Tebanicline hydrochloride (also known as ABT-594, Ebanicline and ABT594 hydrochloride) is a novel, potent, orally bioavailable nAChR modulator with effective analgesic activity. It inhibits the binding of cytisine to α4β2 neuronal nAChRs with a Ki of 37 pM. Tebanicline is a potent synthetic nicotinic (non-opioid) analgesic drug developed by Abbott as a less toxic analogue of the potent poison dart frog-derived compound epibatidine, which is some 200x stronger than morphine as an analgesic but produces extremely dangerous toxic side effects. Like epibatidine, tebanicline showed potent analgesic activity against neuropathic pain in both animal and human trials, but with far less toxicity than its parent compound. It acts as a partial agonist at neuronal nicotinic acetylcholine receptors, binding to both the α3β4 and the α4β2 subtypes.
Physicochemical Properties
| Molecular Formula | C9H12CL2N2O |
| Molecular Weight | 235.1104 |
| Exact Mass | 234.033 |
| CAS # | 203564-54-9 |
| Related CAS # | Tebanicline dihydrochloride;209326-19-2 |
| PubChem CID | 72193963 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 2.606 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 14 |
| Complexity | 168 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | ClC1C([H])=C([H])C(=C([H])N=1)OC([H])([H])[C@@]1([H])C([H])([H])C([H])([H])N1[H].Cl[H] |
| InChi Key | GYVARJONEFSAJB-OGFXRTJISA-N |
| InChi Code | InChI=1S/C9H11ClN2O.ClH/c10-9-2-1-8(5-12-9)13-6-7-3-4-11-7;/h1-2,5,7,11H,3-4,6H2;1H/t7-;/m1./s1 |
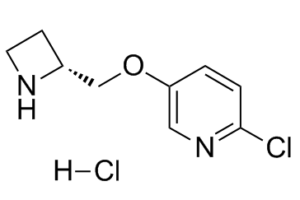
| Chemical Name | 5-[[(2R)-azetidin-2-yl]methoxy]-2-chloropyridine hydrochloride |
| Synonyms | ABT-594 HCl; ABT 594; ABT594; Ebanicline. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Neuronal nicotinic acetylcholine receptor agonist . [3] |
| ln Vitro |
ABT-594 is a potent inhibitor of the binding of [3H](-)-cytisine to alpha 4 beta 2 neuronal nAChRs (Ki = 37 pM, rat brain; Ki = 55 pM, transfected human receptor). ABT-594 has weak affinity in binding assays for adrenoreceptor subtypes alpha-1B、 alpha-2B 、and alpha-2C , and it has negligible affinity (Ki > 1000 nM) for approximately 70 other receptors, enzyme and transporter binding sites.Functionally, tebanicline is an agonist. At the transfected human α4β2 neuronal nAChR in K177 cells, with increased 86Rb efflux as a measure of cation efflux, ABT-594 has an EC+50 value of 140 nM with an intrinsic activitycompared with (−)-nicotine of 130% A-98593 is 2- to 3-fold more potent and displays approximately 50% greater intrinsic activity than ABT-594 in all four functional assays. In terms of potency, ABT-594 is 8- to 64-fold less active than (+/-)-epibatidine and also has less IA in these functional assays. ABT-594 (30 microM) inhibits the release of calcitonin gene-related peptide from C-fibers terminating in the dorsal horn of the spinal cord, an effect mediated via nAChRs. Pharmacologically, ABT-594 has an in vitro profile distinct from that of the prototypic nicotinic analgesic (+/-)-epibatidine, with the potential for substantially reduced side-effect liability and, as such, represents a potentially novel therapeutic approach to pain management.[1] In vitro studies have demonstrated that ABT-594 can attenuate capsaicin-induced release of calcitonin gene-related peptide (CGRP) from spinal cord slices. [4] |
| ln Vivo |
In the hot box and formalin test (i.e., phase 1 and 2), acute treatment with ABT-594 (0.03, 0.1 and 0.3 mumol/kg i.p.) produced significant dose-dependent antinociceptive effects. In the hot box, the efficacy of ABT-594 was maintained after a repeated dosing paradigm (5 days b.i.d.i.p.). ABT-594 was fully efficacious in the formalin test when administered before formalin, and also retained significant efficacy (0.3 mumol/kg i.p.) when administered after formalin injection. The antinociceptive effects of ABT-594 in the hot box and formalin tests were attenuated by pretreatment with the nAChR antagonist, mecamylamine, and in animals treated with the nAChR antagonist chlorisondamine, given centrally (10 micrograms/rat i.c.v. 5 days before), but not in animals pretreated with the opioid receptor antagonist, naltrexone. Acute treatment with ABT-594 produced an initial decrease in open-field locomotor activity, which was absent in animals dosed repeatedly (5 days b.i.d.) with ABT-594. Also, acute treatment with ABT-594 decreased body temperature and decreased the amount of time the animals could maintain balance in an edge-balance test. These effects were no longer present in animals dosed repeatedly with ABT-594. At antinociceptive doses, ABT-594 produced activation of free running EEG in contrast to the sedative-like effects of morphine.Full antinociceptive efficacy was maintained in both the hot box and formalin tests after oral administration, whereas the effects on motoric performance were attenuated. In conclusion, these data demonstrate that ABT-594 is a potent antinociceptive agent with full efficacy in models of acute and persistent pain