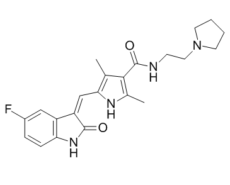
Toceranib (formerly known as PHA-291639, or SU-11654) is an inhibitor of receptor tyrosine kinase (RTK) with anticancer activities. It inhibits Flk-1/KDR and PDGFRβ with Ki values of 6 nM and 5 nM, respectively. Toceranib (brand name: Palladia) has been authorized for use as a veterinary medication to treat canine mast cell tumors, also known as mastocytomas. Toceranib may also have an anti-angiogenic effect, but its main mechanism of action is probably inhibition of kit tyrosine kinase. is an anticancer and antineoplastic drug used to treat canine mast cell tumors.
Physicochemical Properties
| Molecular Formula | C22H25FN4O2 |
| Molecular Weight | 396.47 |
| Exact Mass | 396.196 |
| Elemental Analysis | C, 66.65; H, 6.36; F, 4.79; N, 14.13; O, 8.07 |
| CAS # | 356068-94-5 |
| Related CAS # | Toceranib phosphate;874819-74-6;Toceranib-d8;1795134-78-9 |
| PubChem CID | 5329106 |
| Appearance | Yellow to orange solid powder |
| LogP | 3.686 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 29 |
| Complexity | 663 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | FC1C([H])=C([H])C2=C(C=1[H])/C(/C(N2[H])=O)=C(\[H])/C1=C(C([H])([H])[H])C(=C(C([H])([H])[H])N1[H])C(N([H])C([H])([H])C([H])([H])N1C([H])([H])C([H])([H])C([H])([H])C1([H])[H])=O |
| InChi Key | SRSGVKWWVXWSJT-ATVHPVEESA-N |
| InChi Code | InChI=1S/C22H25FN4O2/c1-13-19(12-17-16-11-15(23)5-6-18(16)26-21(17)28)25-14(2)20(13)22(29)24-7-10-27-8-3-4-9-27/h5-6,11-12,25H,3-4,7-10H2,1-2H3,(H,24,29)(H,26,28)/b17-12- |
| Chemical Name | 5-[(Z)-(5-fluoro-2-oxo-1H-indol-3-ylidene)methyl]-2,4-dimethyl-N-(2-pyrrolidin-1-ylethyl)-1H-pyrrole-3-carboxamide |
| Synonyms | SU-11654; PHA291639; SU 11654; PHA 291639; SU11654; PHA-291639; Toceranib; Toceranib [USAN]; (Z)-5-((5-fluoro-2-oxoindolin-3-ylidene)methyl)-2,4-dimethyl-N-(2-(pyrrolidin-1-yl)ethyl)-1H-pyrrole-3-carboxamide; (Z)-5-(5-Fluoro-2-oxo-2,3-dihydro-1H-indol-3-ylidenemethyl)-2,4-dimethyl-N-[2-(1-pyrrolidinyl)ethyl]-1H-pyrrole-3-carboxamide; Trade name: Palladia |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
PDGFRβ (Ki = 5 nM); Flk-1 (Ki = 6 nM)
KIT (IC₅₀=0.03 μM) [1] Vascular Endothelial Growth Factor Receptor 2 (VEGFR2, KDR) (IC₅₀=0.05 μM) [1] Platelet-Derived Growth Factor Receptor β (PDGFRβ) (IC₅₀=0.01 μM) [1] Colony-Stimulating Factor 1 Receptor (CSF1R, FMS) (IC₅₀=0.11 μM) [1] |
| ln Vitro |
Toceranib (SU11654) is a selective inhibitor of PDGFR and Flk-1/KDR tyrosine kinase activity, as well as other members of the split kinase RTK family, with a Kis of 5 and 6 nM, respectively[1]. The Toceranib-sensitive exon 11 ITD c-kit mutant C2 cell line is used to create three resistant sublines, TR1, TR2, and TR3, which are then used to investigate the mechanisms of acquired Toceranib (TOC) resistance in canine MCT. With an IC50 of less than 10 nM, toceranib inhibits the growth of the parental C2 cells in a dose-dependent manner. Conversely, sublines TR1, TR2, and TR3 exhibit resistance against Toceranib's inhibition (IC50>1,000 nM). Similar to the reported resistance to toceranib, sensitivity to three additional KIT RTK inhibitors is seen. Vinblastine (VBL) and CCNU are cytotoxic agents that still cause sensitivity in the parental line and all three sublines. After being cultured for 72 hours in the presence of increasing Toceranib concentrations, treatment-naïve parental C2 cells separate from the culture flask and, as Toceranib exposure increases, they become clumped, shrunken, and rounded. In contrast, it is