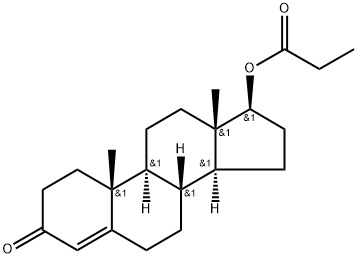
Testosterone propionate is a synthetic androgen and anabolic steroid (AAS) medication sold under the brand name Testoviron among others, and used as a controlled release drug for the treatment of low testosterone levels in men. It has also been used to treat breast cancer in women. It is given by injection into muscle usually once every two to three days. Testosterone propionate is a single ester testosterone compound and represents one of the most important testosterone compounds every manufactured. Although synthetic it is a perfect replica of the primary naturally produced male androgen testosterone. By design, the hormone is attached to the Propionate (propionic acid) ester, a small/short ester that enables the hormone’s release time to be controlled. Without an ester, the hormone would disperse and dissipate rapidly post administration. By attaching the ester, this promotes a controlled release and allows the individual to inject the hormone less frequently. Once Testosterone Propionate is injected, the ester slowly begins to detach from the hormone. As the ester is detached the testosterone hormone begins to release into the blood. The half-life of Testosterone Propionate is approximately two days, which is substantially longer than ester free testosterone, which carries a half-life a little less than 24 hours.
Testosterone propionate is regulated as a Schedule III compound in the USA. This product is intended for research use only.Physicochemical Properties
| Molecular Formula | C22H32O3 |
| Molecular Weight | 344.495 |
| Exact Mass | 344.235 |
| CAS # | 57-85-2 |
| PubChem CID | 5995 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 454.6±45.0 °C at 760 mmHg |
| Melting Point | 118-123 °C |
| Flash Point | 196.3±28.8 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.538 |
| LogP | 4.9 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 25 |
| Complexity | 621 |
| Defined Atom Stereocenter Count | 6 |
| SMILES | O(C(C([H])([H])C([H])([H])[H])=O)[C@@]1([H])C([H])([H])C([H])([H])[C@@]2([H])[C@]3([H])C([H])([H])C([H])([H])C4=C([H])C(C([H])([H])C([H])([H])[C@]4(C([H])([H])[H])[C@@]3([H])C([H])([H])C([H])([H])[C@@]21C([H])([H])[H])=O |
| InChi Key | PDMMFKSKQVNJMI-BLQWBTBKSA-N |
| InChi Code | InChI=1S/C22H32O3/c1-4-20(24)25-19-8-7-17-16-6-5-14-13-15(23)9-11-21(14,2)18(16)10-12-22(17,19)3/h13,16-19H,4-12H2,1-3H3/t16-,17-,18-,19-,21-,22-/m0/s1 |
| Chemical Name | [(8R,9S,10R,13S,14S,17S)-10,13-dimethyl-3-oxo-1,2,6,7,8,9,11,12,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-17-yl] propanoate |
| Synonyms | Enarmon Androlon Testosterone propionate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Androgen Receptor |
| ln Vitro | As all testosterone esters, testosterone propionate is rapidly hydrolysed into free testosterone in plasma. Testosterone is metabolized to 17-keto steroids through two different pathways. The major active metabolites are estradiol and dihydrotestosterone (DHT).Testosterone propionate is rapidly hydrolysed into testosterone. Testosterone is metabolized to 17-keto steroids through two different pathways. The major active metabolites are estradiol and dihydrotestosterone (DHT). Route of Elimination: About 90% of a dose of testosterone given intramuscularly is excreted in the urine as glucuronic and sulfuric acid conjugates of testosterone and its metabolites; about 6% of a dose is excreted in the feces, mostly in the unconjugated form. |
| ln Vivo | Testosterone propionate is used in veterinary practice in heifers in order to stimulate maximal growth. The administration of testosterone propionate can induce production of proteins related to male sexual development. Clinical trials have shown a decrease in plasma LH after the administration of testosterone propionate. |
| Cell Assay |
