Physicochemical Properties
| Molecular Formula | C7H6O2 |
| Molecular Weight | 122.12 |
| Exact Mass | 122.036 |
| CAS # | 533-75-5 |
| PubChem CID | 10789 |
| Appearance | Light yellow to brown solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 290.1±33.0 °C at 760 mmHg |
| Melting Point | 50-52 °C(lit.) |
| Flash Point | 122.0±18.0 °C |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C |
| Index of Refraction | 1.603 |
| LogP | 0.42 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 9 |
| Complexity | 209 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | MDYOLVRUBBJPFM-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C7H6O2/c8-6-4-2-1-3-5-7(6)9/h1-5H,(H,8,9) |
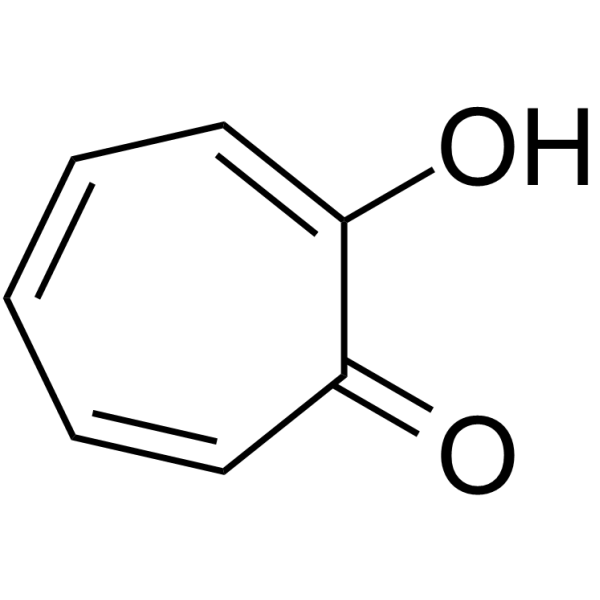
| Chemical Name | 2-hydroxycyclohepta-2,4,6-trien-1-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References |
[1]. In vitro evaluation of tropolone absorption, metabolism, and clearance. Antiviral Res. 2023 Dec;220:105762. [2]. Investigation of the activity of a novel tropolone in osteosarcoma. Drug Dev Res. 2024 Feb;85(1):e22129. [3]. Synthesis of bicyclo[5.3.0]azulene derivatives. Nat Protoc. 2009; 4(8): 1113–1117. [4]. Inhibition of mushroom tyrosinase by tropolone Phytochemistry. Volume 24, Issue 5, 1985, Pages 905-908. |
| Additional Infomation |
Tropolone is a cyclic ketone that is cyclohepta-2,4,6-trien-1-one substituted by a hydroxy group at position 2. It is a toxin produced by the agricultural pathogen Burkholderia plantarii. It has a role as a bacterial metabolite, a toxin and a fungicide. It is a cyclic ketone, an enol and an alpha-hydroxy ketone. It derives from a hydride of a cyclohepta-1,3,5-triene. A seven-membered aromatic ring compound. It is structurally related to a number of naturally occurring antifungal compounds (ANTIFUNGAL AGENTS). |
Solubility Data
| Solubility (In Vitro) |
DMSO : ≥ 100 mg/mL (~818.87 mM) H2O : ~25 mg/mL (~204.72 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (20.47 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (20.47 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (20.47 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 20 mg/mL (163.77 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 8.1887 mL | 40.9433 mL | 81.8867 mL | |
| 5 mM | 1.6377 mL | 8.1887 mL | 16.3773 mL | |
| 10 mM | 0.8189 mL | 4.0943 mL | 8.1887 mL |