not possible to identify morphologic differences induced by ceranib in the resistant sublines[2]. Toceranib (SU11654) is a small-molecule multi-targeted receptor tyrosine kinase inhibitor that potently inhibits the kinase activity of KIT, VEGFR2, PDGFRβ, and CSF1R in vitro [1] - In canine mast cell tumor (MCT) cell lines (C2, NI-1, and Madin-Darby canine kidney (MDCK) cells transfected with wild-type or mutant KIT), it inhibits cell proliferation with IC₅₀ values ranging from 0.1 to 0.5 μM; cells harboring KIT D816V mutation show reduced sensitivity (IC₅₀=0.8 μM) compared to wild-type KIT-expressing cells (IC₅₀=0.2 μM) [2] - Long-term exposure of C2 MCT cells to increasing concentrations of Toceranib (0.01–0.5 μM over 6 months) induces acquired resistance, with the resistant cell line (C2-TocR) showing a 4.2-fold higher IC₅₀ (0.84 μM) than the parental C2 cells (0.2 μM) [2] - Toceranib inhibits KIT phosphorylation and downstream signaling (AKT, ERK1/2) in parental C2 cells but not in C2-TocR cells; sequencing reveals a KIT D816V mutation in C2-TocR cells [2] - In vitro immunomodulatory assays show that Toceranib (0.1–1 μM) increases the proliferation of canine peripheral blood mononuclear cells (PBMCs) stimulated by concanavalin A (ConA) and enhances the cytotoxic activity of natural killer (NK) cells against canine tumor cell lines [3] - It reduces the frequency of regulatory T cells (Tregs) in ConA-stimulated canine PBMCs in a dose-dependent manner (1 μM: 35% reduction vs vehicle control) [3] |
| ln Vivo |
Fifty-seven dogs with a variety of cancers were enrolled; of these, 10 experienced progressive disease within the first 3 weeks. Measurable objective responses were observed in 16 dogs (including 6 complete responses), primarily in mast cell tumors (n = 11), mixed mammary carcinomas (n = 2), soft tissue sarcomas (n = 2), and multiple myeloma (n = 1), for an overall response rate of 28% (16 of 57). Stable disease of sufficient duration to be considered clinically meaningful (>10 weeks) was seen in an additional 15 dogs, for a resultant overall biological activity of 54% (31 of 57). Conclusions: This study provides the first evidence that p.o. administered kinase inhibitors can exhibit activity against a variety of spontaneous malignancies. Given the similarities of canine and human cancers with regard to tumor biology and the presence of analogous RTK dysregulation, it is likely that such agents will demonstrate comparable antineoplastic activity in people.[1] Administration of toceranib significantly decreased the number and percentage of Treg in the peripheral blood of dogs with cancer. Dogs receiving toceranib and CYC demonstrated a significant increase in serum concentrations of IFN-γ, which was inversely correlated with Treg numbers after 6 weeks of combination treatment. Conclusions: In addition to antitumor effects, these data support further investigations into the immunomodulatory effects of toceranib, administered alone or in combination with CYC in dogs with cancer.