Physicochemical Properties
| Molecular Formula | C15H24N2O4S |
| Molecular Weight | 328.42706 |
| Exact Mass | 328.146 |
| CAS # | 51012-32-9 |
| Related CAS # | Tiapride hydrochloride;51012-33-0 |
| PubChem CID | 5467 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.15 g/cm3 |
| Boiling Point | 498.1ºC at 760 mmHg |
| Melting Point | 120ºC |
| Flash Point | 255.1ºC |
| LogP | 2.825 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 22 |
| Complexity | 443 |
| Defined Atom Stereocenter Count | 0 |
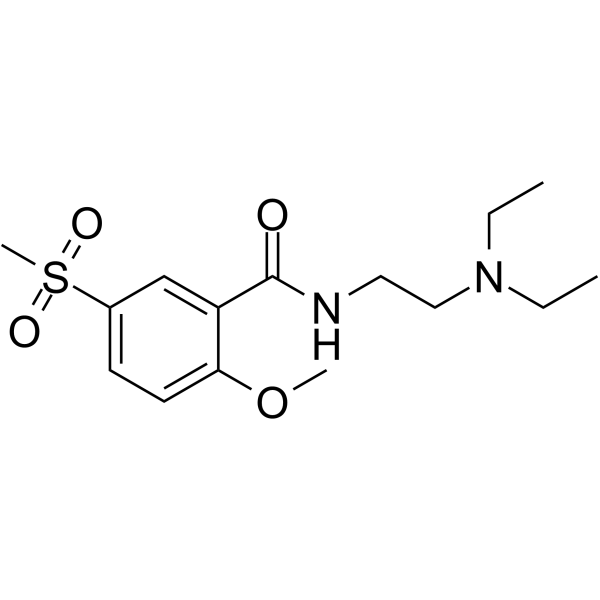
| SMILES | CCN(CCNC(C1=C(OC)C=CC(S(=O)(C)=O)=C1)=O)CC |
| InChi Key | JTVPZMFULRWINT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C15H24N2O4S/c1-5-17(6-2)10-9-16-15(18)13-11-12(22(4,19)20)7-8-14(13)21-3/h7-8,11H,5-6,9-10H2,1-4H3,(H,16,18) |
| Chemical Name | N-[2-(diethylamino)ethyl]-2-methoxy-5-methylsulfonylbenzamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion The bioavailability of tiapride is approximately 75 percent. It has a Tmax is 0.4-1.5 hours and Tss is 24-48 hours with 3 time daily dosing. Benzamide and its derivatives are highly water-soluble but known to cross the blood-brain barrier, necessitating carrier-mediated transport. Urine (70% as unchanged tiapride) Tiapride distributes rapidly and exhibits virtually no binding to plasma proteins, giving it a relatively high volume of distribution 16.6 l/h. Metabolism / Metabolites Tiapride is minimally metabolized in humans, 70 % of the drug is eliminated in unchanged form in the urine within 24 hours. Only low concentration of N-desethyl tiapride and tiapride N-oxide and no phase II metabolites were detected. Biological Half-Life 2.9–3.6 hours |
| Toxicity/Toxicokinetics |
Protein Binding Negligible |
| Additional Infomation |
N-[2-(diethylamino)ethyl]-2-methoxy-5-methylsulfonylbenzamide is a member of benzamides. Tiapride is a selective D2 and D3 dopamine receptor blocker in the brain. A benzamide derivative that is used as a dopamine antagonist. See also: Tiapride Hydrochloride (annotation moved to). Drug Indication Tiapride is indicated for the treatment of a variety of neurological and psychiatric disorders including dyskinesia, alcohol withdrawal syndrome, negative symptoms of psychosis, and agitation and aggression in the elderly. Mechanism of Action Tiapride is a selective dopamine D2 and D3 receptor antagonist, offering an advantage over other neuroleptic drugs, such as haloperidol and risperidone, which bind a range of targets including four of the five known dopamine receptor subtypes (D1-4), serotonin (5-HT2A, 2C), α1- and α2-adrenergic, and histamine H1 receptors. Compared to these drugs, tiapride has a relatively moderate affinity for its target receptors, displacing 50 percent of 3H-raclopride binding at a concentration of 320 nM at D2 receptors and a concentration of 180 nM at D3 receptors. Pharmacodynamics Tiapride has a high degree of regional selectivity for limbic areas. One study found that tiapride shows over three times as much affinity for limbic areas than striatal areas as opposed to the near equal selectivity for limbic and striatal regions shown by haloperidol. Another study in rats found tiapride's affinity for the septum, a limbic region, to be over thirty times as high as for the striatum. Efficacy at the D2 receptor is moderate, with 80 percent of receptors occupied even in the presence of excess tiapride concentrations. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0448 mL | 15.2239 mL | 30.4479 mL | |
| 5 mM | 0.6090 mL | 3.0448 mL | 6.0896 mL | |
| 10 mM | 0.3045 mL | 1.5224 mL | 3.0448 mL |