Preparation of testosterone propionate (TP) [13] Testosterone propionate (TP) was dissolved in an acetone-water mixture (1:3 v/v) by heating at 70 °C for 15 minutes. The solution was cooled and mixed with complete culture media to obtain TP at 10-10, 10-9, 10-8, 10-7, and 10-6 M at final concentrations. The final volume of acetone in cell culture was 0.3%, and culture medium with 0.3% acetone addition was used as vehicle. BM-MSCs were incubated with either TP at concentrations between 10-6 M and 10-10 M or vehicle for 24 hours while untreated cells were used as control. Cell Proliferation Measurements for MB-MSCs [13] The proliferation rates of BM-MSCs were determined by carboxyfluorescein succinimidyl ester (CFSE) staining, which relied on the equal sharing between two daughter cells upon cell division. For this study, 2x105 BM-MSC were seeded into 6-well tissue culture plates as triplicates and incubated overnight to allow attachment. For labeling cells, the complete medium was discarded, and cells were incubated in 1 ml DMEM containing CFSE at the final concentration of 5 µM at 37 °C for 20 minutes. Media containing CFSE were discarded, and cells were either treated with TP at concentrations between 10-6 M and 10-10 M or vehicle. At the end of the 24-hour incubation period, cells were detached by trypsinization, washed once with DPBS, and read by Beckman Coulter FC500 flow cytometry system. Analyses were performed on CXP software. Cell Viability and Apoptosis Measurements for MB-MSCs [13] Effects of TP at various concentrations on MSCs’ viability and apoptosis rates were determined by Annexin V/Propidium Iodide (PI) staining by ApopNexin Annexin V FITC Apoptosis Kit. This procedure relies on the simultaneous analysis of membrane asymmetry and integrity that are gradually lost during apoptosis. For this purpose, 2x105 BM-MSCs were seeded into 6-well tissue culture plates as triplicates and treated with TP at concentrations between 10-6 M and 10-10 M or vehicle for 24 hours, while untreated cells were used as control. At the end of incubation, cells were detached by trypsinization, washed once with DPBS, and labeled with Annexin V-FITC and PI according to the manufacturer’s instructions. Briefly, cells were suspended in 1 ml Annexin V binding buffer, and then incubated with 3 µl Annexin V FITC and 2 µl PI provided by the kit at room temperature under dark conditions for 15 minutes. The cells were placed on ice at the end of incubation and immediately analyzed with Beckman Coulter FC500 flow cytometry system. Analysis was performed on CXP software. Characterization of BM-MSCs Upon TP Treatment [13] Impact of TP at 10-8 M concentration on the characteristic features of BM-MSCs were determined by investigating cell surface markers and by evaluating BM-MSCs’ differentiation capacities. For immunophenotyping, 2x105 BM-MSCs were seeded into 6-well tissue culture plates as triplicates, incubated in the presence of 10-8 M TP for 24 hours followed by collecting cells with trypsinization and labeling with antibodies abovementioned. Cells were immediately read with Beckman Coulter DxFLEX flow cytometry system and analysis was performed on CytExpert software. For evaluation of BM-MSCs’ differentiation capacities, 1x104 BM-MSCs were seeded into 96-well cell culture plates and once they reached 80% confluency, differentiation was initiated by commercial chondrogenesis, osteogenesis, and adipogenesis kits, either supplemented with 10-8 M TP or not. Differentiation media were replaced twice a week. On the 21st day of differentiation, cells were fixed with 10% neutral-buffered formalin solution. Chondrogenesis, osteogenesis, and adipogenesis were evaluated by Alcian Blue, Alizarin Red, and Oil Red-O staining, respectively. Co-culture of BM-MSCs with K562 Cell Line [13] K562 cell line (ATCC #CCL-243) was maintained in RPMI medium supplemented with 10% FBS and 1% penicillin/streptomycin antibiotic solution. For evaluating cytotoxic effects of BM-MSCs on the K562 cell line, BM-MSCs were seeded into 35-mm cell culture dishes (1x104 cells/dish) and cultured overnight to allow attachment. Then, the medium was discarded, and BM-MSCs were either treated with 10-8 M TP for 24 hours followed by co-culturing with K562 cells (1x105 cells/dish) for an additional 24 hours; otherwise, K562 cells were added on BM-MSCs in the presence of 10-8 M TP and incubated for 24 hours. The effects of TP on the K562 cells were determined by incubating them with medium supplemented with 10-8 M TP for 24 hours, whereas the effects of BM-MSCs on K562 cells were evaluated by co-culturing