Physicochemical Properties
| Molecular Formula | C10H12O |
| Molecular Weight | 148.2017 |
| Exact Mass | 148.089 |
| CAS # | 4180-23-8 |
| Related CAS # | 26795-32-4 |
| PubChem CID | 637563 |
| Appearance | White to yellow <20°C powder,>21°C liquid |
| Density | 0.9875 |
| Boiling Point | 234-237ºC |
| Melting Point | 23ºC |
| Flash Point | 90ºC |
| Index of Refraction | 1.559-1.562 |
| LogP | 2.728 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 11 |
| Complexity | 121 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O(C([H])([H])[H])C1C([H])=C([H])C(/C(/[H])=C(\[H])/C([H])([H])[H])=C([H])C=1[H] |
| InChi Key | RUVINXPYWBROJD-ONEGZZNKSA-N |
| InChi Code | InChI=1S/C10H12O/c1-3-4-9-5-7-10(11-2)8-6-9/h3-8H,1-2H3/b4-3+ |
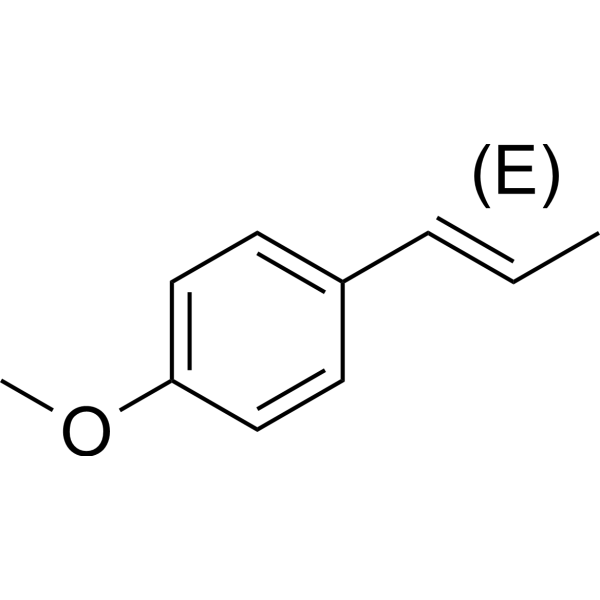
| Chemical Name | 1-methoxy-4-[(E)-prop-1-enyl]benzene |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Trans-Anethole binds to estrogen receptor (ER) and exerts estrogenic activity, with an EC50 of 12.5 μM in recombinant yeast estrogen screen[1] - Trans-Anethole regulates key enzymes of carbohydrate metabolism, including hexokinase (HK), phosphofructokinase (PFK), and glucose-6-phosphatase (G6Pase); it increases HK and PFK activities by 35% and 42%, respectively, and decreases G6Pase activity by 28% in streptozotocin (STZ)-induced diabetic rats[5] - Trans-Anethole inhibits the growth of Aspergillus flavus (fungal target: cell membrane integrity), with a minimum inhibitory concentration (MIC) of 0.5 mg/mL[7] |
| ln Vitro |
- Estrogenic activity in recombinant yeast: Trans-Anethole induced β-galactosidase activity in ER-transfected recombinant yeast cells in a concentration-dependent manner. At 12.5 μM (EC50), the enzyme activity was 50% of the maximum response; at 100 μM, the activity was 8.2-fold higher than the blank control, confirming its estrogenic activity[1] - Cytotoxic and xenoestrogenic effects on cells: In in vitro biotransformation experiments with isolated rat hepatocytes, 100 μM Trans-Anethole was metabolized to products that increased the proliferation rate of MCF-7 human breast cancer cells by 25% (xenoestrogenic effect). Without biotransformation, 100 μM Trans-Anethole had no significant effect on MCF-7 proliferation. Additionally, 200 μM Trans-Anethole reduced the viability of isolated rat hepatocytes by 18% after 24-hour incubation (cytotoxicity)[2] - Antifungal activity against Aspergillus flavus: Trans-Anethole inhibited spore germination and mycelial growth of Aspergillus flavus in vitro. At 0.5 mg/mL (MIC), it completely inhibited spore germination; at 1.0 mg/mL, it suppressed mycelial growth by 85% and reduced aflatoxin production by 90% compared to the control[7] - Regulation of carbohydrate-metabolizing enzymes: In liver homogenates from STZ-induced diabetic rats, Trans-Anethole (100 μg/mL, in vitro incubation) increased HK activity by 30% and PFK activity by 38%, while decreasing G6Pase activity by 25% compared to the diabetic control homogenate[5] |
| ln Vivo |
- Protective effect in experimental thrombosis: Male Wistar rats were subjected to ferric chloride-induced carotid artery thrombosis. Oral administration of Trans-Anethole at 50 mg/kg and 100 mg/kg for 7 days prior to thrombosis induction prolonged the time to complete occlusion by 45% and 68%, respectively, compared to the control group. It also reduced thrombus weight by 32% (50 mg/kg) and 55% (100 mg/kg) and decreased plasma levels of thromboxane B2 (TXB2) by 28% (100 mg/kg)[4] - Antihyperglycemic effect in diabetic rats: STZ-induced diabetic rats were treated with Trans-Anethole (20 mg/kg and 40 mg/kg, oral gavage) daily for 21 days. The 40 mg/kg dose reduced fasting blood glucose levels by 52% compared to the diabetic control group. It also normalized carbohydrate-metabolizing enzymes: HK activity in liver increased by 42%, PFK by 38%, and G6Pase decreased by 28%. Additionally, serum insulin levels increased by 35% in the 40 mg/kg group[5] - Anti-fertility activity in rats: Male albino rats were administered Trans-Anethole (100 mg/kg, oral) daily for 60 days. Sperm count decreased by 65%, sperm motility by 58%, and serum testosterone levels by 42% compared to the control group. Histological examination showed reduced seminiferous tubule diameter and germ cell count in the testes[6] |