[3] The amount and proportion of Treg in cancer-stricken dogs' peripheral blood dramatically decrease after toceranib administration. After 6 weeks of combination therapy, dogs receiving toceranib and cyclophosphamide (CYC) show a significant increase in serum concentrations of IFN-γ, which is inversely correlated with Treg numbers[3]. In a phase I dose-escalation study in dogs with spontaneous malignancies (including MCT, sarcomas, carcinomas), oral Toceranib at doses of 1.0 mg/kg, 1.5 mg/kg, and 2.0 mg/kg once daily for 28 days shows antitumor activity: 12/34 dogs (35.3%) achieve partial response (PR), 14/34 (41.2%) achieve stable disease (SD), and 8/34 (23.5%) show progressive disease (PD); the highest response rate is observed in MCT (5/8 dogs, 62.5% PR) [1] - Tumor growth inhibition is dose-dependent, with 2.0 mg/kg group showing significantly higher PR rate than 1.0 mg/kg group (p<0.05) [1] - In dogs with spontaneous cancer treated with Toceranib (2.0 mg/kg PO q24h) combined with low-dose cyclophosphamide (10 mg/m² PO q72h) for 8 weeks, 9/18 dogs (50%) achieve PR, 7/18 (38.9%) achieve SD, and 2/18 (11.1%) show PD; the combination enhances antitumor efficacy compared to historical Toceranib monotherapy data [3] - The combination therapy increases peripheral blood CD4⁺ T cells and NK cells, reduces Tregs, and enhances the production of interferon-γ (IFN-γ) and tumor necrosis factor-α (TNF-α) in dogs [3] - In nude mice xenografted with C2-TocR cells, oral Toceranib (40 mg/kg once daily for 21 days) fails to inhibit tumor growth, while parental C2 cell xenografts show 75% TGI; treatment with a dual KIT/BCR-ABL inhibitor inhibits C2-TocR xenograft growth [2] |
| Enzyme Assay |
Toceranib (SU11654) is a selective inhibitor of PDGFR and Flk-1/KDR tyrosine kinase activity, as well as other members of the split kinase RTK family, with a Kis of 5 and 6 nM, respectively. For PDGFR and Flk-1/KDR, two members of the split kinase RTK family, toceranib (SU11654) selectively inhibits tyrosine kinase activity at 5 and 6 nM, respectively.
The canine C2 mastocytoma cell line contains an activating mutation in c-kit. Three Toceranib (TOC)-resistant C2 sublines (TR1, TR2, TR3) were established over seven months by growing cells in increasing concentrations of TOC. TOC inhibited KIT phosphorylation and cell proliferation in a dose-dependent manner in the treatment-naïve, parental C2 line (IC50 < 10 nM). In contrast, the three sublines were resistant to growth inhibition by TOC (IC50 > 1,000 nM) and phosphorylation of the KIT receptor was less inhibited compared to the TOC-sensitive C2 cells. Interestingly, sensitivity to three structurally distinct KIT RTK inhibitors was variable among the sublines, and all 3 sublines retained sensitivity to the cytotoxic agents vinblastine and lomustine. Sequencing of c-kit revealed secondary mutations in the juxtamembrane and tyrosine kinase domains of the resistant sublines. These included point mutations in TR1 (Q574R, M835T), TR2 (K724R), and TR3 (K580R, R584G, A620S). Additionally, chronic TOC exposure resulted in c-kit mRNA and KIT protein overexpression in the TOC-resistant sublines compared to the parental line. C2, TR1, TR2, and TR3 cells demonstrated minimal P-glycoprotein (P-gp) activity and no functional P-gp. Conclusions: This study demonstrates the development of an in vitro model of acquired resistance to targeted therapy in canine MCTs harboring a c-kit-activating mutation. This model may be used to investigate the molecular basis of and strategies to overcome TOC resistance [2]. Recombinant kinase activity assay: Recombinant human KIT, VEGFR2, PDGFRβ, and CSF1R kinases are diluted in assay buffer (50 mM Tris-HCl pH 7.5, 10 mM MgCl₂, 1 mM EGTA, 0.01% BSA, 1 mM DTT). Serial 3-fold dilutions of Toceranib (0.001–10 μM) are mixed with each kinase and pre-incubated for 30 minutes at room temperature. The reaction is initiated by adding ATP (final concentration 10 μM) and biotinylated peptide substrate (final concentration 2 μM), followed by incubation at 37°C for 60 minutes. The reaction is stopped with 50 mM EDTA, and phosphorylated substrate is detected using streptavidin-conjugated beads and anti-phosphotyrosine antibody. Fluorescence intensity is measured, and IC₅₀ values are calculated via nonlinear regression [1] |
