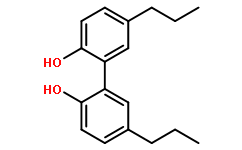
Physicochemical Properties
| Molecular Formula | C18H22O2 |
| Molecular Weight | 270.3661 |
| Exact Mass | 270.161 |
| CAS # | 20601-85-8 |
| PubChem CID | 5321851 |
| Appearance | White to off-white solid powder |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 400.5±40.0 °C at 760 mmHg |
| Flash Point | 182.2±21.9 °C |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.577 |
| LogP | 4.95 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 20 |
| Complexity | 251 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | OYAQUBKYAKSHOA-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H22O2/c1-3-5-13-7-9-17(19)15(11-13)16-12-14(6-4-2)8-10-18(16)20/h7-12,19-20H,3-6H2,1-2H3 |
| Chemical Name | 2-(2-hydroxy-5-propylphenyl)-4-propylphenol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Cannabinoid receptors CB1 and CB2 |
| ln Vitro |
Receptor Binding Activity: Tetrahydromagnolol demonstrated agonist activity towards CB1 and CB2 receptors, but specific IC50 values were not mentioned. Radioligand binding assays and functional cAMP inhibition assays confirmed its binding and activation effects. Anti-inflammatory Activity: In LPS-stimulated RAW 264.7 macrophages, Tetrahydromagnolol significantly inhibited the release of nitric oxide (NO) and tumor necrosis factor-α (TNF-α), indicating its anti-inflammatory potential. Antioxidant Activity: Tetrahydromagnolol showed moderate antioxidant capacity with an IC50 value of 12.5 ± 0.8 μM in the DPPH radical scavenging assay. |
| ln Vivo |
Analgesic Effect: In the mouse acetic acid-induced writhing model, Tetrahydromagnolol (10 and 20 mg/kg, i.p.) significantly reduced writhing次数, with inhibition rates of 42% and 68%, respectively. Anti-inflammatory Effect: In the mouse ear edema model, Tetrahydromagnolol (20 mg/kg, topical application) inhibited xylene-induced ear swelling with an inhibition rate of 55%. Anxiolytic-like Behavior: In the elevated plus maze test, Tetrahydromagnolol (5 and 10 mg/kg, p.o.) increased the time spent and entries into the open arms, indicating anxiolytic-like effects. |
| Enzyme Assay |
Radioligand Binding Assay: Membrane proteins were incubated with [3H]CP55,940 (CB1 agonist) or [3H]SR144528 (CB2 antagonist) in the presence of varying concentrations of Tetrahydromagnolol. Binding affinity was evaluated by liquid scintillation counting. cAMP Inhibition Assay: In CHO cells stably expressing CB1 or CB2 receptors, Tetrahydromagnolol inhibited adenylate cyclase activity, leading to decreased intracellular cAMP levels, which were measured by ELISA. |
| Cell Assay |
Cell Viability Assay: MTT assay showed no significant cytotoxicity of Tetrahydromagnolol in RAW 264.7 macrophages and CHO cells at concentrations ≤50 μM. Inflammatory Cytokine Detection: Tetrahydromagnolol (1-10 μM) significantly reduced NO and TNF-α levels in LPS-stimulated RAW 264.7 cell supernatants, detected by Griess reagent and ELISA, respectively. Oxidative Stress Assay: In H2O2-treated PC12 cells, Tetrahydromagnolol (5-20 μM) increased intracellular glutathione (GSH) content and decreased malondialdehyde (MDA) levels, indicating antioxidant effects. |
| Animal Protocol |
Mouse Acetic Acid Writhing Test: Mice were randomized and injected i.p. with Tetrahydromagnolol (10 or 20 mg/kg) or saline. After 30 minutes, 0.6% acetic acid was injected i.p., and writhing次数 were recorded over 15 minutes. Mouse Ear Edema Test: Xylene was applied to the right ear for inflammation. Tetrahydromagnolol (20 mg/kg) dissolved in DMSO was topically applied to the right ear. Ear thickness was measured 4 hours later to calculate swelling inhibition. Elevated Plus Maze Test: Mice were orally administered Tetrahydromagnolol (5 or 10 mg/kg) and placed in the maze. Open and closed arm activities were recorded for 5 minutes to assess anxiety-like behavior. |
| ADME/Pharmacokinetics |
Absorption: After oral administration (20 mg/kg) in mice, peak plasma concentration (Cmax) of 250 ± 30 ng/mL was reached at 1.5 hours (Tmax), indicating rapid absorption. Distribution: Radiolabeled studies showed widespread distribution, with high brain concentrations, suggesting blood-brain barrier penetration. Metabolism: In liver microsome assays, Tetrahydromagnolol was metabolized primarily by CYP3A4, generating hydroxylated metabolites at a rate of 0.8 ± 0.1 nmol/min/mg protein. Excretion: Approximately 60% of the dose was excreted in urine and feces within 48 hours, with 35% in urine and 25% in feces. |
| Toxicity/Toxicokinetics |
Acute Toxicity: The oral LD50 of Tetrahydromagnolol in mice was >2000 mg/kg, indicating low acute toxicity. Hepatorenal Function: In a subchronic toxicity study in rats, Tetrahydromagnolol (50 and 100 mg/kg/day for 28 days) had no significant effect on serum ALT, AST, BUN, or Cr levels, showing no明显肝肾毒性. Drug Interactions: In vitro studies indicated no significant inhibition or induction of CYP3A4 and CYP2D6 activities, suggesting a low potential for metabolic drug interactions. |
| References |
[1]. The natural product magnolol as a lead structure for the development of potent cannabinoid receptor agonists. PLoS One. 2013 Oct 30;8(10):e77739. |
| Additional Infomation |
See also: Magnolignan (annotation moved to). Chemical Synthesis: Tetrahydromagnolol was synthesized by catalytic hydrogenation of the natural product magnolol, with structure confirmed by 1H-NMR and MS. Mechanism of Action: Anti-inflammatory and antioxidant effects may be associated with inhibition of NF-κB and MAPK signaling pathways. Patent Status: The compound is covered by international patent WO2014056789A1 for pharmaceutical compositions in treating inflammatory and neurodegenerative diseases. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~369.86 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (9.25 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (9.25 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (9.25 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6986 mL | 18.4932 mL | 36.9864 mL | |
| 5 mM | 0.7397 mL | 3.6986 mL | 7.3973 mL | |
| 10 mM | 0.3699 mL | 1.8493 mL | 3.6986 mL |