| Enzyme Assay |
- Estrogen receptor-mediated β-galactosidase activity assay: Recombinant yeast cells transfected with human ER and β-galactosidase reporter gene were inoculated into culture medium containing different concentrations of Trans-Anethole (0.1–200 μM). The cells were incubated at 30°C for 24 hours, then lysed with lysis buffer. β-galactosidase activity was measured by adding o-nitrophenyl-β-D-galactopyranoside (ONPG) substrate, incubating at 37°C for 30 minutes, and reading absorbance at 420 nm. The EC50 was calculated from the dose-response curve[1] - Carbohydrate-metabolizing enzyme activity assay: Liver tissue from STZ-induced diabetic rats was homogenized in ice-cold buffer. The homogenate was centrifuged at 10,000 × g for 15 minutes, and the supernatant was used for enzyme assays. For HK activity: the reaction mixture contained supernatant, glucose, ATP, and NADP+; absorbance was measured at 340 nm to monitor NADPH formation. For G6Pase activity: the mixture contained supernatant and glucose-6-phosphate; released phosphate was measured using ammonium molybdate reagent at 620 nm. PFK activity was measured by monitoring NADH oxidation at 340 nm in a reaction mixture with supernatant, fructose-6-phosphate, ATP, and NADH-linked enzymes[5] - Fungal enzyme inhibition assay (for Aspergillus flavus ): Aspergillus flavus mycelia were homogenized in buffer and centrifuged to obtain crude enzyme extract. The extract was incubated with Trans-Anethole (0.25–2.0 mg/mL) and cell wall synthesis-related enzyme substrates (e.g., UDP-glucose). Enzyme activity was measured by quantifying remaining substrate using HPLC. At 0.5 mg/mL Trans-Anethole, cell wall synthase activity was inhibited by 60%[7] |
| Cell Assay |
- Recombinant yeast estrogen screen assay: ER-transfected yeast cells were seeded into 96-well plates with medium containing Trans-Anethole (0.1–200 μM). After 24-hour incubation at 30°C, cell viability was checked by trypan blue exclusion (to ensure no cytotoxicity), and β-galactosidase activity was assayed as described in Enzyme Assay. 17β-Estradiol was used as a positive control[1] - MCF-7 cell proliferation assay: MCF-7 cells were seeded into 96-well plates at 5×10³ cells/well. After 24-hour adherence, the medium was replaced with medium containing biotransformed Trans-Anethole (25–200 μM, obtained from rat hepatocyte metabolism) or non-biotransformed Trans-Anethole. After 72-hour incubation, cell proliferation was measured by MTT assay (absorbance at 570 nm). The proliferation rate was calculated relative to the control group[2] - Isolated rat hepatocyte assay: Hepatocytes were isolated from male Wistar rats by collagenase perfusion and seeded into 6-well plates. After 4-hour attachment, Trans-Anethole (50–200 μM) was added. After 24-hour incubation, cell viability was measured by lactate dehydrogenase (LDH) release assay (absorbance at 490 nm) and trypan blue staining. The culture medium was collected to analyze Trans-Anethole metabolites by HPLC[2] - Aspergillus flavus spore germination assay: Aspergillus flavus spores were suspended in potato dextrose broth (PDB) containing Trans-Anethole (0.125–2.0 mg/mL). The suspension was incubated at 28°C, and spore germination was observed under a microscope at 6, 12, and 24 hours. Germination rate was calculated as the percentage of spores with germ tubes longer than the spore diameter[7] |
