Physicochemical Properties
| Molecular Formula | C11H11CL2N3O5S |
| Molecular Weight | 368.193139314651 |
| Exact Mass | 366.979 |
| CAS # | 178908-09-3 |
| PubChem CID | 9842188 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 0.2 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 22 |
| Complexity | 567 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | BGJHUMNCODFLCH-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H11Cl2N3O5S/c1-22(20,21)16(2-3-17)9-7(13)5(12)4-6-8(9)15-11(19)10(18)14-6/h4,17H,2-3H2,1H3,(H,14,18)(H,15,19) |
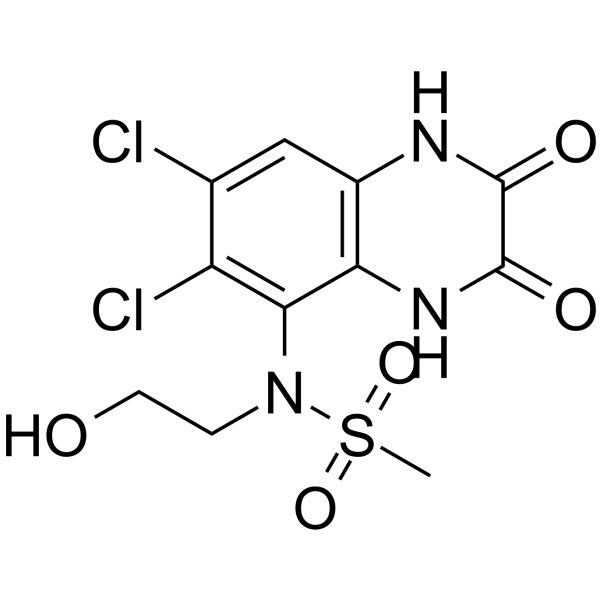
| Chemical Name | N-(6,7-dichloro-2,3-dioxo-1,4-dihydroquinoxalin-5-yl)-N-(2-hydroxyethyl)methanesulfonamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
Caco-2 Permeability: UK-240,455 showed poor flux across a Caco-2 cell monolayer. The apparent permeability (Papp) was 2 × 10⁻⁶ cm s⁻¹ in the apical-to-basolateral direction and 3 × 10⁻⁶ cm s⁻¹ in the basolateral-to-apical direction, suggesting it may be a substrate for active efflux transporters. [1] Plasma Protein Binding: The plasma protein binding of UK-240,455 was determined by equilibrium dialysis. Binding was 97% in rat plasma (free fraction 3%), 85% in dog plasma (free fraction 15%), and 87% in human plasma (free fraction 13%). [1] |
| ln Vivo |
An effective and highly specific N-methyl D-aspartate (NMDA) glycine site antagonist is UK-240455. Male rats were injected intravenously with UK-240455; the clearance rate was 12 mL/min/kg and the volume of distribution was 0.4 L/kg. UK-240455 having an apparent half-life of 0.4 hours and lower plasma concentrations. An study of unmodified UK-240455 in urine (0 to 24 hours) revealed that 57% of the administered dose was excreted unchanged in the urine. As a result, rats using UK-240455 have a urine clearance of 7 mL/min/kg. The study reports that the apparent elimination half-life of UK-240455 from plasma following oral treatment in male rats was 1.6 hours [1]. CNS Penetration in Rats: Following continuous intravenous infusion in rats, UK-240,455 rapidly penetrated the blood-brain barrier. The cerebrospinal fluid (CSF) to plasma concentration ratio was 4.3%, which was essentially equal to the plasma-free fraction (3%), indicating that free plasma concentration equilibrated with CSF. Brain tissue concentrations were low, with a brain-to-plasma ratio of 0.7%. [1] |
| Enzyme Assay |
pKa Determination: The ionization constant of UK-240,455 was determined by potentiometric titration. Multiple semi-aqueous solutions of the compound in methanol/water mixtures were acidified and then titrated with standardized KOH. Bjerrum difference plots were used to calculate apparent pKa values in the mixed solvent, which were then extrapolated to zero co-solvent using the Yasuda-Shedlovsky procedure. The determined pKa was 7.9. [1] Log D7.4 Determination: The log D7.4 (octanol/water) was determined by mixing a solution of UK-240,455 in phosphate buffer (pH 7.4) with an equal volume of octanol. After mixing and phase separation, the concentration in each phase was analyzed by HPLC. The determined log D7.4 was 0.1. [1] |
| Cell Assay |
Caco-2 Cell Permeability Assay: Caco-2 cells were grown to form confluent monolayers in 24-well plates. Before the assay, monolayers were washed. UK-240,455 (25 µM in transport buffer) was added to either the apical or basolateral donor compartment. After a 2-hour incubation at 37°C, samples were taken from the acceptor compartment, mixed with acetonitrile, and analyzed by LC-MS/MS. Apparent permeability (Papp) was calculated based on the amount transported, initial concentration, time, and surface area. Membrane integrity was verified using Lucifer yellow. [1] |
| Animal Protocol |
Rat IV and Oral PK: Male Sprague-Dawley rats received UK-240,455 via tail vein injection (2 mg/kg dissolved in sterile saline) or oral gavage (2 mg/kg dissolved in water). Blood samples were collected at multiple time points post-dose for plasma analysis. Urine was also collected over 24 hours from separate groups. [1] Rat CNS Distribution Study: Male Sprague-Dawley rats with jugular vein catheters received a loading bolus of UK-240,455 (2.5 mg/kg in saline) followed by a continuous infusion (4.5 mg/kg/h). At specified time points, animals were sacrificed, and blood, cerebrospinal fluid (via cisterna magna puncture), and whole brains were collected for analysis. [1] Rat Biliary Excretion: Anesthetized male Sprague-Dawley rats underwent bile duct cannulation. UK-240,455 (1 mg in saline) was administered via tail vein, and bile was collected for 2 hours. [1] Rat Whole-Body Autoradiography (WBA): Male Lister Hooded rats received a single intravenous dose of [¹⁴C]-UK-240,455 (10 mg/kg, ~100 µCi/animal) in saline. Animals were sacrificed at 0.1, 1, 24, and 48 hours post-dose, frozen, and sagittal sections were prepared using a cryomicrotome. Sections were apposed to phosphor plates for imaging and quantitative analysis using calibrated radioactivity standards. [1] Rat Disposition/Mass Balance: Male and female