Physicochemical Properties
| Molecular Formula | C14H18N4O3.HCL |
| Molecular Weight | 326.77866 |
| Exact Mass | 326.115 |
| CAS # | 60834-30-2 |
| Related CAS # | Trimethoprim;738-70-5;Trimethoprim lactate;23256-42-0;Trimethoprim-d3;1189923-38-3;Trimethoprim sulfate;56585-33-2;Trimethoprim-13C3;1189970-95-3 |
| PubChem CID | 173769 |
| Appearance | Typically exists as solid at room temperature |
| Boiling Point | 526ºC at 760 mmHg |
| Flash Point | 271.9ºC |
| LogP | 1.919 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 22 |
| Complexity | 307 |
| Defined Atom Stereocenter Count | 0 |
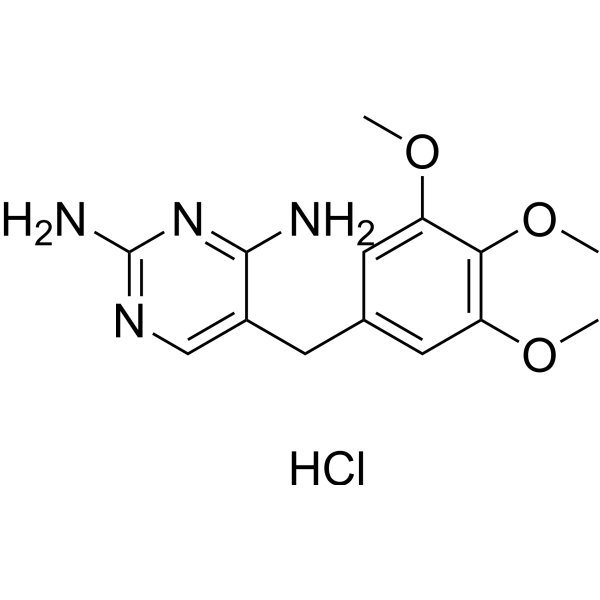
| SMILES | Cl.COC1=CC(CC2=CN=C(N)N=C2N)=CC(OC)=C1OC |
| InChi Key | YLCCEQZHUHUYPA-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C14H18N4O3.ClH/c1-19-10-5-8(6-11(20-2)12(10)21-3)4-9-7-17-14(16)18-13(9)15;/h5-7H,4H2,1-3H3,(H4,15,16,17,18);1H |
| Chemical Name | 5-[(3,4,5-trimethoxyphenyl)methyl]pyrimidine-2,4-diamine;hydrochloride |
| Synonyms | TRIMETHOPRIM HYDROCHLORIDE; Trimethoprim HCl; Trimplex; Primsol; 60834-30-2; Trimplex 200; UNII-9XE000OU9B; 9XE000OU9B; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | DHFR/Dihydrofolate reductase; Influenza A virus |
| ln Vitro | By preventing dihydrofolate reductase (DHFR) from functioning, trimethoprim disrupts the metabolism of folate and converts dihydrofolate to tetrahydrofolate (THF) [1]. In E. coli, trimethoprim (3 μg/mL; 1 h) causes significant heat shock proteins (Hsps) and protein aggregation. coli cells, indicating that protein misfolding is brought on by trimethoprim sulfate [1]. E. coli produces DnaK, DnaJ, GroEL, ClpB, and IbpA/B Hsps when treated with trimethoprim (1.5–3 μg/mL; 1 hour). Coli cells subjected to heat stress and folate [1]. |
| ln Vivo | Trimethoprim (10 mg/kg; IV; every 12 hours; 3 days) demonstrated antibacterial activity against Neisseria meningitidis, Escherichia coli, Haemophilus influenzae, and Streptococcus pneumoniae in infected mice[2]. Trimethoprim has a half-life of about an hour in full serum and a MIC value of about 1 μM against E. coli. It can be associated with thiomaltose (TM-TMP) and shows stability. Coli [2]. The injection of trimethoprim-zinc mixed suspension (10 mg/mL; 0.5 mL) lowers virus titers and increases the survival rate of chicken embryos [4]. |
| Enzyme Assay |
Influenza virus was isolated from patients and propagated in eggs. We determined viral load that infects 50% of eggs (50% egg lethal dose, ELD50). We introduced 10 ELD50 into embryonated eggs and repeated the experiments using 100 ELD50. A mixture of zinc oxide (Zn) and trimethoprim (TMP) (weight/weight ratios ranged from 0.01 to 0.3, Zn/TMP with increment of 0.1) was tested for embryo survival of the infection (n = 12 per ratio, in triplicates). Embryo survival was determined by candling eggs daily for 7 days. Controls of Zn, TMP, saline or convalescent serum were conducted in parallel. The effect of Tri-Z on virus binding to its cell surface receptor was evaluated in a hemagglutination inhibition (HAI) assay using chicken red cells. Tri-Z was prepared to concentration of 10 mg TMP and 1.8 mg Zn per ml, then serial dilutions were made. HAI effect was expressed as scores where ++++ = no effect; 0 = complete HAI effect. Results: TMP, Zn or saline separately had no effect on embryo survival, none of the embryos survived influenza virus infection. All embryos treated with convalescent serum survived. Tri-Z, at ratio range of 0.15-0.2 (optimal ratio of 0.18) Zn/TMP, enabled embryos to survive influenza virus despite increasing viral load (> 80% survival at optimal ratio). At concentration of 15 µg/ml of optimal ratio, Tri-Z had total HAI effect (scored 0). However, at clinical concentration of 5 µg/ml, Tri-Z had partial HAI effect (+ +). Conclusion: Acting on host cells, Tri-Z at optimal ratio can reduce the lethal effect of influenza A virus in chick embryo. Tri-Z has HAI effect. These findings suggest that combination of trimethoprim and zinc at optimal ratio can be provided as treatment for influenza and possibly other respiratory RNA viruses infection in man.[1] |