and that these effects are mediated predominately by an action at central neuronal nAChRs. In addition, antinociceptive effects were maintained after repeated dosing, whereas effects of ABT-594 on motor and temperature measures were attenuated in animals treated repeatedly with ABT-594. [2] ABT-594 produced significant antinociceptive effects in mice against acute noxious thermal stimulation (hot-plate and cold-plate tests) and persistent visceral irritation (abdominal constriction assay). The maximally effective dose in each test was 0.62 μmol/kg (i.p.). The effect peaked at 30 min after i.p. administration and was still present at 60 min, but not at 120 min. The compound was also orally active but about 10-fold less potent than after i.p. administration. The antinociceptive effect was prevented by the noncompetitive neuronal nicotinic acetylcholine receptor antagonist mecamylamine (5 μmol/kg, i.p.) but not by hexamethonium (10 μmol/kg, i.p.) or naltrexone (0.8 μmol/kg, i.p.). The antinociceptive effects of morphine and ABT-594 appeared additive in the hot-plate test. ABT-594 also produced anxiolytic-like effects in the elevated plus-maze test at 0.019 and 0.062 μmol/kg (i.p.). [3] |
| Animal Protocol |
Hot-plate test: Mice were placed in test chambers on a heated (55°C) copper plate, and the latency to jump 10 times was recorded. Test compounds were administered 30 min prior to testing. A cut-off latency of 180 s was used. [3] Cold-plate test: A glass beaker was placed on crushed ice until the bottom temperature reached approximately 1°C. Mice were placed in the beaker, and the latency to engage in shaking/rubbing or jumping behavior was recorded. Compounds were administered 30 min before testing. [3] Abdominal constriction assay: Mice received an i.p. injection of phenyl-p-quinone (68 μmol/kg). Stretching or writhing responses were recorded during a 10-min period starting 5 min after irritant injection. [3] Locomotor activity, motor coordination, and body temperature: Locomotor activity was monitored in an open field for 5 min, followed by rotarod testing (accelerating from 0 to 40 rpm over 120 s) and rectal temperature measurement. Tests were performed 25–35 min after i.p. injection. [3] Elevated plus-maze test: Mice were placed in the center of a plus-maze consisting of two open and two enclosed arms. Time spent on open arms and total distance traveled were recorded for 5 min, starting 30 min after i.p. injection. [3] Ethanol- and pentobarbital-induced hypnotic effects: ABT-594 or saline was injected i.p., followed 15 min later by ethanol (87 mmol/kg) or pentobarbital (160 μmol/kg). The time to regain the righting reflex was recorded as “sleep time.” [3] Respiratory depression assessment: Mice were sacrificed by cervical dislocation, and trunk blood was collected for pCO₂ measurement using a blood gas analyzer. [3] Seizure and lethality assessment: Mice were observed for overt seizure signs for at least 15 min after i.p. injection. Lethality was recorded out to 7 days after dosing. [3] |
| ADME/Pharmacokinetics |
ABT-594 was orally active but about 10-fold less potent than after i.p. administration, suggesting lower oral bioavailability. [3] |
| Toxicity/Toxicokinetics |
The LD₅₀ of ABT-594 in mice was 19.1 μmol/kg (i.p.). The ED₅₀ for overt seizure production was 1.9 μmol/kg (i.p.). ABT-594 significantly increased the duration of ethanol-induced hypnotic effects but did not affect pentobarbital-induced lethality. The compound reduced body temperature and spontaneous exploration at antinociceptive doses but did not reliably impair motor coordination in the rotarod test. [3] |
| References |
[1]. ABT-594 [(R)-5-(2-azetidinylmethoxy)-2-chloropyridine]: a novel, orally effective analgesic acting via neuronal nicotinic acetylcholine receptors: I. In vitro characterization.J Pharmacol Exp Ther. 1998 May;285(2):777-86. [2]. ABT-594 [(R)-5-(2-azetidinylmethoxy)-2-chloropyridine]: a novel, orally effective antinociceptive agent acting via neuronal nicotinic acetylcholine receptors: II. In vivo characterization. J Pharmacol Exp Ther. 1998 May;285(2):787-94. [3]. Antinociceptive effects of the novel neuronal nicotinic acetylcholine receptor agonist, ABT-594, in mice. Eur J Pharmacol. 1998 Apr 3;346(1):23-33. [4]. The role of neuronal nicotinic acetylcholine receptors in antinociception: effects of ABT-594. J Physiol Paris. 1998 Jun-Aug;92(3-4):221-4. |
| Additional Infomation |
ABT-594 is a novel neuronal nicotinic acetylcholine receptor agonist with potent antinociceptive and anxiolytic-like effects in mice. Its antinociceptive effects are mediated through central nicotinic receptors and do not involve opioid receptor activation. The compound shows a broader spectrum of activity and improved safety profile compared to (−)-nicotine and (±)-epibatidine. [3] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.2533 mL | 21.2666 mL | 42.5333 mL | |
| 5 mM | 0.8507 mL | 4.2533 mL | 8.5067 mL | |
| 10 mM | 0.4253 mL | 2.1267 mL | 4.2533 mL |