Physicochemical Properties
| Molecular Formula | C16H17N3O5S |
| Molecular Weight | 363.38800 |
| Exact Mass | 363.088 |
| CAS # | 210631-68-8 |
| PubChem CID | 11302979 |
| Appearance | Light beige liquid |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 590.5±60.0 °C at 760 mmHg |
| Flash Point | 310.9±32.9 °C |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.670 |
| LogP | 0.96 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 25 |
| Complexity | 752 |
| Defined Atom Stereocenter Count | 0 |
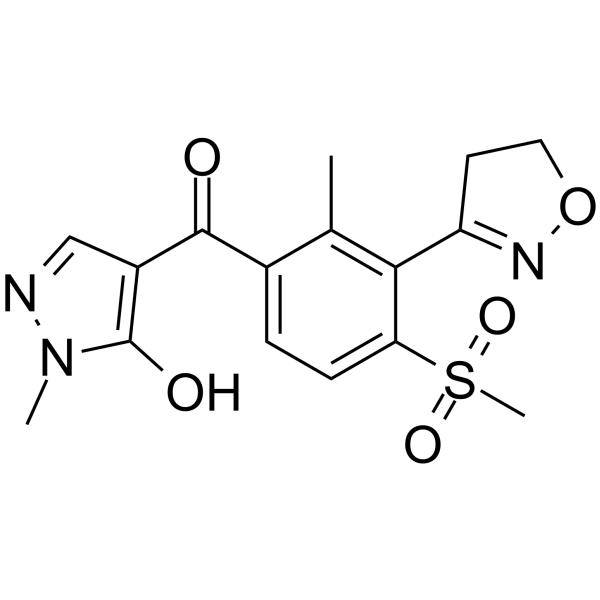
| SMILES | Cc1c(ccc(c1C2=NOCC2)S(=O)(=O)C)C(=O)c3cnn(c3O)C |
| InChi Key | BPPVUXSMLBXYGG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H17N3O5S/c1-9-10(15(20)11-8-17-19(2)16(11)21)4-5-13(25(3,22)23)14(9)12-6-7-24-18-12/h4-5,8,17H,6-7H2,1-3H3 |
| Chemical Name | 4-[3-(4,5-dihydro-1,2-oxazol-3-yl)-2-methyl-4-methylsulfonylbenzoyl]-2-methyl-1H-pyrazol-3-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Topramezone (5, 10, 20, 30, 45 mg/L; 24-96 hours) reduces the formation of C. vulgaris algal blooms by causing oxidative stress, which alters the shape of cells and their ability to photosynthesize [1]. Topramezone (20 mg/L; 24, 48, 72, and 96 hours) depletes the photosynthetic system by decreasing the amount of pigments involved in photosynthesis and causes a time-dependent rise in MDA and ROS (oxidative stress) [1]. Topramezone (20 mg/L; 96 hours) damages algal cell membranes and chloroplasts, impairing cell integrity and inducing death [1]. Topramezone (0.1 nM-0.1 mM; 30 min) has IC50s of 15 nM for setaria, 23 nM for Arabidopsis thaliana, and 180 nM for maize. It is selective and inhibits 4-HPPD (recombinase protein) activity in vitro [2]. Corn metabolizes topramezone (foliar spray; contains 0.29 µg [14C]topramezone; 24 and 48 hours) more quickly than weeds [2]. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Metabolism and pharmacokinetics. Absorption of 14C-topramezone following a single oral dose was rapid but limited, with the highest plasma concentrations observed at 1 hour (first time point measured). Oral absorption is estimated to be approximately 20% of the administered dose. The majority of the dose was recovered within 48 hours in the feces (73- 91% dose) and urine (8-29% dose) /From table/[USEPA; 40 CFR Part 180 Dermal penetration. The majority of the applied dose for each group was not absorbed (91.0-98.3% dose), with the greatest amount of the non-absorbed material being recovered from the skin wash (90.8-96.0% dose). Absorbed radioactivity was low and accounted for 0.16-2.60% of the dose for all groups for all exposures /From table/[USEPA; 40 CFR Part 180 |
| Toxicity/Toxicokinetics |
Toxicity Summary Topramezone has a low acute toxicity via the oral, dermal, or inhalation route. It is a slight eye and dermal irritant, and it is not a skin sensitizer. Following oral administration, topramezone is rapidly absorbed and excreted via urine and feces. Topramezone is an inhibitor of 4-hydroxyphenylpyruvate dioxygenase (4-HPPD); this results in elevated serum tyrosine levels. However, no data could determine at what level increases of tyrosine levels would result in detrimental (adverse) effects. As a consequence of the elevated tyrosine levels, topramezone has been shown to cause adverse effects in the