| Cell Assay | Trimethoprim (TMP), an inhibitor of dihydrofolate reductase, decreases the level of tetrahydrofolate supplying one-carbon units for biosynthesis of nucleotides, proteins, and panthotenate. We have demonstrated for the first time that one of the effects of the TMP action in E. coli cells is protein aggregation and induction of heat shock proteins (Hsps). TMP caused induction of DnaK, DnaJ, GroEL, ClpB, and IbpA/B Hsps. Among these Hsps, IbpA/B were most efficiently induced by TMP and coaggregated with the insoluble proteins. Upon folate stress, deletion of the delta ibpA/B operon resulted in increased protein aggregation but did not influence cell viability.[1] |
| Animal Protocol |
Animal/Disease Models: Female C3H/HeOuJ mice (transurethral infection in 50 μL suspension containing 1-2×107 CFU E. coli under 3% isoflurane) [2] Doses: 10 mg/kg Route of Administration: intravenous (iv) (iv)injection ; Once every 12 hrs (hrs (hours)); 3 days Experimental Results:Antibacterial activity against Haemophilus influenzae, Streptococcus pneumoniae, Escherichia coli and Neisseria meningitidis. The CD50 of the infected person was 150 mg/kg, 335 mg/kg, 27.5 respectively. mg/kg and 8.4 mg/kg mice. Animal/Disease Models: Fertilized eggs (H3N2 virus is injected into the amniotic membrane and allantoic cavity on day 8) [4] Doses: 10 mg/mL; 0.5 mL Route of Administration: Trimethoprim-zinc composite suspension is injected into the air sac; single dose Experimental Results: The virus titer was diminished and the survival rate of chicken embryos was improved. Survival rates peaked at a ratio of approximately 0.18 (Zn/trimethoprim). |
| ADME/Pharmacokinetics |
Absorption Steady-state concentrations are achieved after approximately 3 days of repeat administration. Average peak serum concentrations of approximately 1 µg/mL (Cmax) are achieved within 1 to 4 hours (Tmax) following the administration of a single 100mg dose. Trimethoprim appears to follow first-order pharmacokinetics, as a single 200mg dose results in serum concentrations approximately double that of a 100mg dose. The steady-state AUC of orally administered trimethoprim is approximately 30 mg/L·h. Route of Elimination Approximately 10-20% of an ingested trimethoprim dose is metabolized, primarily in the liver, while a large portion of the remainder is excreted unchanged in the urine. Following oral administration, 50% to 60% of trimethoprim is excreted in the urine within 24 hours, approximately 80% of which is unchanged parent drug. Volume of Distribution Trimethoprim is extensively distributed into various tissues following oral administration. It distributes well into sputum, middle ear fluid, and bronchial secretions. Trimethoprim distributes efficiently into vaginal fluids, with observed concentrations approximately 1.6-fold higher than those seen in the serum. It may pass the placental barrier and into breast milk. Trimethoprim is also sufficiently excreted in the feces to markedly reduce and/or eliminate trimethoprim-susceptible fecal flora. Clearance Following oral administration, the renal clearance of trimethoprim has been variably reported between 51.7 - 91.3 mL/min. Trimethoprim is widely distributed into body tissues & fluids including the aqueous humor, middle ear fluid, saliva, lung tissue, sputum, seminal fluid, prostatic tissue & fluid, vaginal secretions, bile, bone, & /cerebrospinal fluid/. The apparent volume of distribution of trimethoprim in adults with normal renal function ranges from 100-120 l. ... Trimethoprim is 42-46% bound to plasma