Tezosentan (RO610612; Veletr; RO-610612) is a novel, non-selective and dual antagonist/inhibitor of endothelian (ETA/ETB) receptor (pA2s of 9.5, 7.7 for ETA and ETB) with the potential to be used for the treatment of congestive heart failure. Tezosentan acts as a vasodilator and was designed as a therapy for patients with acute heart failure. Tezosentan inhibits uptake of proinflammatory endothelin-1 in stenotic aortic valves. Tezosentan increases nitric oxide signaling via enhanced hydrogen peroxide generation in lambs with surgically induced acute increases in pulmonary blood flow. Tezosentan reduces the renal injury induced by abdominal aortic ischemia-reperfusion in rats.
Physicochemical Properties
| Molecular Formula | C27H27N9O6S |
| Molecular Weight | 605.62498 |
| Exact Mass | 605.18 |
| CAS # | 180384-57-0 |
| PubChem CID | 151174 |
| Appearance | Off-white to gray solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 761.2±70.0 °C at 760 mmHg |
| Flash Point | 414.2±35.7 °C |
| Vapour Pressure | 0.0±2.7 mmHg at 25°C |
| Index of Refraction | 1.634 |
| LogP | 0.89 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 12 |
| Heavy Atom Count | 43 |
| Complexity | 962 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | TUYWTLTWNJOZNY-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C27H27N9O6S/c1-16(2)18-8-9-22(29-15-18)43(38,39)34-26-23(42-21-7-5-4-6-20(21)40-3)27(41-13-12-37)31-24(30-26)17-10-11-28-19(14-17)25-32-35-36-33-25/h4-11,14-16,37H,12-13H2,1-3H3,(H,30,31,34)(H,32,33,35,36) |
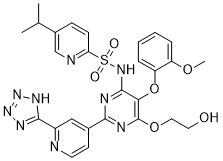
| Chemical Name | N-[6-(2-hydroxyethoxy)-5-(2-methoxyphenoxy)-2-[2-(2H-tetrazol-5-yl)pyridin-4-yl]pyrimidin-4-yl]-5-propan-2-ylpyridine-2-sulfonamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Tezosentan's ET receptor affinity was assessed in a variety of cell types and tissues. The selective binding of 125I-labeled ET-1 to ETA receptors is inhibited by tesosentan, with an inhibitory potency (Ki) of 18 nM on insect cell membranes infected with baculovirus and 0.3 nM on CHO cells. Similarly, tezosentan has an inhibitory affinity of 10 to 21 nM for the specific binding of 125I-labeled ET-1, ET-3, or sarafotoxin S6c to the ETB receptor. Tezosentan did not show any binding inhibitory activity at doses up to 1 μM in 27 radioligand binding experiments that varied with ET binding. Less than 20% mild inhibition of H1 centers, serotonin 2A, and vasopressin V1 receptors is produced by tezosentan (1 μM) [1]. |
| ln Vivo | In Wistar rats, the pressor effects of massive ET-1 were dose-dependently reduced by tesosentan (P<0.001 at all doses). At the lowest studied dose of 1 mg/kg, tezosentan reduced the pressor effects of different doses of big ET-1 by 50% to 80%. In these rats, tezosentan by itself had no effect on blood pressure. In rat models of acute renal failure, tezosentan exhibits remarkable efficacy. In rats with acute renal ischemia, ET antagonists have been demonstrated to prevent vasoconstriction and renal failure [1]. |
| ADME/Pharmacokinetics |
Biological Half-Life A pronounced and rapid disposition phase (half-life 6 min), accounting for the major part of the elimination, was followed by a slower phase (half-life 3 h), probably caused by distribution from tissues. |
| References |
[1]. Pharmacology of tezosentan, new endothelin receptor antagonist designed for parenteral use. J Pharmacol Exp Ther. 1999 Aug;290(2):840-6. |
| Additional Infomation |
Tezosentan is an intravenous endothelin receptor A/B antagonist. Tezosentan was initially developed for vasodilation in patients with acute heart failure but studies have shown that it does not assist in the treatment of dyspnea or prevent cardiovascular events. Drug Indication Investigated for use/treatment in congestive heart failure, liver disease, and heart disease. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~82.56 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6512 mL | 8.2559 mL | 16.5117 mL | |
| 5 mM | 0.3302 mL | 1.6512 mL | 3.3023 mL | |
| 10 mM | 0.1651 mL | 0.8256 mL | 1.6512 mL |