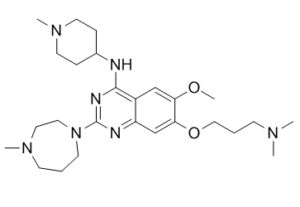
UNC0321 (UNC-0321) is a novel, potent and selective G9a histone methyltransferase inhibitor with anticancer activity. It inhibits G9a with Ki of 63 pM, UNC0321 was discovered by optimizing the 7-dimethylaminopropoxy side chain on the basis of the structural insights revealed by UNC0224-G9a cocrystal structure. UNC0321 is the first G9a inhibitor with picomolar potency.
Physicochemical Properties
| Molecular Formula | C27H45N7O3 |
| Molecular Weight | 515.6913 |
| Exact Mass | 515.358 |
| CAS # | 1238673-32-9 |
| PubChem CID | 46901937 |
| Appearance | White to yellow solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 664.7±65.0 °C at 760 mmHg |
| Flash Point | 355.8±34.3 °C |
| Vapour Pressure | 0.0±2.0 mmHg at 25°C |
| Index of Refraction | 1.582 |
| LogP | 0.36 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 37 |
| Complexity | 652 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | AULLUGALUBVBDD-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C27H45N7O3/c1-31(2)15-16-36-17-18-37-25-20-23-22(19-24(25)35-5)26(28-21-7-11-33(4)12-8-21)30-27(29-23)34-10-6-9-32(3)13-14-34/h19-21H,6-18H2,1-5H3,(H,28,29,30) |
| Chemical Name | 7-(2-(2-(dimethylamino)ethoxy)ethoxy)-6-methoxy-2-(4-methyl-1,4-diazepan-1-yl)-N-(1-methylpiperidin-4-yl)quinazolin-4-amine |
| Synonyms | UNC0321, CHEBI:785916, NCGC0018778901, UNC-0321, CHEMBL1214066, UNC 0321, KB81388 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Compound 29 (UNC0321) has IC50 values of 9 nM, 6 nM, 15 nM, and 23 nM, respectively, which inhibit the activities of G9a ECSD, G9a CLOT, GLP ECSD, and GLP CLOT. In HUVECs in MDA-MB-231 cells, UNC0321 (Compound 3) decreases Rab4 expression. Rab4 expression is markedly suppressed in HUVEC by UNC0321 (200 pM; 48 h) [3]. |
| Cell Assay |
Western Blot Analysis[3] Cell Types: HUVEC. Level of H3K9me2, IC50 value is 11 µM[2]. Tested Concentrations: 50 pM, 100 pM, 200 pM. Incubation Duration: 48 hrs (hours). Experimental Results: Glucose-induced reduction in Cleared Caspase3 and Bax expression. Release glucose from inhibiting the expression of Bcl-2, Caspase3, p-AKT, p-mTOR and p70. Promote proliferation and inhibit apoptosis by inhibiting Rab4. Apoptosis analysis[3] Cell Types: HUVEC. Tested Concentrations: 50 pM, 100 pM, 200 pM. Incubation Duration: 48 hrs (hours). Experimental Results: Inhibition of glucose-induced apoptosis. Cell proliferation assay [3] Cell Types: HUVEC. Tested Concentrations: 50 pM, 100 pM, 200 pM. Incubation Duration: 48 hrs (hours). Experimental Results: The inhibitory effect of glucose on HUVEC proliferation was relieved. |
| References |
[1]. Protein lysine methyltransferase G9a inhibitors: design, synthesis, and structure activity relationships of 2,4-diamino-7-aminoalkoxy-quinazolines. J Med Chem. 2010 Aug 12;53(15):5844-57. [2]. Optimization of cellular activity of G9a inhibitors 7-aminoalkoxy-quinazolines. J Med Chem. 2011 Sep 8;54(17):6139-50. [3]. Nie J. UNC0321 inhibits high glucose induced apoptosis in HUVEC by targeting Rab4. Biomed Pharmacother. 2020 Nov;131:110662. |
| Additional Infomation | 7-[2-[2-(dimethylamino)ethoxy]ethoxy]-6-methoxy-2-(4-methyl-1,4-diazepan-1-yl)-N-(1-methyl-4-piperidinyl)-4-quinazolinamine is a member of quinazolines. |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 31 mg/mL (~60.11 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.03 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.03 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.03 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9391 mL | 9.6957 mL | 19.3915 mL | |
| 5 mM | 0.3878 mL | 1.9391 mL | 3.8783 mL | |
| 10 mM | 0.1939 mL | 0.9696 mL | 1.9391 mL |