proteins. Trimethoprim readily crosses the placenta, & amniotic fluid concns are reported to be 80% of concurrent maternal serum concns. Only small amounts of trimethoprim are excreted in feces via biliary elimination. Trimethoprim may be moderately removed by hemodialysis. Trimethoprim is readily & almost completely absorbed from the GI tract. Peak serum concns of approx 1, 1.6, & 2 ug/ml are reached in 1-4 hr after single 100-, 160-, & 200 mg oral doses of trimethoprim. Following multiple-dose oral admin, steady-state peak serum concns of trimethoprim usually are 50% greater than those obtained after single-dose admin of the drug. Steady-state serum concns range from 1.2-3.2 ug/ml following oral admin of 160 mg of trimethoprim every 12 hr in adults with renal function. Rapidly and widely distributed to various tissues and fluids, including kidneys, liver, spleen, bronchial secretions, saliva, and seminal fluid. Trimethoprim has also been demonstrated in bile; aqueous humor; bone marrow and spongy, but not compact, bone. View More
Metabolism / Metabolites
Biological Half-Life Trimethoprim half-life ranges from 8-10 hours, but may be prolonged in patients with renal dysfunction. Trimethoprim has a serum half-life of approx 8-11 hr in adults with normal renal function. In adults with creatinine clearances of 10-30 or 0-10 ml/min, serum half-life of the drug may incr to 15 hr or >26 hr, respectively. Trimethoprim serum half-lives of about 7.7 & 5.5 hr have been reported in children <1 yr of age & between 1 & 10 yr of age, respectively. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation
◉ Summary of Use during Lactation Because of the low levels of trimethoprim in breastmilk, amounts ingested by the infant are small and would not be expected to cause any adverse effects in breastfed infants. ◉ Effects in Breastfed Infants In one study, no adverse effects were noted in infants during 4 days of maternal therapy with co-trimoxazole. In a telephone follow-up study, 12 nursing mothers reported taking co-trimoxazole (dosage unspecified). Two mothers reported poor feeding in their infants. Diarrhea was not reported among the exposed infants. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Interactions Concomitant admin of trimethoprim or trimethoprim/sulfamethoxazole with methotrexate may incr bone marrow suppression, probably as an additive antifolate effect. Concurrent use with trimethoprim or use of trimethoprim between courses of other folic acid antagonists, such as methotrexate or pyrimethamine, is not recommended because of the possibility of an increased incidence of megaloblastic anemia. Trimethoprim may inhibit the metab of phenytoin, increasing the half-life of phenytoin by up to 50% & decreasing its clearance by 30% Non-Human Toxicity Values LD50 Mice oral 7000 mg/kg LD50 Rat oral 200 mg/kg LD50 Mouse oral 3960 mg/kg Protein Binding Trimethoprim is 44% bound to plasma proteins, though the specific proteins to which it binds have not been elucidated. |
| References |
[1]. Trimethoprim induces heat shock proteins and protein aggregation in E. coli cells. Curr Microbiol, 2003. 47(4): p. 286-9. [2]. Trimethoprim: a review of its antibacterial activity, pharmacokinetics and therapeutic use in urinary tract infections. Drugs, 1982. 23(6): p. 405-30. [3]. A Trimethoprim Conjugate of Thiomaltose Has Enhanced Antibacterial Efficacy In Vivo. Bioconjug Chem. 2018 May 16;29(5):1729-1735.\. [4]. El Habbal MH. Combination therapy of zinc and trimethoprim inhibits infection of influenza A virus in chick embryo. Virol J. 2021 Jun 3;18(1):113. |
| Additional Infomation | See also: Trimethoprim (has active moiety). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0602 mL | 15.3008 mL | 30.6016 mL | |
| 5 mM | 0.6120 mL | 3.0602 mL | 6.1203 mL | |
| 10 mM | 0.3060 mL | 1.5301 mL | 3.0602 mL |