Physicochemical Properties
| Molecular Formula | C₂₁H₂₄N₂O₇S |
| Molecular Weight | 448.49 |
| Exact Mass | 448.13 |
| CAS # | 113658-85-8 |
| PubChem CID | 124236 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.327g/cm3 |
| Boiling Point | 623.2ºC at 760mmHg |
| Flash Point | 330.7ºC |
| Vapour Pressure | 1.88E-15mmHg at 25°C |
| Index of Refraction | 1.571 |
| LogP | 3.072 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 31 |
| Complexity | 945 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | S1(C2=C([H])C([H])=C([H])C([H])=C2C(N1C([H])([H])C([H])([H])OC(C1=C(C([H])([H])[H])N([H])C(C([H])([H])[H])=C(C(=O)OC([H])([H])C([H])([H])[H])C1([H])C([H])([H])[H])=O)=O)(=O)=O |
| InChi Key | MCNAAGLIGWJLQX-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H24N2O7S/c1-5-29-20(25)17-12(2)18(14(4)22-13(17)3)21(26)30-11-10-23-19(24)15-8-6-7-9-16(15)31(23,27)28/h6-9,12,22H,5,10-11H2,1-4H3 |
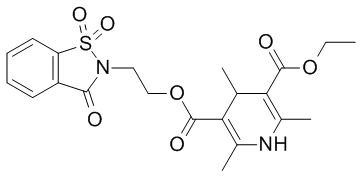
| Chemical Name | 3-O-ethyl 5-O-[2-(1,1,3-trioxo-1,2-benzothiazol-2-yl)ethyl] 2,4,6-trimethyl-1,4-dihydropyridine-3,5-dicarboxylate |
| Synonyms | PCA-4230PCA4230PCA 4230 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
One antithrombotic medication is trombodipine. After being exposed to 50 μM trombodipine, there was a noticeable growth stop. On days 3, 5, and 7 of the growth curve, the inhibition percentages of 1 μM tripodipine were 22.0±4.5, 32.0±3.0, and 29.4±6.0%, respectively (P<0.05). Trombodipine has a reversible inhibitory impact on cell growth; removal of the drug causes cell proliferation to resume [1]. During the course of four to six days of culture, the addition of trombodipine resulted in a dose-dependent reduction in cell counts. 5 μM trombodipine decreased E19P cell and human VSMC numbers by 25% and 21%, respectively, compared to control cultures at the most recent research time point, whereas 50 μM trombodipine decreased E19P and human VSMC numbers by 85% and 74%, respectively. 2]. PCA-4230 reversibly inhibited the proliferation of cultured A10 rat vascular smooth muscle cells (VSMCs). The increase in cell number was significantly reduced in the presence of 1 and 50 µM PCA-4230 over a 7-day period. [1] PCA-4230 concentration-dependently inhibited serum-induced DNA synthesis in synchronized A10 VSMCs, with an IC₅₀ value of 13 µM. This inhibition was also observed in primary rabbit aortic VSMCs. [1] Serum-induced DNA synthesis in bovine aortic endothelial cells was not significantly affected by PCA-4230 (0.5-50 µM). [1] PCA-4230 (50 µM) caused a significant drop in platelet-derived growth factor-BB (PDGF-BB)-mediated DNA synthesis in A10 VSMCs. This inhibition could not be reversed by increasing the PDGF-BB concentration. [1] The antiproliferative effect of PCA-4230 (10 µM) on serum-induced DNA synthesis was similar in magnitude to that of the dihydropyridine nifedipine at an equimolar concentration. [1] Flow cytometry analysis revealed that PCA-4230 (50 µM) blocks serum-induced cell cycle progression at the early G₀/G₁ phase in synchronized A10 VSMCs. The inhibitory effect was maximal when added with the serum stimulus but was abolished if added 12 hours after serum repletion. [1] Northern blot analysis showed that PCA-4230 (50 µM) caused a 95% decrease in serum-induced c-fos mRNA expression and a 90% decrease in c-jun mRNA expression in A10 VSMCs. [1] The inhibitory effects of PCA-4230 on cell proliferation and DNA synthesis were reversible upon drug washout. Trypan blue exclusion tests indicated no loss of cell viability (<2% dye uptake) at the concentrations tested. [1] |
| Cell Assay |
