Thevetiaflavone is a naturally occurring and bioactive flavonoid that can upregulate the expression of Bcl‑2 and downregulate that of Bax and caspase‑3.
Physicochemical Properties
| Molecular Formula | C16H12O5 |
| Molecular Weight | 284.26300 |
| Exact Mass | 284.068 |
| CAS # | 29376-68-9 |
| PubChem CID | 5315202 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 567.9±50.0 °C at 760 mmHg |
| Melting Point | 263℃ |
| Flash Point | 218.3±23.6 °C |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.669 |
| LogP | 2.58 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 21 |
| Complexity | 424 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | YQABHAHJGSNVQR-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H12O5/c1-20-14-6-11(18)7-15-16(14)12(19)8-13(21-15)9-2-4-10(17)5-3-9/h2-8,17-18H,1H3 |
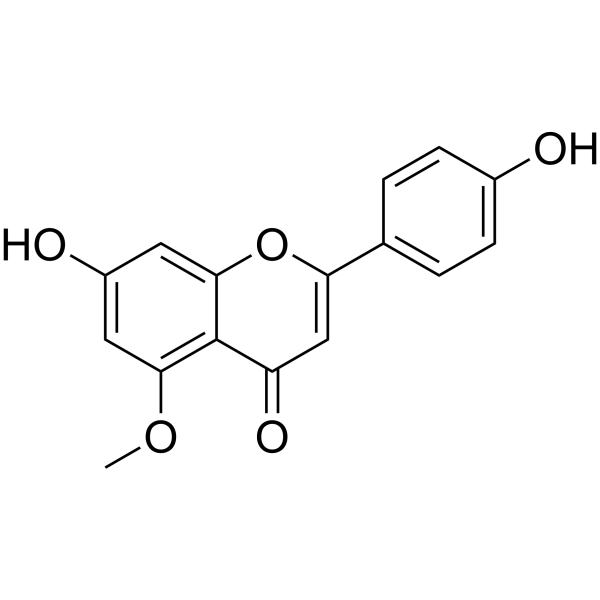
| Chemical Name | 7-hydroxy-2-(4-hydroxyphenyl)-5-methoxychromen-4-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- ROS-mediated mitochondrial dysfunction [1] |
| ln Vitro |
Thevetiaflavone is a natural flavonoid derived from Wikstroemia indica, which can promote cell viability and block the leakage of lactate dehydrogenase from the cytoplasm. Further examination of this process demonstrated that Thevetiaflavone lowers the excessive production of ROS and improves ROS-mediated mitochondrial dysfunction, including the collapse of mitochondrial membrane potential and the opening of mitochondrial permeability transition pores. Thevetiaflavone decreases intracellular Ca2+ levels, which is intimately tied to mitochondrial function and interaction with ROS. In addition, in addition to enhancing the activity of caspase-3, Thevetiaflavone also prevents PC12 cell death by up-regulating the expression of Bcl-2 and down-regulating the expression of Bax and caspase-3. These data further highlight the in vivo protective properties of vetiaflavone and its therapeutic applicability [1]. - Thevetiaflavone protected PC12 cells against oxygen-glucose deprivation/reoxygenation (OGD/R)-induced injury in a dose-dependent manner. At concentrations of 1, 5, and 10 μM, it increased cell viability (measured by MTT assay) by 12.3%, 28.5%, and 42.7%, respectively, compared to the OGD/R model group [1] - The compound significantly reduced intracellular reactive oxygen species (ROS) accumulation in OGD/R-treated PC12 cells. At 10 μM, ROS levels were decreased by 56.8% compared to the model group, as detected by DCFH-DA fluorescence staining [1] - Thevetiaflavone restored mitochondrial membrane potential (ΔΨm) in OGD/R-injured PC12 cells. JC-1 staining showed that the ratio of red/green fluorescence (indicative of ΔΨm) was increased by 35.2% (5 μM) and 61.4% (10 μM) compared to the model group [1] - It inhibited OGD/R-induced apoptosis of PC12 cells. Flow cytometry analysis revealed that the apoptotic rate was reduced from 38.6% (model group) to 26.3% (5 μM) and 15.7% (10 μM) after treatment with Thevetiaflavone [1] - The compound upregulated the activity of endogenous antioxidant enzymes: at 10 μM, superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) activities were increased by 48.3% and 52.6%, respectively, compared to the model group [1] - Thevetiaflavone decreased malondialdehyde (MDA) content (a marker of lipid peroxidation) in OGD/R-treated PC12 cells by 41.2% (5 μM) and 63.5% (10 μM) [1] - Western blot analysis showed that Thevetiaflavone upregulated Bcl-2 protein expression and downregulated Bax protein expression, increasing the Bcl-2/Bax ratio by 1.8-fold (5 μM) and 3.2-fold (10 μM) compared to the model group. It also reduced the release of cytochrome c (Cyt c) from mitochondria to cytoplasm [1] |
