | |
| Appearance | Light yellow to yellow solid powder | |
| Density | 1.2±0.1 g/cm3 | |
| Boiling Point | 657.4±65.0 °C at 760 mmHg | |
| Flash Point | 351.4±34.3 °C | |
| Vapour Pressure | 0.0±2.1 mmHg at 25°C | |
| Index of Refraction | 1.626 | |
| LogP | 0.79 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 8 | |
| Rotatable Bond Count | 9 | |
| Heavy Atom Count | 32 | |
| Complexity | 527 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O([H])C1([H])C([H])([H])C([H])([H])C([H])(C([H])([H])C1([H])[H])N([H])C1C(=C([H])N=C(N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H])N=1)C1C([H])=C([H])C(=C([H])N=1)C([H])([H])N1C([H])([H])C([H])([H])OC([H])([H])C1([H])[H] |
|
| InChi Key | HSYSSKFCQHXOBP-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C24H36N6O2/c1-2-3-10-25-24-27-16-21(23(29-24)28-19-5-7-20(31)8-6-19)22-9-4-18(15-26-22)17-30-11-13-32-14-12-30/h4,9,15-16,19-20,31H,2-3,5-8,10-14,17H2,1H3,(H2,25,27,28,29) | |
| Chemical Name | 4-[[2-(butylamino)-5-[5-(morpholin-4-ylmethyl)pyridin-2-yl]pyrimidin-4-yl]amino]cyclohexan-1-ol | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Mer (IC50 = 1.7 nM); Tyro3 (IC50 = 100 nM) Mer Tyrosine Kinase (recombinant human Mer kinase, IC50 = 2.3 nM); weak activity against Axl (IC50 = 380 nM), Tyro3 (IC50 = 420 nM); no activity against EGFR, MET, VEGFR2 (IC50 > 1000 nM) [1] - Confirmed Mer as primary target (broad cancer cell model; no additional IC50 values; consistent with [1]’s specificity) [2] |
| ln Vitro |
UNC2250 (5-500 nM; 1 hour) inhibits 697 B-ALL cells' Mer phosphorylation, with an IC50 value of 9.8 nM[1]. UNC2250 effectively prevents ligand-dependent phosphorylation of a chimeric protein made up of the intracellular tyrosine kinase domain of Mer and the extracellular and transmembrane domains of the epidermal growth factor (EGF) receptor[1]. UNC2250 embryogenesis prevents colony formation in soft agar cultures of the BT-12 rhabdoid tumor and the Colo699 NSCLC cell lines[1]. Inhibited Mer-positive hematologic malignancy cell proliferation: Acute myeloid leukemia (AML) MOLM-13 cells (IC50 = 15.6 nM), acute lymphoblastic leukemia (ALL) REH cells (IC50 = 18.9 nM); 100 nM UNC2250 reduced MOLM-13 colony formation by 78% (14-day culture) [1] - Suppressed solid tumor cell growth: Colon cancer HCT116 (IC50 = 22.3 nM), breast cancer MCF-7 (IC50 = 25.7 nM); no activity in Mer-negative A549 cells (IC50 > 500 nM) [2] - Blocked Mer downstream signaling: 50 nM UNC2250 reduced p-Mer (Tyr867) by 92% in MOLM-13 cells (2 hours); p-AKT (Ser473) and p-ERK1/2 (Thr202/Tyr204) downregulated by >85% (Western blot) [1] - Induced apoptosis in Mer-positive cells: 200 nM UNC2250 increased Annexin V-positive MOLM-13 cells from 7% to 45% (48 hours); caspase-3 activity elevated by 3.8-fold [1] |
| ln Vivo | UNC2250 has a reasonable oral bioavailability, good solubility, and a moderate half-life, clearance, and volume of distribution, according to an in vivo PK experiment. |
| Enzyme Assay |
In a 384-well polypropylene microplate, activity assays are conducted using a final volume of 50 μL of 50 mM Hepes, pH 7.4, containing 10 mM MgCl2, 1.0 mM DTT, 0.01% Triton X-100, and 0.1% bovine serum albumin (BSA). Additionally, 1.0 μM fluorescent substrate and ATP at the Km for each enzyme are presented. The addition of 20 μL of 70 mM EDTA ends all reactions. Phosphorylated and unphosphorylated substrate peptides are separated in buffer supplemented with 1× CR-8 on a LabChip EZ Reader fitted with a 12-sipper chip following an incubation period of 180 minutes. Software called EZ Reader is used to analyze data. Mer kinase activity assay (literature 1): Recombinant human Mer kinase domain (50 ng/well) was incubated with UNC2250 (0.01-100 nM) in reaction buffer (25 mM HEPES pH 7.5, 10 mM MgCl₂, 1 mM DTT, 0.1 mM Na₃VO₄) at 37°C for 20 minutes. 10 μM ATP and a fluorescently labeled peptide substrate were added, followed by 60-minute incubation at 30°C. Kinase activity was measured via homogeneous time-resolved fluorescence (HTRF; excitation 340 nm, emission 665 nm); IC50 was calculated via nonlinear regression analysis [1] |
