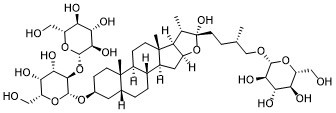
Timosaponin BII (TB-II) is a steroidal saponin-based antioxidant and an anti-inflammatory agent isolated from Anemarrhenae with diverse biological activities, e.g. neuronal protective, anti-inflammatory and antioxidant activities.
Physicochemical Properties
| Molecular Formula | C45H76O19 |
| Molecular Weight | 921.0736 |
| Exact Mass | 920.498 |
| CAS # | 136656-07-0 |
| PubChem CID | 4483043 |
| Appearance | White to off-white solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 1033.6±65.0 °C at 760 mmHg |
| Melting Point | 187 - 189 °C |
| Flash Point | 578.9±34.3 °C |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.627 |
| LogP | 0.34 |
| Hydrogen Bond Donor Count | 12 |
| Hydrogen Bond Acceptor Count | 19 |
| Rotatable Bond Count | 13 |
| Heavy Atom Count | 64 |
| Complexity | 1560 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | SORUXVRKWOHYEO-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C45H76O19/c1-19(18-58-40-37(55)34(52)31(49)27(15-46)60-40)7-12-45(57)20(2)30-26(64-45)14-25-23-6-5-21-13-22(8-10-43(21,3)24(23)9-11-44(25,30)4)59-42-39(36(54)33(51)29(17-48)62-42)63-41-38(56)35(53)32(50)28(16-47)61-41/h19-42,46-57H,5-18H2,1-4H3 |
| Chemical Name | 2-[4-[16-[4,5-dihydroxy-6-(hydroxymethyl)-3-[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyoxan-2-yl]oxy-6-hydroxy-7,9,13-trimethyl-5-oxapentacyclo[10.8.0.02,9.04,8.013,18]icosan-6-yl]-2-methylbutoxy]-6-(hydroxymethyl)oxane-3,4,5-triol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
With an IC50 value of 15.5 μg/mL in HL-60 cells, timosaponin BII, a steroidal glycoside isolated from Anemarrhenae, was found to be effective against human cancer cell lines, including HT-29 (colon), HepG2 and Bel-7402 (liver), HL-60 (leukemia), and MDA-MB-468 (breast cancer)[1]. Timosaponin BII exhibited cytotoxic activity against four human cancer cell lines in vitro as measured by the Sulforhodamine B (SRB) assay. The IC50 values were 2.46 µM against human hepatic carcinoma BEL-7402 cells, 1.65 µM against human colon carcinoma HT-29 cells, and 5.05 µM against human breast carcinoma MDA-MB-468 cells. It showed no significant activity (IC50 > 100 µM) against human cervix HeLa adenocarcinoma cells. [1] Among the isolated and modified compounds, Timosaponin BII (compound 2) and Timosaponin AIII (compound 4) showed the best cytotoxic activities against BEL-7402, HT-29, and MDA-MB-468 cell lines, with mean IC50 values of 3.05 µM and 1.82 µM, respectively. [1] Preliminary structure-activity relationship analysis indicated that the anti-proliferative activity of Timosaponin BII was reduced upon acid hydrolysis to yield derivatives 2a-2f. Among them, only derivative 2b showed certain cytotoxic activities against BEL-7402 and HT-29 cell lines, but these were weaker than the parent compound Timosaponin BII. The activity was significantly diminished when the E-ring was saturated (compound 3, derived from Timosaponin BII, showed no activity). [1] |
| ln Vivo | Rats in both the vehicle control group and the model group had considerably higher levels of BACE1 expression in their retinas. Aβ1-40, β-CTF, and malondialdehyde (MDA) all rose at the same period. In contrast to the vehicle control group, the retinas from the tiemaponin-BII treated group exhibited considerably lower levels of BACE1 and collected less Aβ1-40 or β-CTF. Additionally, a notable reduction in MDA levels and an elevation in partial thromboplastin time are seen [2]. |
| Cell Assay | The cytotoxic activities of the compounds were evaluated against human cancer cell lines (BEL-7402, HT-29, HeLa, and MDA-MB-468) using the Sulforhodamine B (SRB) protein coloration method. Briefly, cells were seeded in 96-well plates at optimal densities and incubated overnight. Triplicate wells were then treated with various concentrations of the test compounds and incubated for 72 hours. Doxorubicin was used as a positive control. After incubation, the culture medium was discarded, and the cells were fixed with pre-cooled 10% trichloroacetic acid at 4°C for 1 hour. The fixed cells were washed with distilled water and stained with 0.4% SRB in 1% acetic acid for 15 minutes. Unbound dye was removed by washing with 1% acetic acid. The plates were air-dried, and the bound dye was dissolved in 10 mM Tris base (pH 10.5). The absorbance of each well was measured at 560 nm using a multiwall spectrophotometer. The IC50 values were calculated using the Logit method. [1] |
| References |
[1]. Cytotoxic activities of chemical constituents from rhizomes of Anemarrhena asphodeloides and their analogues. Arch Pharm Res. 2015;38(5):598-603. [2]. Timosaponin-BII inhibits the up-regulation of BACE1 induced by ferric chloride in rat retina. BMC Complement Altern Med. 2012 Oct 22;12:189. |
| Additional Infomation |
Melongoside N is a steroid saponin. Officinalisinin I has been reported in Solanum melongena with data available. Timosaponin BII is a steroidal saponin (furostanol type) isolated from the rhizomes of Anemarrhena asphodeloides. [1] Timosaponin BII was one of the major constituents in the crude extract used in the study. [1] In the study, Timosaponin BII (1.0 g) was subjected to acid hydrolysis using a 0.5% nitric acid aqueous solution at 100°C for 4 hours to produce several derivatives (2a-2f) for structure-activity relationship studies. [1] Preliminary structure-activity relationship analysis suggested that for the timosaponins, a saturated E-ring significantly reduced cytotoxic activity, while diglycosylation at the C-3 position appeared to increase activity. Whether the F-ring of the saponin was closed (spirostanol) or open (furostanol) did not have an important influence on its activity, as evidenced by the similar potent activities of Timosaponin BII (furostanol) and Timosaponin AIII (spirostanol). [1] |
Solubility Data
| Solubility (In Vitro) |
H2O : ~100 mg/mL (~108.57 mM) DMSO : ~100 mg/mL (~108.57 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (2.71 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (2.71 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (2.71 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 50 mg/mL (54.28 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.0857 mL | 5.4285 mL | 10.8569 mL | |
| 5 mM | 0.2171 mL | 1.0857 mL | 2.1714 mL | |
| 10 mM | 0.1086 mL | 0.5428 mL | 1.0857 mL |