| Animal Protocol |
- Experimental thrombosis model (rat): Male Wistar rats (200–220 g) were divided into 3 groups (n=6/group): control (saline), Trans-Anethole 50 mg/kg, and Trans-Anethole 100 mg/kg. Trans-Anethole was dissolved in 0.5% carboxymethyl cellulose (CMC) and administered by oral gavage daily for 7 days. On day 8, the left carotid artery was exposed, and a filter paper soaked in 40% ferric chloride was applied to the artery for 10 minutes to induce thrombosis. Blood flow was monitored using a Doppler flowmeter to record time to complete occlusion. After sacrifice, the artery was excised to weigh the thrombus[4] - STZ-induced diabetic rat model: Male Sprague-Dawley rats (180–200 g) were rendered diabetic by a single intraperitoneal injection of STZ (60 mg/kg, dissolved in citrate buffer, pH 4.5). After 7 days, rats with fasting blood glucose >250 mg/dL were selected and divided into 3 groups (n=6/group): diabetic control (saline), Trans-Anethole 20 mg/kg, and Trans-Anethole 40 mg/kg. Trans-Anethole was dissolved in 0.5% CMC and given by oral gavage daily for 21 days. Fasting blood glucose was measured weekly using a glucometer. At the end of the study, rats were sacrificed, and liver tissue was collected for enzyme assays; serum was collected for insulin measurement[5] - Anti-fertility model (rat): Male albino rats (180–200 g) were divided into 2 groups (n=5/group): control (saline) and Trans-Anethole 100 mg/kg. Trans-Anethole was dissolved in corn oil and administered by oral gavage daily for 60 days. Every 15 days, blood samples were collected from the tail vein to measure serum testosterone levels by ELISA. After 60 days, rats were sacrificed; testes were excised for histological examination, and epididymides were collected to count sperm and assess motility[6] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion /IN BIOTRANSFORMATION OF ANETHOLE/...ANISIC ACID...CONSTITUTED THE MAJOR URINARY METABOLITE. ...THERE WAS A QUANTITATIVE RECOVERY OF THE DOSE ADMIN. TRANS-ANETHOLE WAS AMONG 4 FOOD ADDITIVES STUDIED & FOUND LARGELY ABSORBED FROM DIGESTIVE TRACT BY PASSIVE DIFFUSION. ABSORPTION KINETICS VARY PARTIALLY BY DIFFERENCES OF COMPD IN COEFFICIENTS OF LIPOSOLUBILITY. ...TRANS-ANETHOLE USED IN PREPN OF ANIS-FLAVORED ALCOHOLIC BEVERAGES WAS STUDIED IN THE RABBIT & RAT AFTER IV & ORAL ADMIN. IT WAS EXCRETED RAPIDLY FROM THE ANIMAL REGARDLESS OF /ROUTE/ OF ADMIN. AFTER IV INJECTION, IT WAS FOUND CONCENTRATED IN LIVER, LUNGS & BRAIN; AFTER ORAL ADMIN...MOST OF IT REMAINED IN STOMACH. TRANS-ANETHOLE IS ONE OF THE COMPOUNDS LARGELY ABSORBED FROM THE DIGESTIVE TRACT BY MECHANISM OF PASSIVE DIFFUSION. THE METABOLITES OF ANETHOLE IN RATS WERE IDENTIFIED AND DETERMINED. MOST OF THE URINARY METABOLITES WERE ALSO DETECTED IN THE BILE. Metabolism / Metabolites ANETHOLE YIELDS PARA-PROPENYLPHENOL IN RABBIT, MOUSE, GUINEA PIG, & DOG. ANETHOLE YIELDS PARA-PROPENYLPHENOL IN RAT. /FROM TABLE/ ANETHOLE YIELDS ANISIC ACID IN MAN, RABBIT, & RAT. ANETHOLE YIELDS PARA-METHOXYCINNAMYL ALCOHOL IN RAT. OXIDATIVE O-DEMETHYLATION LEADING TO P-HYDROXYPROPENYLBENZENE & P-HYDROXYCINNAMIC ACID WAS FOUND TO BE A MAJOR BIOTRANSFORMATION OF ANETHOLE. METABOLISM VIA CINNAMOYL DERIV WAS MORE EXTENSIVE WITH THE PROPENYL SUBSTITUENT THAN FOR THE ALLYL COMPD; & FURTHER METABOLISM BY BETA-OXIDATION ALSO OCCURRED. - Biotransformation in rat hepatocytes: Isolated rat hepatocytes metabolized Trans-Anethole (100 μM) to two major metabolites (identified by HPLC-MS). The half-life of Trans-Anethole in hepatocyte culture was 4.2 hours, and 65% of the parent compound was metabolized within 12 hours[2] - Oral absorption in rats: After oral administration of Trans-Anethole (40 mg/kg) to diabetic rats, peak plasma concentration (Cmax) of 1.8 μg/mL was reached at 1.5 hours (Tmax), with an apparent volume of distribution (Vd) of 25 L/kg. Plasma half-life (t1/2) was 3.8 hours[5] |