eye, liver, kidney, pancreas, and thyroid. Histopathological evaluations showed dose-dependent increases of adverse effects in the thyroid (follicular cell hyperplasia) in rats and dogs, pancreas (diffuse degeneration) in rats, liver (hepatocellular hypertrophy and focal necrosis) in rats and mice, and eyes (chronic keratitis) in rats. The reproductive toxicity study in rats did not demonstrate adverse reproductive effects; however, developmental toxicity studies in rats and rabbits showed increased incidences of skeletal variation and alterations in skeletal ossification sites. Animal studies show that skeletal variations are associated with 4-HPPD inhibitor herbicides (mesotrione and isoxaflutole). Mutagenicity studies conducted on technical topramezone and its major metabolites did not demonstrate any mutagenic potential. Increased incidences of thyroid follicular cell adenomas and adenoma and/or adenocarcinomas combined were observed in the carcinogenicity study in rats of both sexes. In accordance with the EPA Final Guidelines for Carcinogen Risk Assessment (March 29, 2005), the /EPA Health Effects Division (HED)/ classified topramezone as "not likely to be carcinogenic to humans at doses that do not alter rat thyroid hormone homeostasis". HED determined that quantification of human cancer risk is not required since the NOAEL (0.4 mg/kg/day) for non-cancer risk assessment is not expected to alter thyroid hormone homeostasis nor result in thyroid tumor formation. |
| References |
[1]. Zhao F, et al. Evaluation of the toxicity of herbicide topramezone to Chlorella vulgaris: Oxidative stress, cell morphology and photosynthetic activity. Ecotoxicol Environ Saf. 2017 Sep. 143:129-135. [2]. Grossmann K, et al. On the mechanism of action and selectivity of the corn herbicide topramezone: a new inhibitor of 4-hydroxyphenylpyruvate dioxygenase. Pest Manag Sci. 2007 May. 63(5):429-39. |
| Additional Infomation |
Topramezone is an aromatic ketone that is phenyl 1H-pyrazol-4-yl ketone in which the pyrazolyl group is substituted at positions 1 and 5 by methyl and hydroxy groups, respectively, and in which the phenyl group is substituted at positions 2, 3, and 4 by methyl, 4,5-dihydro-1,2-oxazol-3-yl, and methylsulfonyl groups, respectively. A potent inhibitor of 4-hydroxyphenylpyruvate dioxygenase (HPPD) that is rapily metabolised by corn to non-active substances, it is used as a herbicide for the treatment of broadleaf weeds. It has a role as a herbicide, an agrochemical, an EC 1.13.11.27 (4-hydroxyphenylpyruvate dioxygenase) inhibitor and a carotenoid biosynthesis inhibitor. It is a sulfone, a member of isoxazoles, an aromatic ketone and a pyrazolone. Topramezone is under investigation in clinical trial NCT00559520 (The Role of Preoperative Oral Immunonutrition in Major Vascular Surgery). Mechanism of Action Topramezone has herbicidal activity against broadleaf weeds and grasses. It's efficacy is the result of the inhibition of the enzyme 4-hydroxyphenylpyruvate dioxygenase (HPPD) enzyme in target plants. Following treatment in sensitive plants carotenoid pigment formation, membrane structure and photosynthesis is disrupted. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7519 mL | 13.7593 mL | 27.5186 mL | |
| 5 mM | 0.5504 mL | 2.7519 mL | 5.5037 mL | |
| 10 mM | 0.2752 mL | 1.3759 mL | 2.7519 mL |