Physicochemical Properties
| Molecular Formula | C15H14NO3-.NA+ |
| Molecular Weight | 279.26636 |
| Exact Mass | 279.087 |
| CAS # | 35711-34-3 |
| Related CAS # | Tolmetin sodium dihydrate;64490-92-2;Tolmetin;26171-23-3 |
| PubChem CID | 23665411 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.18g/cm3 |
| Boiling Point | 483.2ºC at 760 mmHg |
| Melting Point | 155-157ºC (DECOMPOSES) |
| Flash Point | 246ºC |
| LogP | 0.856 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 20 |
| Complexity | 353 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CC1=CC=C(C=C1)C(=O)C2=CC=C(CC(=O)O)N2C.[Na].[H] |
| InChi Key | QGUALMNFRILWRA-UHFFFAOYSA-M |
| InChi Code | InChI=1S/C15H15NO3.Na/c1-10-3-5-11(6-4-10)15(19)13-8-7-12(16(13)2)9-14(17)18;/h3-8H,9H2,1-2H3,(H,17,18);/q;+1/p-1 |
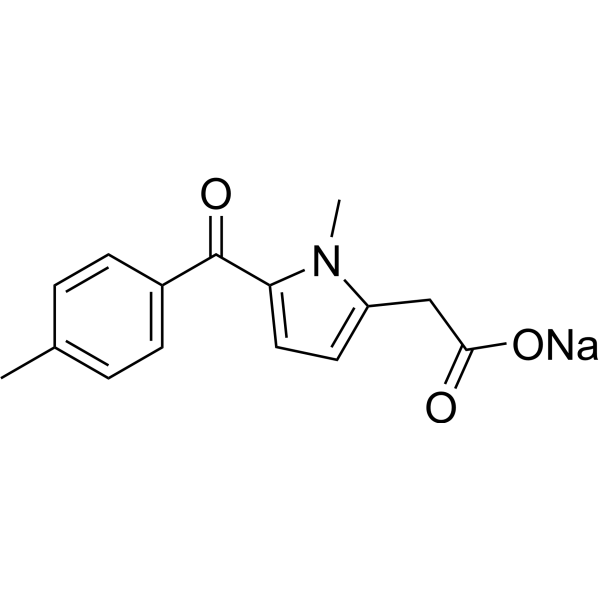
| Chemical Name | sodium;2-[1-methyl-5-(4-methylbenzoyl)pyrrol-2-yl]acetate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Rat brain homogenates show no reduction in lipid peroxidation when tolmetin sodium (0.25 mM) is added. In rat brain homogenate, tolmetin (0.25, 0.5, 0.75, and 1 mM) demonstrates free radical scavenging activities without producing superoxide anion [3]. The HT-29 colon cancer cell line is susceptible to dose-dependent anticancer action from tolmetin sodium (0.001-100 μM) [4]. The growth of osteoblasts is not affected by tolmetin sodium (0-100 μM) [5]. |
| ln Vivo | Maximum ulcerogenicity in males weighing 180–180 kg was observed 4 hours after a single dose of tolmetin sodium (30,100 mg/kg; gavage; once dose or twice daily for 3 and 14 days). The ulcerogenic effect was significantly reduced in Wistar rats after repeated administration for 3 and 14 days. 200 grams. At 100 mg/kg, tolmetin can cause stomach damage [2]. The neurotoxicity caused by quinolinic acid (QA) was significantly reduced when tolmetin sodium (5 mg/kg twice daily) was administered prior to the experiment [3]. |
| References |
[1]. Nonsteroid drug selectivities for cyclo-oxygenase-1 rather than cyclo-oxygenase-2 are associated with human gastrointestinal toxicity: a full in vitro analysis. Proc Natl Acad Sci U S A. 1999 Jun 22;96(13):7563-8. [2]. Three new vanadyl(IV) complexes with non-steroidal anti-inflammatory drugs (Ibuprofen, Naproxen and Tolmetin). Bioactivity on osteoblast-like cells in culture. J Inorg Biochem. 2002 Jan 1;88(1):94-100. [3]. Synthesis and anticancer activity of some novel tolmetin thiosemicarbazides. Marmara Pharmaceutical Journal 19(3) • April 2015. |
| Additional Infomation |
Tolmetin sodium is an organic sodium salt that is the monosodium salt of tolmetin. Used in the form of its dihydrate as a nonselective nonsteroidal anti-inflammatory drug. It has a role as an EC 1.14.99.1 (prostaglandin-endoperoxide synthase) inhibitor and a non-steroidal anti-inflammatory drug. It contains a tolmetin(1-). Tolmetin Sodium is the sodium salt form of tolmetin, an arylalkanoic acid and non-steroidal anti-inflammatory drug (NSAID) with analgesic, anti-inflammatory and antipyretic activities. Although the exact mechanism through which tolmetin exerts its effects has yet to be fully elucidated, this agent appears to inhibit the enzyme prostaglandin synthase. This prevents the formation of prostaglandins from prostaglandin precursors, including the synthesis of the inflammatory prostaglandin E2 (PGE2) from the precursor prostaglandin H2 (PGH2). This prevents prostaglandin-mediated effects, including pain, inflammation and fever. A non-steroidal anti-inflammatory agent (ANTI-INFLAMMATORY AGENTS, NON-STEROIDAL) similar in mode of action to INDOMETHACIN. See also: Tolmetin (has active moiety). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5808 mL | 17.9038 mL | 35.8076 mL | |
| 5 mM | 0.7162 mL | 3.5808 mL | 7.1615 mL | |
| 10 mM | 0.3581 mL | 1.7904 mL | 3.5808 mL |