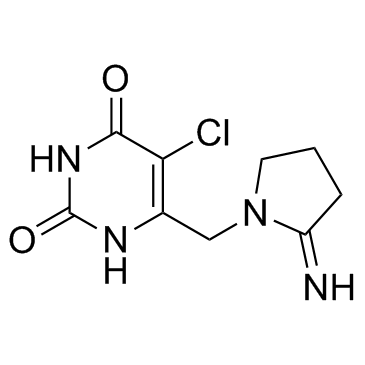
Tipiracil (TPI), one of the main ingredients in TAS-102 (trade name Lonsurf, a mixture of Trifluridine and tipiracil hydrochloride in a 1:0.5 molar ratio), is a potent thymidine phosphorylase (TPase) inhibitor with anticancer activity. TAS-102 has been approved in Japan in 2015.
Physicochemical Properties
| Molecular Formula | C9H11N4O2CL |
| Molecular Weight | 242.66224 |
| Exact Mass | 242.057 |
| CAS # | 183204-74-2 |
| Related CAS # | Trifluridine/tipiracil hydrochloride mixture;733030-01-8;Tipiracil hydrochloride;183204-72-0 |
| PubChem CID | 6323266 |
| Appearance | White to off-white solid |
| Density | 1.7±0.1 g/cm3 |
| Melting Point | 245ºC (decomposition) |
| Index of Refraction | 1.743 |
| LogP | -1.37 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 16 |
| Complexity | 404 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O=C1NC(C(Cl)=C(CN2C(CCC2)=N)N1)=O |
| InChi Key | QQHMKNYGKVVGCZ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C9H11ClN4O2/c10-7-5(12-9(16)13-8(7)15)4-14-3-1-2-6(14)11/h11H,1-4H2,(H2,12,13,15,16) |
| Chemical Name | 5-chloro-6-[(2-iminopyrrolidin-1-yl)methyl]-1H-pyrimidine-2,4-dione |
| Synonyms | Tipiracil |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | thymidine phosphorylase |
| ln Vitro | Thymidine phosphorylase is inhibited by the drug tipiracil. Tipiracil increases trifluridine exposure by preventing thymidine phosphorylase from metabolizing the drug. A brand-new oral treatment for metastatic colorectal cancer combines trifluridine and piracil[2]. As a thymidine phosphorylase inhibitor, tipiracil has a first-pass effect that prevents FTD from degrading[3]. |
| Cell Assay | HeLa cells are plated in triplicate in 96-well plates at a density of 500 cells/180 μL/well and pre-cultured for 24 hours before receiving 20 μL of each drug solution for 24 or 72 hours. Following the 24 h treatment, cells are washed with phosphate-buffered saline (PBS) before being added to each well with drug-free medium and being incubated for an additional 48 h. A Cell Counting Kit-8 is used to assess the inhibition of cell growth. SAS[3] is used to determine the 50% inhibitory concentration (IC50) values from the absorbance data. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Absorption of tipiracil is suggested to be done by the gastrointestinal tract. Administration of a single 35 mg/m2 dose of TAS-102 containing tipiracil and trifluridine, generates the absoprtion rates of tipiracil of AUC 301 ng h/ml, maximum observed plasma concentration (Cmax) 69 ng/ml and time for maximum observed plasma concentration (Tmax) 3 h. The consumption of a high-fat and high-calorie meal can decrease Cmax and AUC by 40%. A standardized high-fat, high-calorie meal decreased tipiracil Cmax and AUC by approximately 40% in patients with cancer following administration of a single dose of LONSURF 35 mg. After single oral administration of LONSURF (60 mg) with [14C]-tipiracil hydrochloride, recovered radioactivity was 77% of the dose, which consisted of 27% urinary excretion and 50% fecal excretion. Tipiracil was the major component and 6-HMU was the major metabolite in urine and feces. Following a single dose of LONSURF (35 mg/m2) in patients with advanced solid tumours, the apparent volume of distribution (Vd/F) for tipiracil hydrochloride was 333 L. Following a single dose of LONSURF (35 mg/m2) in patients with advanced solid tumours, the oral clearance (CL/F) for tipiracil hydrochloride was 109 L/hr. Metabolism / Metabolites Tipiracil does not undergo much metabolism upon first pass. It is not metabolized by the liver or hepatocytes, nor by the cytochrome P450 enzymes. The only tipiracil-derived metabolite found in very small quantities in human plasma, urine or faeces is 6-hydroxymethyluracil (6-HMU) which is not unique of tipiracil. This metabolite is though to be formed either by enterobacterial metabolism. In plasma, this two metabolites can be found in a proportion of tipiracil 53.1% and 6-HMU 30.9%. Biological Half-Life After administration of LONSURF 35 mg/m2, the mean elimination and steady-state half-life (t1/2) of tipiracil was 2.1 hours and 2.4 hours respectively. |