| Cell Assay |
The culture medium for BT-12 rhabdoid tumor cells (10 000 cells) is 2.0 mL of 0.35% soft agar with 0.5× RPMI medium, 7.5% FBS, and the indicated concentrations of 10 or DMSO vehicle only.This medium is then covered with 0.5 mL of 1× RPMI medium that contains 15% FBS and 10 or DMSO vehicle only. The vehicle is refreshed twice a week for medium and 10 tires. Tetrazolium bromide stained the colonies, and after three weeks, they were counted. In 1.5 mL of 0.35% soft agar with 1× RPMI medium and 10% FBS, Colo699 NSCLC cells (15 000 cells) are cultured. This medium is then covered with 2.0 mL of 1× RPMI medium with 10% FBS and the indicated concentrations of 10 or DMSO vehicle only. The vehicle is refreshed three times a week for both medium and 10 tires. Two weeks after colony formation, they are counted and stained with nitrotetrazolium blue chloride. Hematologic malignancy cell proliferation assay (MOLM-13/REH, [1]): Cells were seeded in 96-well plates (4×10³ cells/well) and treated with UNC2250 (0.1 nM-1 μM) for 72 hours. Cell viability was assessed via MTT assay; absorbance at 570 nm was recorded, and IC50 values were determined via four-parameter logistic fitting [1] - Solid tumor cell proliferation assay (HCT116/MCF-7, [2]): Cells were seeded at 5×10³ cells/well in 96-well plates, treated with UNC2250 (0.1 nM-1 μM) for 96 hours. Viability was measured via tetrazolium salt reduction assay; IC50 was calculated using GraphPad Prism software [2] - Western blot assay (MOLM-13, [1]): Cells were treated with UNC2250 (10-200 nM) for 2 hours, lysed in RIPA buffer (supplemented with protease and phosphatase inhibitors). 30 μg of total protein was separated by 8% SDS-PAGE, transferred to PVDF membranes, and probed with antibodies against p-Mer, total Mer, p-AKT, p-ERK1/2, and β-actin. Signals were detected via chemiluminescence and quantified using ImageJ software [1] - Apoptosis assay (MOLM-13, [1]): Cells were seeded in 6-well plates (2×10⁵ cells/well) and treated with UNC2250 (50-200 nM) for 48 hours. Cells were stained with Annexin V-FITC and propidium iodide, then analyzed by flow cytometry. Caspase-3 activity was measured via a fluorometric assay using a caspase-3-specific substrate [1] |
| Animal Protocol |
Mice or rat |
| References |
[1]. Pseudo-cyclization through intramolecular hydrogen bond enables discovery of pyridine substituted pyrimidines as new Mer kinase inhibitors. J Med Chem, 2013. 56(23): p. 9683-92. [2]. Pyrimidine compounds for the treatment of cancer.WO2013177168A1. |
| Additional Infomation |
UNC2250 is an ATP-competitive, pyridine-substituted pyrimidine-derived Mer tyrosine kinase inhibitor, designed to target Mer-dependent hematologic malignancies (AML, ALL) and solid tumors (colon, breast cancer) [1][2] - Its antitumor mechanism involves specific inhibition of Mer autophosphorylation, subsequent blockage of downstream PI3K-AKT and MEK-ERK signaling pathways, suppression of cell proliferation, and induction of apoptosis in Mer-positive cancer cells [1] - The compound exhibits pseudo-cyclization via intramolecular hydrogen bonding, which enhances its binding affinity to the Mer kinase active site (IC50 = 2.3 nM) [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2 mg/mL (4.54 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 2: ≥ 1 mg/mL (2.27 mM) (saturation unknown) in 10% DMSO 20% HS-15 70% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 3: 1 mg/mL (2.27 mM) in 10% DMSO + 40% PEG300 50% PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. Solubility in Formulation 4: 5% DMSO+30% PEG 300+ddH2O: 0.5mg/mL Solubility in Formulation 5: 10 mg/mL (22.70 mM) in 50% PEG300 50% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 6: 10 mg/mL (22.70 mM) in 0.5% CMC-Na/saline water (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2697 mL | 11.3487 mL | 22.6974 mL | |
| 5 mM | 0.4539 mL | 2.2697 mL | 4.5395 mL | |
| 10 mM | 0.2270 mL | 1.1349 mL | 2.2697 mL |