| Cell Assay |
- Cell viability (MTT) assay: PC12 cells were seeded in 96-well plates at 5×10³ cells/well and cultured overnight. Cells were divided into control group (normal glucose and oxygen), OGD/R model group (glucose-free medium, 95% N₂ + 5% CO₂ for 4 h, then reoxygenation for 24 h), and Thevetiaflavone treatment groups (pretreated with 1, 5, 10 μM Thevetiaflavone for 2 h before OGD/R). After reoxygenation, MTT reagent was added, and absorbance was measured at 490 nm to calculate cell viability [1] - ROS detection assay: PC12 cells were seeded in 6-well plates and treated as above. After reoxygenation, DCFH-DA probe was added and incubated for 30 min at 37°C. Cells were washed with PBS, and fluorescence intensity (ex/em = 488/525 nm) was measured by flow cytometry to quantify ROS levels [1] - Mitochondrial membrane potential assay: Treated PC12 cells were stained with JC-1 dye for 20 min at 37°C. After washing, red (aggregated JC-1, high ΔΨm) and green (monomeric JC-1, low ΔΨm) fluorescence intensities were detected by flow cytometry, and the red/green fluorescence ratio was calculated [1] - Apoptosis assay: PC12 cells were treated as described, harvested, and stained with Annexin V-FITC and propidium iodide (PI) for 15 min in the dark. Apoptotic cells were quantified by flow cytometry, distinguishing early (Annexin V⁺/PI⁻) and late (Annexin V⁺/PI⁺) apoptosis [1] - Antioxidant enzyme and MDA assay: PC12 cells were lysed after treatment, and the supernatant was collected. SOD and GSH-Px activities were measured by colorimetric methods, and MDA content was determined using a thiobarbituric acid reaction assay [1] - Western blot assay: Mitochondrial and cytoplasmic fractions of treated PC12 cells were isolated. Proteins (Bcl-2, Bax, Cyt c, β-actin) were separated by SDS-PAGE, transferred to membranes, and incubated with primary and secondary antibodies. Band intensities were quantified by densitometry [1] |
| References |
[1]. Thevetiaflavone from Wikstroemia indica ameliorates PC12 cells injury induced by OGD/R via improving ROS-mediated mitochondrial dysfunction. Mol Med Rep. 2017 Dec;16(6):9197-9202. |
| Additional Infomation |
Thevetiaflavone has been reported in Thevetia neriifolia, Heliotropium sinuatum, and other organisms with data available. - Thevetiaflavone is a flavonoid compound isolated from Wikstroemia indica [1] - The neuroprotective mechanism of Thevetiaflavone involves inhibiting ROS overproduction, improving mitochondrial function (restoring ΔΨm, reducing Cyt c release), enhancing antioxidant capacity (upregulating SOD and GSH-Px), and suppressing apoptosis (regulating Bcl-2/Bax pathway) [1] - Thevetiaflavone shows potential as a therapeutic agent for ischemic neuronal injury, as it ameliorates OGD/R-induced PC12 cell damage, a cellular model of cerebral ischemia-reperfusion injury [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5179 mL | 17.5895 mL | 35.1791 mL | |
| 5 mM | 0.7036 mL | 3.5179 mL | 7.0358 mL | |
| 10 mM | 0.3518 mL | 1.7590 mL | 3.5179 mL |