| Toxicity/Toxicokinetics |
Interactions MEAN SLEEPING TIME IN MICE TREATED WITH 50 MG/KG NA-PENTOBARBITAL ABOUT DOUBLED WITH 20 & 50 MG OF SOME COMPD; ANETHOLE WAS LESS ACTIVE. - Hepatocyte toxicity: Trans-Anethole showed dose-dependent cytotoxicity in isolated rat hepatocytes. At 100 μM, LDH release increased by 12% (mild toxicity); at 200 μM, LDH release increased by 35%, and trypan blue-positive cells reached 22% (moderate toxicity)[2] - Reproductive toxicity: Oral administration of Trans-Anethole (100 mg/kg) to male rats for 60 days caused reproductive toxicity, including reduced sperm count (65%), motility (58%), and testosterone levels (42%), with no significant changes in body weight or liver/kidney weight (indicating no systemic toxicity)[6] |
| References |
[1]. Estrogenic activity of isolated compounds and essential oils of Pimpinella species from Turkey, evaluated using a recombinant yeast screen. Planta Med. 2004 Aug;70(8):728-35. [2]. Cytotoxic and xenoestrogenic effects via biotransformation of trans-anethole on isolated rat hepatocytes and cultured MCF-7 human breast cancer cells. Biochem Pharmacol. 2003 Jul 1;66(1):63-73. [3]. "Flavors and Fragrances", Ullmann's Encyclopedia of Industrial Chemistry, Weinheim: Wiley-VCH. [4]. Protective effect of Foeniculum vulgare essential oil and anethole in an experimental model of thrombosis. Pharmacological Research 2007; 56: 254-260. [5]. Trans-anethole, a terpenoid ameliorates hyperglycemia by regulating key enzymes of carbohydrate metabolism in streptozotocin induced diabetic rats. Biochimie 112 (2015): 57-65. [6]. Anti-fertility activity and hormonal profile of trans-anethole in rats. Indian journal of physiology and pharmacology 39 (1995): 63-63. [7]. "Antifungal effects of trans-anethole, the main constituent of Illicium verum fruit volatiles, on Aspergillus flavus in stored wheat." Food Control 149 (2023): 109721. |
| Additional Infomation |
Anethole appears as white crystals or a liquid. Odor of anise oil and a sweet taste. (NTP, 1992) Anethole is a monomethoxybenzene that is methoxybenzene substituted by a prop-1-en-1-yl group at position 4. It has a role as a plant metabolite. Anethole has been reported in Malus, Asarum epigynum, and other organisms with data available. Anethole is a metabolite found in or produced by Saccharomyces cerevisiae. - Trans-Anethole is a major component of essential oils from Pimpinella species (Turkey), Foeniculum vulgare (fennel), and Illicium verum (star anise)[1,4,7] - Trans-Anethole is widely used in the food industry as a flavoring agent and in the cosmetics industry as a fragrance component, with a generally recognized as safe (GRAS) status for food use[3] - The estrogenic activity of Trans-Anethole is mediated by ER binding, which activates estrogen-responsive genes (e.g., β-galactosidase in recombinant yeast)[1] - The anti-thrombotic effect of Trans-Anethole is associated with reduced TXB2 levels, which inhibits platelet aggregation and thrombus formation[4] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~674.76 mM) H2O : ~1 mg/mL (~6.75 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (16.87 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (16.87 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (16.87 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 50 mg/mL (337.38 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 6.7476 mL | 33.7382 mL | 67.4764 mL | |
| 5 mM | 1.3495 mL | 6.7476 mL | 13.4953 mL | |
| 10 mM | 0.6748 mL | 3.3738 mL | 6.7476 mL |