Cell Culture and Proliferation: Rat A10 vascular smooth muscle cells were cultured in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal calf serum (FCS) and antibiotics. To assess proliferation, cells were seeded at 2.5x10⁴ cells/well. After allowing attachment, they were incubated in the presence or absence of PCA-4230 for up to 7 days. Media were changed daily. Cells were detached with trypsin and counted manually using a hemocytometer. [1] DNA Synthesis (BrdU Incorporation): Cells were seeded on coverslips, allowed to attach, and then synchronized by incubation in serum-free medium with 0.2% bovine serum albumin (BSA) for 48 hours. Quiescent cells were then stimulated with 10% FCS (or 10 ng/ml PDGF-BB) in the presence or absence of various concentrations of PCA-4230 for 18 hours. 5-bromo-2'-deoxyuridine (BrdU) was added for the final 2 hours. Cells were fixed, and BrdU incorporation was detected immunocytochemically using an anti-BrdU monoclonal antibody followed by a peroxidase-conjugated secondary antibody. Diaminobenzidine was used as the chromogen. The percentage of positively stained nuclei was determined by light microscopy, counting at least 500 nuclei. [1] Cell Cycle Analysis by Flow Cytometry: Synchronized A10 VSMCs (via 48-hour serum starvation) were stimulated with 10% FCS. PCA-4230 (50 µM) was added at selected time points relative to serum repletion. At specified times, cells were harvested, washed, and fixed. Nuclear DNA was stained with propidium iodide (PI) in a buffer containing RNase and a non-ionic detergent. DNA content per cell was analyzed using a flow cytometer equipped with a 488 nm argon laser. PI fluorescence was collected, and data from 10,000 single-cell events were acquired to determine the percentage of cells in G₀/G₁, S, and G₂/M phases. [1] Oncogene Expression (Northern Blot): Synchronized A10 VSMCs were stimulated with 10% FCS for 30 minutes in the presence or absence of PCA-4230 (50 µM). Total RNA was extracted using an acid-phenol method. RNA (20 µg per sample) was separated by formaldehyde-agarose gel electrophoresis, transferred to a nylon membrane, and immobilized by UV cross-linking and baking. Membranes were pre-hybridized and then hybridized overnight at 65°C with random-primed DNA probes specific for rat c-fos or c-jun. After washing, blots were exposed to X-ray film. To control for loading, blots were re-hybridized with a β-actin probe. Signal intensity was analyzed by densitometry. [1] |
| Toxicity/Toxicokinetics |
In cell culture, trypan blue exclusion tests performed on A10 VSMCs treated with PCA-4230 showed less than 2% dye uptake, indicating no significant loss of cell viability at the concentrations used (up to 50 µM). No floating cells were observed, suggesting the inhibition was not due to cytotoxicity. [1] |
| References |
[1]. Antiproliferative effects of PCA-4230, a new antithrombotic drug, in vascular smooth muscle cells. Br J Pharmacol. 1997 Apr;120(7):1360-6. |
| Additional Infomation |
PCA-4230 is a novel 1,4-dihydropyridine derivative with antiplatelet and antithrombotic activity. It lacks the typical vasodilator effects of classical dihydropyridines due to structural modification (absence of an aryl substituent at position 4 of the dihydropyridine ring). [1] The drug inhibits vascular smooth muscle cell (VSMC) proliferation, a key process in atherosclerosis and restenosis. Its mechanism involves blocking cell cycle progression at the early G₀/G₁ phase and downregulating the expression of immediate-early genes c-fos and c-jun. [1] The antiproliferative effect appears selective for VSMCs, as endothelial cell DNA synthesis was not affected. This selectivity is considered potentially beneficial for treating vascular diseases without impairing endothelial repair. [1] The IC₅₀ for PCA-4230 on collagen-induced platelet aggregation was reported to be approximately 10 µM, which is about 10-fold lower than that of nifedipine. [1] Phase II Clinical Trials suggested beneficial effects in patients with atherosclerotic disease of the lower limbs. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2297 mL | 11.1485 mL | 22.2970 mL | |
| 5 mM | 0.4459 mL | 2.2297 mL | 4.4594 mL | |
| 10 mM | 0.2230 mL | 1.1149 mL | 2.2297 mL |