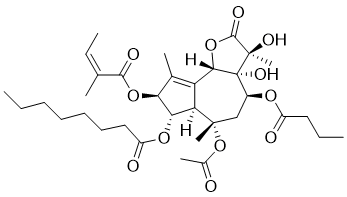
Thapsigargin is a novel and potent non-competitive inhibitor of the sarco/endoplasmic reticulum Ca2+ ATPase (SERCA) and an Endoplasmic Reticulum (ER) stress inducing agent. It is a potent, cell-permeable, IP3-independent intracellular calcium releaser, that works by blocking the transient increase in intracellular Ca2+ induced by angiostatin and endostatin, and inducing apoptosis by disrupting intracellular free Ca2+ levels.
Physicochemical Properties
| Molecular Formula | C34H50O12 |
| Molecular Weight | 650.75 |
| Exact Mass | 650.33 |
| Elemental Analysis | C, 62.75; H, 7.74; O, 29.50 |
| CAS # | 67526-95-8 |
| PubChem CID | 446378 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 699.6±55.0 °C at 760 mmHg |
| Flash Point | 209.0±25.0 °C |
| Vapour Pressure | 0.0±5.0 mmHg at 25°C |
| Index of Refraction | 1.540 |
| LogP | 6.61 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 17 |
| Heavy Atom Count | 46 |
| Complexity | 1270 |
| Defined Atom Stereocenter Count | 8 |
| SMILES | C(C(=O)O[C@H]1[C@]2([H])[C@@](OC(C)=O)(C[C@@H]([C@@]3([C@](C)(O)C(=O)O[C@]3(C2=C(C)[C@@H]1OC(=O)/C(/C)=C\C)[H])O)OC(CCC)=O)C)CCCCCC |
| InChi Key | IXFPJGBNCFXKPI-FSIHEZPISA-N |
| InChi Code | InChI=1S/C34H50O12/c1-9-12-13-14-15-17-24(37)43-28-26-25(20(5)27(28)44-30(38)19(4)11-3)29-34(41,33(8,40)31(39)45-29)22(42-23(36)16-10-2)18-32(26,7)46-21(6)35/h11,22,26-29,40-41H,9-10,12-18H2,1-8H3/b19-11-/t22-,26+,27-,28-,29-,32-,33+,34+/m0/s1 |
| Chemical Name | (3S,3aR,4S,6S,6aR,7S,8S,9bS)-6-(acetyloxy)-4-(butyryloxy)-3,3a-dihydroxy-3,6,9-trimethyl-8-{[(2Z)-2-methylbut-2-enoyl]oxy}-2-oxo-2,3,3a,4,5,6,6a,7,8,9b-decahydroazuleno[4,5-b]furan-7-yl octanoate |
| Synonyms | thapsigargin; 67526-95-8; MFCD00083511; Z96BQ26RZD; CHEMBL96926; CHEBI:9516; (-)-Thapsigargin; (3S,3aR,4S,6S,6aR,7S,8S,9bS)-6-(acetyloxy)-4-(butanoyloxy)-3,3a-dihydroxy-3,6,9-trimethyl-8-{[(2Z)-2-methylbut-2-enoyl]oxy}-2-oxo-2,3,3a,4,5,6,6a,7,8,9b-decahydroazuleno[4,5-b]furan-7-yl octanoate; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Ca2+-ATPase |
| ln Vitro | Thapsigargin (0.001-1 μM; for 2 and 4 days) decreases cell proliferation of MH7A human rheumatoid arthritis synoviocytes in a time- and dose-dependent manner [2]. Thapsigargin (0.001-1 μM; for 2 and 4 days). Thapsigargin (0.001-1 μM; for 2 and 4 days) inhibits mTOR activity and increases cyclin D1 expression in MH7A cells [2]. Thapsigargin reduces Ca2+ entrance into human neutrophils [1]. Thapsigargin inhibits carbachol-evoked [Ca2+]i-transients with (IC50=0.353 nM) or without (IC50=0.448 nM) KCl prestimulation, but not in the absence. An extra minor component was found with KCl prestimulation, resulting in low fractionation (IC50=4814 nM). In contrast, KCl-induced transient changes in [Ca2+]i exhibited just one component, with a very low shift to Thapsigargin in the absence of the mean of both components (IC50=3343 nM) and the presence of carbachol prestimulation (IC50 =6858 nM)[3]. Additionally, thapsigargin inhibits TNF-α via phosphorylating p38 MAPK through SOCE influx of Ca2+. |
| ln Vivo |
Thapsigargin (injection; 0.25 μg/g, 0.5 μg/g, and 1 μg/g; 24 hours) caused a 2- to 5-fold increase in the expression of proinflammatory chemokines. The ability to elicit systemic immunological responses is more sensitive in thapsigargin [4]. To assess ER stress in vivo, we performed interperitoneal injection of either thapsigargin (TG) (0.5ug/g/body weight) or TUN (1ug/g/body weight) in mice. The dose selected for TUN in vivo is in the mid-range of doses used by others and does not elicit cytotoxic effects as assessed by mouse survival from previous studies. Unfortunately, choosing a thapsigargin (TG) dose in vivo has been difficult as literature of thapsigargin (TG) use in vivo is scarce. In order to proceed with using thapsigargin (TG) in a mouse model to evaluate the effectiveness of thapsigargin (TG) in ER stress induction, we needed to demonstrate that our dose of TG (0.5ug/g/body) did not compromise mouse survival. In fact, we found that using thapsigargin (TG) at a dose of 0.5ug/g/body weight was safe and did not elicit any adverse effects on survival in these mice (Supplementary Figure 2). We then examined the induction of ER stress in the liver and adipose tissue of mice treated with either TG or TUN, by assessing the expression of key ER stress and UPR markers.[4] |
| Enzyme Assay |