| Toxicity/Toxicokinetics |
Protein Binding The plasma protein binding of tipiracil is below 8%. |
| References |
[1]. Thymidine phosphorylase influences [(18)F]fluorothymidine uptake in cancer cells and patients with non-small cell lung cancer. Eur J Nucl Med Mol Imaging. 2014 Jul;41(7):1327-35. [2]. Lonsurf (Trifluridine plus Tipiracil): A New Oral Treatment Approved for Patients with Metastatic Colorectal Cancer. Am Health Drug Benefits. 2016 Mar;9(Spec Feature):97-100. [3]. Repeated oral dosing of TAS-102 confers high trifluridine incorporation into DNA and sustained antitumor activity in mouse models. Oncol Rep. 2014 Dec;32(6):2319-26. |
| Additional Infomation |
Tipiracil is a member of the class of pyrimidones that is uracil substituted by chloro and (2-iminopyrrolidin-1-yl)methyl groups at positions 5 and 6 respectively. Used (as the hydrochloride salt) in combination with trifluridine, a nucleoside metabolic inhibitor, for treatment of advanced/relapsed unresectable colorectal cancer. It has a role as an antineoplastic agent and an EC 2.4.2.4 (thymidine phosphorylase) inhibitor. It is a pyrimidone, an organochlorine compound, a carboxamidine and a member of pyrrolidines. It is functionally related to a uracil. It is a conjugate base of a tipiracil(1+). Tipiracil is a thymidine phosphorylase inhibitor. It is used in combination with trifluridine, in a ratio of 1:0.5, to form TAS-102. The main function of Tipiracil in TAS-102 is to increase trifluridine bioavailability by inhibiting its catabolism. TAS-102 is indicated for the treatment of metastatic colorectal cancer which has been previously treated with fluoropyrimidine-, oxaliplatin- and irinotecan-based chemotherapy, or with an anti-VEGF or anti-EGFR therapy. Tipiracil is a Thymidine Phosphorylase Inhibitor. The mechanism of action of tipiracil is as a Thymidine Phosphorylase Inhibitor. Drug Indication Tipiracil is also available as a combination product with [Trifluridine], which is indicated either alone or in combination with [bevacizumab] for the treatment of adult patients with metastatic colorectal cancer who have been previously treated with fluoropyrimidine-, oxaliplatin- and irinotecan-based chemotherapy, an anti-VEGF biological therapy, and if RAS wild-type, an anti-EGFR therapy. This combination product is also used for adult patients with metastatic gastric or gastroesophageal junction adenocarcinoma and were previously treated with at least two prior lines of chemotherapy that included a fluoropyrimidine, a platinum, either a taxane or irinotecan and if appropriate, HER2/neu-targeted therapy. FDA Label Mechanism of Action Tipiracil is a thymidine phosphorylase inhibitor. Its function prevents the breakdownof the active component of trifluridine, thus increasing the bioavailability of trifluridine and boosting its systemic presence. In addition, it is reported that thymidine phosphorylase is an angiogenic factor usually overexpressed in solid tumors. There is a direct association of thymidine phosphorylase with a poor prognosis; where the tumors with an elevated expression of this enzyme tend to present an increased angiogenesis and ergo, be more malignant. Therefore, it has been suggested that tipiracil presents an aditional function by downregulating tumoral angiogenesis. Pharmacodynamics Tipiracil prevents trifluridine conversion into 5-trifluoromethyl-2,4(1H,3H)-pyrimidinedione, which is an inactive major metabolite, by inhibiting the enzyme thymidine phosphorylase. Thus, tipiracil is able to increase trifluridine bioavailability. On the other hand, thymidine phsophorylase is a known platelet-derived endothelial cell growth factor and its inhibition generates an indirect antiangiogenic benefit. |
Solubility Data
| Solubility (In Vitro) |
H2O: ~1 mg/mL (~4.1 mM) DMSO: <1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2 mg/mL (8.24 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication (<60°C). (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.1210 mL | 20.6050 mL | 41.2099 mL | |
| 5 mM | 0.8242 mL | 4.1210 mL | 8.2420 mL | |
| 10 mM | 0.4121 mL | 2.0605 mL | 4.1210 mL |