Sprague-Dawley rats received a single intravenous dose of [¹⁴C]-UK-240,455 (2 mg/kg, ~20 µCi/animal) in saline. Animals were housed in metabolism cages, and urine and feces were collected daily for 6 days. On day 6, animals were sacrificed and carcasses processed. Radioactivity in excreta and carcass was quantified by liquid scintillation counting or combustion. Urine and fecal extracts were analyzed by radio-HPLC. [1] Dog IV and Oral PK: Male and female beagle dogs received UK-240,455 via a 15-minute intravenous infusion into the saphenous vein (1 mg/kg in saline). Blood samples were collected via an indwelling catheter. In a separate phase, the same dogs received an oral dose by gavage (1 mg/kg in water) with blood sampling. Urine was collected over 24 hours after IV dosing from female dogs. [1] Dog Disposition/Mass Balance: One male and one female beagle dog received a single intravenous dose of [¹⁴C]-UK-240,455 (2 mg/kg, ~85 µCi/animal) via a 15-minute infusion. Animals were kept in metabolism cages, and urine and feces were collected daily for 6 days. Blood samples were also collected at specified times. Radioactivity in excreta and blood was quantified. Urine and fecal extracts were analyzed by radio-HPLC. [1] |
| ADME/Pharmacokinetics |
Absorption: Oral bioavailability of UK-240,455 was low in rats (7%) and moderate in dogs (31 ± 16%), attributed to poor intestinal permeability and potential efflux transport. The apparent elimination half-life after oral dosing was longer than after IV dosing, indicating absorption-rate limited kinetics. [1] Distribution: UK-240,455 has a low to moderate volume of distribution (Vss): 0.4 L/kg in rats, 0.8 ± 0.3 L/kg in dogs, and 0.8 ± 0.1 L/kg in humans. This limited distribution correlates with its polarity and plasma protein binding. It rapidly penetrates the blood-brain barrier, with CSF concentrations equilibrating with free plasma concentrations. Brain tissue concentrations, however, are very low (0.7% of plasma). Whole-body autoradiography in rats showed widespread but low tissue concentrations shortly after dosing, with the highest levels in excretory organs (kidney, liver, bile, intestines). [1] Metabolism: UK-240,455 is primarily excreted unchanged. In rats and dogs, no major circulating metabolites were identified; the drug was recovered unchanged in urine and feces (>95%). In humans, a glucuronide conjugate of UK-240,455 was detected in plasma. This metabolite was not observed in rat or dog plasma. [1] Excretion: Following intravenous administration, UK-240,455 is predominantly excreted unchanged in urine (57-61% of dose in rats, dogs, and humans) and feces (remainder of dose). Biliary excretion is significant in rats (23.2 ± 6.5% of dose excreted unchanged in bile over 2 hours). Clearance involves active processes: renal tubular secretion (urinary clearance exceeds glomerular filtration rate) and active hepatobiliary transport. [1] Key PK Parameters (IV): Elimination half-life (t₁/₂): 0.4 h in rats, 1.1 ± 0.4 h in dogs, 1.4 ± 0.2 h in humans. Systemic clearance (CL): 12 mL/min/kg in rats, 13 ± 3 mL/min/kg in dogs, 6 ± 1 mL/min/kg in humans. Unbound clearance (CLu) showed an allometric relationship across species. [1] Sex Differences: A sex difference was observed in rats, with female rats having approximately 30-45% higher clearance than males, attributed to differences in active renal tubular secretion. No sex difference was observed in dogs. [1] |
| Toxicity/Toxicokinetics |
The paper does not provide specific data on acute toxicity (e.g., LD₅₀), organ toxicity, or drug-drug interactions. The short half-life was a design feature to allow rapid cessation of pharmacological activity and remediation of potential side effects. The formation of a human-specific glucuronide metabolite not seen in preclinical species (rat, dog) was noted as a potential development consideration. [1] |
| References | [1]. Webster R, et al. Pharmacokinetics and disposition of a novel NMDA glycine site antagonist (UK-240,455) in rats, dogs and man. Xenobiotica. 2003 May;33(5):541-60 |
| Additional Infomation |
UK-240,455 ((+)-6,7-dichloro-5-(N-(2-hydroxyethyl)methanesulphonamido)-2,3(1H,4H)-quinoxalinedione) is a novel NMDA receptor glycine site antagonist developed for the potential treatment of stroke and head trauma. It was designed to have a short half-life to facilitate rapid offset of action and management of potential CNS-related side effects. Its physicochemical properties include a pKa of 7.9 and a log D7.4 of 0.1, indicating it is a weakly acidic, polar molecule. The compound shows optimal passive diffusion across the blood-brain barrier (free concentration in plasma equals CSF concentration) despite poor intestinal permeability. Allometric scaling of unbound clearance and volume of distribution from animals predicted human pharmacokinetics reasonably well. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7160 mL | 13.5799 mL | 27.1599 mL | |
| 5 mM | 0.5432 mL | 2.7160 mL | 5.4320 mL | |
| 10 mM | 0.2716 mL | 1.3580 mL | 2.7160 mL |