cells for 24 hours. K562 cells’ viability was determined by labeling them with 4′,6-diamidino-2-phenylindole by incubating the cells for 15 minutes under dark conditions at room temperature. The cells were read with Beckman Coulter DxFLEX flow cytometry system, and analysis was performed with CytExpert software. Confocal Microscopy [13] For the evaluation of BM-MSC polarization and the cellular morphology, upon TP treatment, the cells were seeded in 8-well chamber slides at a density of 2x104 cells per well and incubated overnight to allow attachment, followed by treatment with 10-8 M TP for 24 hours. The slides were fixed and permeabilized as previously described23 and labeled either with anti-CXCL9 PE and anti-CXCL5 APC antibodies or with F-actin probe Phalloidin (Alexa Fluor 555 conjugated) by incubating overnight at 4 °C. Cell nuclei were counterstained with DAPI, and the slides were mounted with FluoroShield medium. Micrographs were taken with a Zeiss LSM 780 confocal microscope. |
| Animal Protocol |
Absorption Testosterone propionate presents a slow absorption from the intramuscular site of administration. This slow absorption is due to the presence of the less polar ester group. The absorption rate of testosterone propionate generates a frequent injection requirement when compared with testosterone enanthate or testosterone cypionate. It presents absorption parameters of AUC and residence time of 180-210 ng h/ml and 40-60 h, respectively. Route of Elimination About 90% of a dose of testosterone given intramuscularly is excreted in the urine as glucuronic and sulfuric acid conjugates of testosterone and its metabolites. From the rest of the dose, approximately 6% of a dose is excreted in the feces, mostly in the unconjugated form. Volume of Distribution The registered volume of distribution for testosterone propionate is in the range of 75-120 L/kg. Clearance Testosterone propionate has a reduced clearance rate compared to testosterone. The reported clearance rate is of approximately 2000 ml/min. Biological Half-Life Testosterone propionate possesses a relatively short half-life compared with other testosterone esters at approximately 4.5 days. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Testosterone propionate presents a slow absorption from the intramuscular site of administration. This slow absorption is due to the presence of the less polar ester group. The absorption rate of testosterone propionate generates a frequent injection requirement when compared with testosterone enanthate or testosterone cypionate. It presents absorption parameters of AUC and residence time of 180-210 ng h/ml and 40-60 h, respectively. About 90% of a dose of testosterone given intramuscularly is excreted in the urine as glucuronic and sulfuric acid conjugates of testosterone and its metabolites. From the rest of the dose, approximately 6% of a dose is excreted in the feces, mostly in the unconjugated form. The registered volume of distribution for testosterone propionate is in the range of 75-120 L/kg. Testosterone propionate has a reduced clearance rate compared to testosterone. The reported clearance rate is of approximately 2000 ml/min. Metabolism / Metabolites As all testosterone esters, testosterone propionate is rapidly hydrolysed into free testosterone in plasma. Testosterone is metabolized to 17-keto steroids through two different pathways. The major active metabolites are estradiol and dihydrotestosterone (DHT). Testosterone propionate is rapidly hydrolysed into testosterone. Testosterone is metabolized to 17-keto steroids through two different pathways. The major active metabolites are estradiol and dihydrotestosterone (DHT). Route of Elimination: About 90% of a dose of testosterone given intramuscularly is excreted in the urine as glucuronic and sulfuric acid conjugates of testosterone and its metabolites; about 6% of a dose is excreted in the feces, mostly in the unconjugated form. Biological Half-Life Testosterone propionate possesses a relatively short half-life compared with other testosterone esters at approximately 4.5 days. |
| Toxicity/Toxicokinetics |