| Cell Assay |
The cells of BR, C2, and P815 were counted and then resuspended in complete medium in 24-well plates at a concentration of 0.5 × 106 cells/well (for BR and C2) or 0.25 × 106 cells/well (for P815). Cells were left untreated, or SU11652, SU11654, or SU11655 was added to a final concentration of 0.01 to 1 μM. After being cultured for 24, 48, and 72 hours, cells were taken out and counted to find out how many cells survived. Canine MCT cell proliferation assay: Parental C2, NI-1, KIT-transfected MDCK cells, and C2-TocR cells are seeded in 96-well plates (5×10³ cells/well) and incubated overnight. Serial 3-fold dilutions of Toceranib (0.001–10 μM) are added, and cells are cultured for 72 hours. Cell viability is detected by MTT assay, and IC₅₀ values are calculated [2] - Acquired resistance induction assay: C2 cells are seeded in 6-well plates and cultured with Toceranib at an initial concentration of 0.01 μM. The drug concentration is increased by 0.05 μM every 2 weeks if cell viability exceeds 70%. After 6 months of continuous exposure, the resistant cell line (C2-TocR) is established and validated for resistance [2] - Western blot for signaling pathways: Parental C2 and C2-TocR cells are treated with Toceranib (0.5 μM) for 4 hours, lysed, and proteins are separated by SDS-PAGE. Membranes are probed with antibodies against p-KIT (Tyr719), KIT, p-AKT (Ser473), AKT, p-ERK1/2 (Thr202/Tyr204), ERK1/2, and β-actin [2] - Immunomodulatory cell assay: Canine PBMCs are isolated from healthy dogs and seeded in 96-well plates (2×10⁵ cells/well). Toceranib (0.1–1 μM) and ConA (5 μg/mL) are added, and cells are cultured for 72 hours. Cell proliferation is detected by MTS assay; Treg frequency is analyzed by flow cytometry (CD4⁺CD25⁺Foxp3⁺); NK cell cytotoxicity is measured using a lactate dehydrogenase (LDH) release assay [3] |
| Animal Protocol |
Dogs; Fifteen client-owned dogs with advanced tumors are used. Toceranib is given to dogs once every other day at a dosage of 2.75 mg/kg. Oral cyclophosphamide (CYC) is added at a daily dose of 15 mg/m2 following a 2-week period. Throughout the eight-week study period, flow cytometry is used to quantify the number of Treg and lymphocyte subsets in blood. ELISA is used to gauge IFN- concentrations in serum. Phase I dose-escalation study in dogs: Dogs with histologically confirmed spontaneous malignancies are enrolled into three dose groups (1.0 mg/kg, 1.5 mg/kg, 2.0 mg/kg). Toceranib is formulated as oral capsules and administered once daily for 28 days. Tumor size is measured every 7 days using calipers; response is evaluated per RECIST criteria. Blood samples are collected for PK analysis and toxicity monitoring (hematology, biochemistry) [1] - Combination therapy study in dogs: Dogs with spontaneous cancer are treated with Toceranib (2.0 mg/kg oral once daily) plus cyclophosphamide (10 mg/m² oral every 72 hours) for 8 weeks. Tumor response is assessed every 4 weeks; peripheral blood is collected for immunophenotyping (flow cytometry) and cytokine analysis (ELISA) [3] - Xenograft model in nude mice: Nude mice (6–8 weeks old) are subcutaneously implanted with 5×10⁶ parental C2 or C2-TocR cells. When tumors reach 100–150 mm³, mice are treated with Toceranib (40 mg/kg oral once daily) for 21 days. Tumor volume is measured every 3 days; mice are euthanized at study end, and tumors are collected for histopathological analysis [2] |