Thapsigargin, an inhibitor of microsomal Ca(2+)-ATPase, is an important tool in inducing store-regulated Ca2+ influx. In the present paper we show that, at concentrations above 500 nM, thapsigargin also has an opposite effect: it inhibits store-regulated Ca2+ influx into Fura-2-loaded human neutrophil granulocytes. As thapsigargin has been frequently applied at concentrations up to 2 microM, its inhibitory action on plasma-membrane Ca2+ fluxes deserves consideration.[1] Measurement of [Ca2+], Unless otherwise stated, 106 Fura-2-loaded cells were suspended in 3 ml of Ca2+ medium. Changes in fluorescence were recorded in a Deltascan dual-wavelength spectrofluorimeter using 340 nm and 380 nm for excitation and 505 nm for emission wavelength. Measurements were made at 37 'C with continuous stirring. [Ca2+], was calculated from the ratio of fluorescence excited at 340 and 380 nm, by the method detailed in [18]. Measurement of Mn2+ influx This measurement was performed under the same conditions as for [Ca2+1] except that the excitation wavelength was 360 nm, at which fluorescence of Fura-2 is independent of [Ca2+]. Mn2+ influx was initiated by addition of 100 ,uM MnCl2. |
| Cell Assay |
Cell proliferation assay [2] Cell Types: MH7A human rheumatoid arthritis synoviocytes Tested Concentrations: 0.001, 0.1 and 1 μM Incubation Duration: 2 and 4 days Experimental Results: Prevents cell proliferation in a. time- and dose-dependent manner. Apoptosis analysis[2] Cell Types: MH7A human rheumatoid arthritis synovial cells Tested Concentrations: 0.001, 0.1 and 1 μM Incubation Duration: 2 days and 4 days Experimental Results: Apoptosis was induced in a time and dose dependent manner. Western Blot Analysis[2] Cell Types: MH7A Human Rheumatoid Arthritis Synovial Cells Tested Concentrations: 0.001, 0.1 and 1 μM Incubation Duration: 2 and 4 Days Experimental Results: Impaired mTOR activity and resulted in cyclin D1 expression |
| Animal Protocol |
Animal/Disease Models: Male balb/c (Bagg ALBino) mouse (20-25 g) [4] Doses: 0.25 ug/g, 0.5 ug/g, and 1 ug/g Route of Administration: Injection; 24 hour Experimental Results: Increased chemokine and pro-inflammatory expression 2 to 5 times. Induction of ER stress in vivo[4] Male Balb/c mice were housed and cared in accordance with the Guide for the Care and Use of Laboratory Animals. Tunicamycin and thapsigargin (TG) were purchased from Sigma and dissolved in dimethyl sulfoxide (DMSO) and diluted in sterile 150mM dextrose to obtain a concentration of 10μg/μl. Male Balb/c mice (20-25g) were injected intraperitoneally with tunicamycin solution (1μg/g body mass) as described previously. For thapsigargin solution a dose response was conducted using (0.25ug/g, 0.5ug/g and 1ug/g body mass). As controls, mice were injected intraperitoneally with control buffer (150mM dextrose containing 1% DMSO). Adipose and liver tissues were harvested 24hrs post treatment, where as liver tissues utilized for Oil Red O staining was harvested 5 days post treatment with the ER stress inducing agents. |
| References |
[1]. Thapsigargin inhibits Ca2+ entry into human neutrophil granulocytes. Biochem J. 1995 Jan 15;305 ( Pt 2):525-8. [2]. Effects of thapsigargin on the proliferation and survival of human rheumatoid arthritis synovialcells. ScientificWorldJournal. 2014 Feb 9;2014:605416. [3]. Differential thapsigargin-sensitivities and interaction of Ca2+ stores in human SH-SY5Y neuroblastoma cells. Brain Res. 2004 Jun 18;1011(2):177-86. [4]. Modeling Acute ER Stress in Vivo and in Vitro. Shock. 2017 Apr;47(4):506-513. [5]. Inhibiting coronavirus replication in cultured cells by chemical ER stress. bioRxiv 2020.08.26.266304;. [6]. Store-operated calcium entry (SOCE) contributes to phosphorylation of p38 MAPK and suppression of TNF-α signalling in the intestinal epithelial cells. Cell Signal. 2019 Nov;63:109358. |
| Additional Infomation |
Thapsigargin is an organic heterotricyclic compound that is a hexa-oxygenated 6,7-guaianolide isolated fron the roots of Thapsia garganica L., Apiaceae. A potent skin irritant, it is used in traditional medicine as a counter-irritant. Thapsigargin inhibits Ca(2+)-transporting ATPase mediated uptake of calcium ions into sarcoplasmic reticulum and is used in experimentation examining the impacts of increasing cytosolic calcium concentrations. It has a role as an EC 3.6.3.8 (Ca(2+)-transporting ATPase) inhibitor and a calcium channel blocker. It is a sesquiterpene lactone, an organic heterotricyclic compound and a butyrate ester. Thapsigargin has been reported in Thapsia gymnesica, Thapsia garganica, and Thapsia villosa with data available. A sesquiterpene lactone found in roots of THAPSIA. It inhibits SARCOPLASMIC RETICULUM CALCIUM-TRANSPORTING ATPASES. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~50 mg/mL (~76.83 mM) H2O : < 0.1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: 5 mg/mL (7.68 mM) in 10% DMSO + 90% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (3.20 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (3.20 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5367 mL | 7.6834 mL | 15.3669 mL | |
| 5 mM | 0.3073 mL | 1.5367 mL | 3.0734 mL | |
| 10 mM | 0.1537 mL | 0.7683 mL | 1.5367 mL |