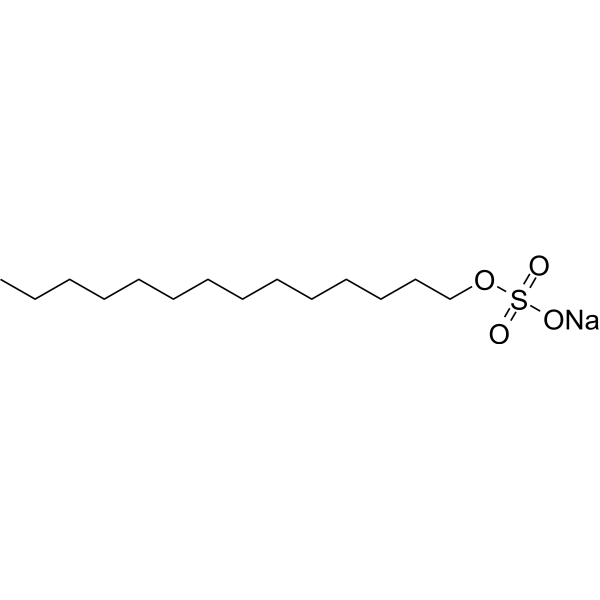
Physicochemical Properties
| Molecular Formula | C14H29NAO4S |
| Molecular Weight | 316.43 |
| Exact Mass | 316.168 |
| CAS # | 1191-50-0 |
| PubChem CID | 23665770 |
| Appearance | White to off-white solid powder |
| Melting Point | 199 °C |
| LogP | 5.245 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 14 |
| Heavy Atom Count | 20 |
| Complexity | 275 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CCCCCCCCCCCCCCOS(=O)(=O)[O-].[Na+] |
| InChi Key | UPUIQOIQVMNQAP-UHFFFAOYSA-M |
| InChi Code | InChI=1S/C14H30O4S.Na/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-18-19(15,16)17;/h2-14H2,1H3,(H,15,16,17);/q;+1/p-1 |
| Chemical Name | sodium;tetradecyl sulfate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the clinical use of sodium tetradecyl sulfate during breastfeeding. Although sodium tetradecyl sulfate is unlikely to adversely affect the breastfed infant, international guidelines recommend that breastfeeding be withheld for 2 days after sclerotherapy. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. |
| References | [1]. Tan KT, et al. Percutaneous sodium tetradecyl sulfate sclerotherapy for peripheral venous vascular malformations: a single-center experience. J Vasc Interv Radiol. 2007;18(3):343-351. |
| Additional Infomation |
An anionic surface-active agent used for its wetting properties in industry and used in medicine as an irritant and sclerosing agent for hemorrhoids and varicose veins. An anionic surface-active agent used for its wetting properties in industry and used in medicine as an irritant and sclerosing agent for hemorrhoids and varicose veins. Drug Indication For the treatment of small uncomplicated varicose veins of the lower extremities that show simple dilation with competent valves. Mechanism of Action Sodium tetradecyl sulfate is a potent toxin for endothelial cells in that brief exposure to even low concentrations are effective in stripping endothelium over a considerable distance and exposing highly thrombogenic endothelium in the process. Diluted sodium tetradecyl sulfate is also able to induce a hypercoagulable state, possibly by selective inhibition of protein C, and can also promote platelet aggregation. Pharmacodynamics Sodium tetradecyl sulfate is an anionic surfactant which occurs as a white, waxy solid. It is used as a sclerosing agent in sclerotherapy. Sclerotherapy is an injection of a sclerosing agent directly through the skin into a lesion and is used primarily for slow-flow vascular anomalies, particularly for venous malformation and lymphatic malformation. Intravenous injection causes intima inflammation and thrombus formation. This usually occludes the injected vein. Subsequent formation of fibrous tissue results in partial or complete vein obliteration that may or may not be permanent. |
Solubility Data
| Solubility (In Vitro) | DMSO: 100 mg/mL (316.03 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.90 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.90 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (7.90 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1603 mL | 15.8013 mL | 31.6026 mL | |
| 5 mM | 0.6321 mL | 3.1603 mL | 6.3205 mL | |
| 10 mM | 0.3160 mL | 1.5801 mL | 3.1603 mL |