| ADME/Pharmacokinetics |
In dogs (phase I study): Oral bioavailability (F) = 60–70%, Cmax = 0.8–1.2 μg/mL (2.0 mg/kg dose), AUC₀–24h = 8.5–10.2 μg·h/mL, t₁/₂ = 12.3–14.5 hours; steady-state concentrations are achieved by day 7 of daily dosing [1] - Tissue distribution: In dogs, Toceranib distributes widely to tumor tissues, with tumor/plasma concentration ratio of 2.3–3.1 at 4 hours post-dose [1] - Plasma protein binding rate: 91–93% in canine plasma (equilibrium dialysis, 0.1–10 μg/mL) [1] - Metabolism: Predominantly metabolized by hepatic cytochrome P450 enzymes (CYP3A4/5) in dogs; two major metabolites are identified, with no significant kinase inhibitory activity [1] |
| Toxicity/Toxicokinetics |
Phase I study in dogs: Dose-limiting toxicity (DLT) is grade 3 neutropenia (observed at 2.0 mg/kg); common treatment-related adverse events (TRAEs) include grade 1–2 diarrhea (47%), vomiting (35%), anorexia (29%), and grade 1 neutropenia (26%); no grade 4 TRAEs or treatment-related deaths are reported [1] - Liver function: Mild, transient increases in ALT (18%) and AST (12%) are observed, resolving without treatment interruption [1] - Combination therapy toxicity: Dogs treated with Toceranib + cyclophosphamide show similar TRAEs (diarrhea, vomiting, neutropenia) to Toceranib monotherapy, with no increase in severity [3] - No significant nephrotoxicity (BUN, creatinine unchanged) is observed in any study [1][3] |
| References |
[1]. Phase I dose-escalating study of SU11654, a small molecule receptor tyrosine kinase inhibitor, in dogs with spontaneous malignancies. Clin Cancer Res. 2003 Jul;9(7):2755-68. [2]. Development of an in vitro model of acquired resistance to toceranib phosphate (Palladia?) in canine mast cell tumor. BMC Vet Res. 2014 May 6;10:105. [3]. Clinical and immunomodulatory effects of toceranib combined with low-dose cyclophosphamide in dogs with cancer. J Vet Intern Med. 2012 Mar-Apr;26(2):355-62. |
| Additional Infomation |
See also: Toceranib Phosphate (has salt form). Drug Indication Treatment of non-resectable Patnaik grade-II (intermediate-grade) or -III (high-grade), recurrent, cutaneous mast-cell tumours in dogs. Toceranib (SU11654), marketed as Palladia®, is the first small-molecule receptor tyrosine kinase inhibitor approved by the FDA for the treatment of canine cancer (specifically, unresectable, recurrent, or metastatic canine MCT) [1] - Its antitumor mechanism involves dual inhibition of tumor cell proliferation (via KIT, PDGFRβ) and angiogenesis (via VEGFR2), as well as immunomodulatory effects (enhancing T cell and NK cell function, reducing Tregs) [1][3] - Acquired resistance to Toceranib in canine MCT is primarily mediated by KIT D816V mutation, which can be overcome by dual KIT/BCR-ABL inhibitors [2] - Low-dose cyclophosphamide enhances the immunomodulatory and antitumor effects of Toceranib without increasing toxicity, providing a potential combination strategy for canine cancer [3] - It is well-tolerated in dogs at the recommended dose of 2.0 mg/kg once daily, with manageable gastrointestinal and hematological toxicity [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5223 mL | 12.6113 mL | 25.2226 mL | |
| 5 mM | 0.5045 mL | 2.5223 mL | 5.0445 mL | |
| 10 mM | 0.2522 mL | 1.2611 mL | 2.5223 mL |