Toxicity Summary The effects of testosterone in humans and other vertebrates occur by way of two main mechanisms: by activation of the androgen receptor (directly or as DHT), and by conversion to estradiol and activation of certain estrogen receptors. Free testosterone (T) is transported into the cytoplasm of target tissue cells, where it can bind to the androgen receptor, or can be reduced to 5α-dihydrotestosterone (DHT) by the cytoplasmic enzyme 5α-reductase. DHT binds to the same androgen receptor even more strongly than T, so that its androgenic potency is about 2.5 times that of T. The T-receptor or DHT-receptor complex undergoes a structural change that allows it to move into the cell nucleus and bind directly to specific nucleotide sequences of the chromosomal DNA. The areas of binding are called hormone response elements (HREs), and influence transcriptional activity of certain genes, producing the androgen effects. Protein Binding Even 98% of testosterone in plasma is bound to sex hormone-binding globulin and 2% remains unbound or bound to albumin and other proteins. |
| References |
[1] Archives of General Psychiatry, 57, 133-140; [2] Personality and Individual Differences, 28, 437-445; [3] Am J Physiol Endocrinol Metab 2003 Jan 7; [4] J Investig Med. 1997 Oct;45(8):441-7; [5] J Clin Endocrinol Metab. 1986 Dec;63(6):1361-4; [6] J Clin Endocrinol Metab. 1997 Feb;82(2):407-13; [7] Am J Physiol Endocrinol Metab. 2002 Mar;282(3):E601-7; [8] Curr Opin Clin Nutr Metab Care. 2004 May;7(3):271-7; [9] Curr Pharm Biotechnol. 2004 Oct;5(5):459-70; [10] Metabolism. 1991 Apr;40(4):368-77; [11]] J Lab Clin Med. 1995 Mar;125(3):326-33; [12] Zhonghua Nan Ke Xue. 2003;9(4):248-51. [13] Balkan Med J. 2023 Mar 8;40(2):117–123. |
| Additional Infomation |
Testosterone propionate appears as odorless white or yellowish-white crystals or a white or creamy-white crystalline powder. (NTP, 1992) Testosterone propionate is a steroid ester. Testosterone propionate is a slower-releasing anabolic steroid with a short half-life. It is a synthetic androstane steroid derivative of testosterone in the form of 17β propionate ester of testosterone. Testosterone propionate was developed initially by Watson labs, and FDA approved on February 5, 1974. Currently, this drug has been discontinued in humans, but the vet application is still available as an OTC. Testosterone Propionate is a short acting oil-based injectable formulation of testosterone. Testosterone inhibits gonadotropin secretion from the pituitary gland and ablates estrogen production in the ovaries, thereby decreasing endogenous estrogen levels. In addition, this agent promotes the maintenance of male sex characteristics and is indicated for testosterone replacement in hypogonadal males. (NCI04) An ester of testosterone with a propionate substitution at the 17-beta position. An ester of TESTOSTERONE with a propionate substitution at the 17-beta position. See also: Testosterone (has active moiety); Estradiol Benzoate; Testosterone Propionate (component of). Drug Indication Testosterone propionate is used in veterinary practice in heifers in order to stimulate maximal growth. Mechanism of Action The effects of testosterone in humans and other vertebrates occur by way of two main mechanisms: by activation of the androgen receptor (directly or as DHT), and by conversion to estradiol and activation of certain estrogen receptors. Free testosterone (T) is transported into the cytoplasm of target tissue cells, where it can bind to the androgen receptor, or can be reduced to 5alpha-dihydrotestosterone (DHT) by the cytoplasmic enzyme 5alpha-reductase. The areas of binding are called hormone response elements (HREs), and influence transcriptional activity of certain genes, producing the androgen effects. Pharmacodynamics The administration of testosterone propionate can induce production of proteins related to male sexual development. Clinical trials have shown a decrease in plasma LH after the administration of testosterone propionate. |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~30 mg/ml DMF: ~30 mg/ml Ethanol: ~2 mg/ml |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9028 mL | 14.5138 mL | 29.0276 mL | |
| 5 mM | 0.5806 mL | 2.9028 mL | 5.8055 mL | |
| 10 mM | 0.2903 mL | 1.4514 mL | 2.9028 mL |