Physicochemical Properties
| Molecular Formula | C15H14IN3O6 |
| Molecular Weight | 516.65614 |
| Exact Mass | 458.993 |
| CAS # | 569371-10-4 |
| Related CAS # | UBP301 hydrochloride |
| PubChem CID | 6604913 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 0.473 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 25 |
| Complexity | 614 |
| Defined Atom Stereocenter Count | 1 |
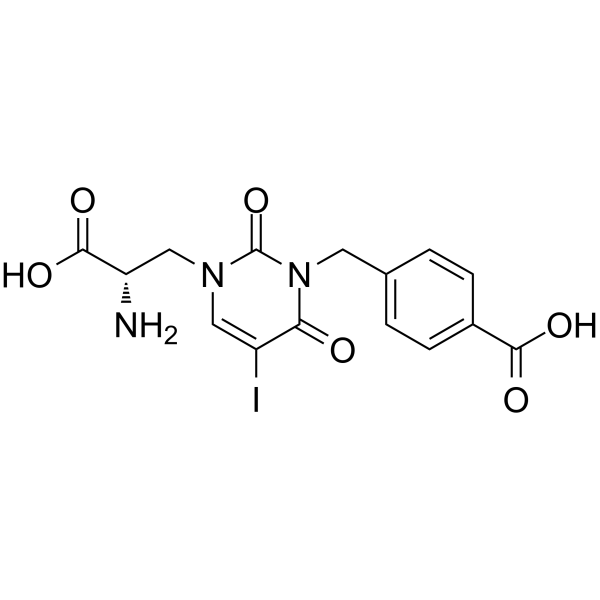
| SMILES | C1=C(C=CC(=C1)C(=O)O)CN2C(=O)C(=CN(C[C@@H](C(=O)O)N)C2=O)I |
| InChi Key | JHSYCOCOIYSZGI-NSHDSACASA-N |
| InChi Code | InChI=1S/C15H14IN3O6/c16-10-6-18(7-11(17)14(23)24)15(25)19(12(10)20)5-8-1-3-9(4-2-8)13(21)22/h1-4,6,11H,5,7,17H2,(H,21,22)(H,23,24)/t11-/m0/s1 |
| Chemical Name | 4-[[3-[(2S)-2-amino-2-carboxyethyl]-5-iodo-2,6-dioxopyrimidin-1-yl]methyl]benzoic acid |
| Synonyms | UBP-301; 569371-10-4; UBP 301; UBP301; (AS)-alpha-AMINO-3-[(4-CARBOXYPHENYL)METHYL]-3,4-DIHYDRO-5-IODO-2,4-DIOXO-1(2H)-PYRIMIDINEPROPANOIC ACID; 4-[[3-[(2S)-2-amino-2-carboxyethyl]-5-iodo-2,6-dioxopyrimidin-1-yl]methyl]benzoic acid; CHEMBL200309; NCGC00025285-01; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
UBP301 acts as a competitive antagonist at both AMPA (GluA2 subunit, Ki = 8.2 μM) and kainate (GluK1 subunit, Ki = 2.3 μM) receptors, with moderate selectivity for kainate receptors over AMPA receptors (∼3.5-fold) [1] |
| ln Vitro |
In Vitro: [1] <|place▁holder▁no▁797|> In recombinant HEK293 cells expressing GluK1 receptors, UBP301 (10 μM) inhibited kainate-evoked currents by 92 ± 3% (IC50 = 0.9 μM), showing potent antagonism. Schild plot analysis yielded a slope of 1.02 ± 0.05, confirming competitive antagonism at the glutamate binding site. For AMPA receptors (GluA2-expressing cells), UBP301 (100 μM) reduced AMPA-induced currents by 85 ± 4% (IC50 = 32 μM), demonstrating weaker activity compared to kainate receptors. The pA2 value was 4.7 ± 0.2, indicating lower affinity [1] |
| Enzyme Assay |
Enzyme Assay: [1] Radioligand displacement assays were conducted using [3H]kainate on rat cortical membranes. Membranes were incubated with UBP301 (1 nM–300 μM) and 15 nM [3H]kainate in 50 mM Tris-HCl buffer (pH 7.4, 4°C) for 45 min. Bound ligand was separated via vacuum filtration through GF/B filters, washed with ice-cold buffer, and quantified by liquid scintillation spectrometry to calculate Ki values [1] |
| References |
[1]. Structural requirements for novel willardiine derivatives acting as AMPA and kainate receptor antagonists. Br J Pharmacol. 2003;138(6):1093-1100. |
| Additional Infomation |
Additional Info: [1] UBP301 is a 5-iodowillardiine analog designed to optimize steric bulk at the N3-position for enhanced receptor subtype discrimination. Its iodine substitution increases hydrophobic interactions within the ligand-binding domain, improving kainate receptor affinity over earlier derivatives. Serves as a key pharmacological tool for dissecting AMPA/kainate receptor contributions to excitatory neurotransmission, particularly in studies of synaptic plasticity and excitotoxicity [1] |
Solubility Data
| Solubility (In Vitro) | DMSO: 100 mg/mL (217.77 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 5 mg/mL (10.89 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 5 mg/mL (10.89 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 5 mg/mL (10.89 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9355 mL | 9.6775 mL | 19.3551 mL | |
| 5 mM | 0.3871 mL | 1.9355 mL | 3.8710 mL | |
| 10 mM | 0.1936 mL | 0.9678 mL | 1.9355 mL |