Physicochemical Properties
| Molecular Formula | C33H50O10 |
| Molecular Weight | 606.74 |
| Exact Mass | 606.34 |
| CAS # | 119757-73-2 |
| PubChem CID | 6439037 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.148g/cm3 |
| Boiling Point | 763.7ºC at 760mmHg |
| Flash Point | 233.9ºC |
| Vapour Pressure | 6.37E-27mmHg at 25°C |
| Index of Refraction | 1.518 |
| LogP | 3.946 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 21 |
| Heavy Atom Count | 43 |
| Complexity | 1090 |
| Defined Atom Stereocenter Count | 0 |
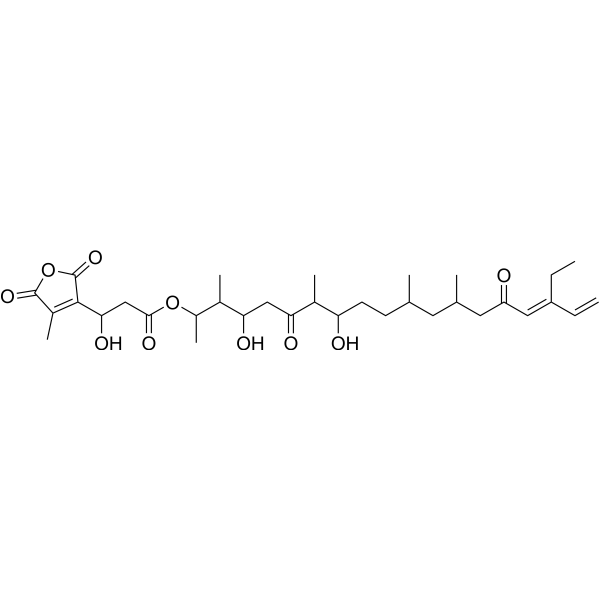
| SMILES | O([H])C([H])(C([H])(C([H])([H])[H])C(C([H])([H])C([H])(C([H])(C([H])([H])[H])C([H])(C([H])([H])[H])OC(C([H])([H])C([H])(C1C(=O)OC(C=1C([H])([H])[H])=O)O[H])=O)O[H])=O)C([H])([H])C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C(/C(/[H])=C(/C([H])=C([H])[H])\C([H])([H])C([H])([H])[H])=O |
| InChi Key | VAIBGAONSFVVKI-IWIPYMOSSA-N |
| InChi Code | InChI=1S/C33H50O10/c1-9-24(10-2)15-25(34)14-19(4)13-18(3)11-12-26(35)21(6)28(37)16-27(36)20(5)23(8)42-30(39)17-29(38)31-22(7)32(40)43-33(31)41/h9,15,18-21,23,26-27,29,35-36,38H,1,10-14,16-17H2,2-8H3/b24-15- |
| Chemical Name | [(16E)-17-ethyl-4,8-dihydroxy-3,7,11,13-tetramethyl-6,15-dioxononadeca-16,18-dien-2-yl] 3-hydroxy-3-(4-methyl-2,5-dioxofuran-3-yl)propanoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | In the murine mixed lymphocyte response (MLR) experiments, tautomycetin (100 ng/ml–10 μg/ml; 48 hours) inhibits T cell growth. TMC has a 100-fold greater amount of MLR inhibition than CsA, and its IC50 is 7.8 nM[1]. Tautomycetin (1 μg/ml) has immunosuppressive properties that prevent T cell proliferation by preventing human primary T cells from secreting IL-2[1]. /ml; pretreated for 5 hours) stimulates T cells with immobilized OKT3 mAb, and it phosphorylates tyrosine residues on a number of particular cellular proteins in a manner that is dependent on both time and dose[1]. In the Jurkat TAg cell line, tautomycetin (1 μg/ml; 5 hours) substantially and dose-dependently suppresses the phosphorylation of Akt and BAD[1]. |
| ln Vivo | Tautomycetin (intraperitoneal injection; 0.03 mg/kg–5 mg/kg; 30 days) suppresses the immune system in vivo after the transplantation of a cardiac allograft. Furthermore, no appreciable liver or renal damage was observed in rats given TMC at doses up to 5 mg/kg for 30 days, according to a biochemical study of GOP, GTP, glucose, and creatine levels in blood[1]. |
| Cell Assay |
Western Blot Analysis[1] Cell Types: Human primary B cells Tested Concentrations: 1 μg/ml Incubation Duration: Pretreated for 5 hrs (hours), and then stimulated with immobilized OKT3 mAb (10 μg/ml) for 0, 5, 10 miuntes Experimental Results: Blocked tyrosine phosphorylation of intracellular signal mediators. Apoptosis Analysis[1] Cell Types: Jurkat T cells Tested Concentrations: 1 μg/ml Incubation Duration: 5 hour Experimental Results: Induces cell apoptosis. |
| Animal Protocol |
Animal/Disease Models: Rat[2] Doses: 0.03 mg/kg, 0.5 mg/kg, 5 mg/kg; 30 days Route of Administration: intraperitoneal (ip)injection Experimental Results: demonstrated immunosuppressive effects in vivo |
| References |
[1]. S Mitsuhashi, et al. Tautomycetin is a novel and specific inhibitor of serine/threonine protein phosphatase type 1, PP1. Biochem Biophys Res Commun. 2001 Sep 21;287(2):328-31. [2]. Jae-Hyuck Shim, et al. Immunosuppressive effects of tautomycetin in vivo and in vitro via T cell-specific apoptosis induction. Proc Natl Acad Sci U S A. 2002 Aug 6;99(16):10617-22. |
| Additional Infomation |
Tautomycetin is a sesquiterpenoid. [(16E)-17-ethyl-4,8-dihydroxy-3,7,11,13-tetramethyl-6,15-dioxononadeca-16,18-dien-2-yl] 3-hydroxy-3-(4-methyl-2,5-dioxofuran-3-yl)propanoate has been reported in Streptomyces griseochromogenes with data available. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6482 mL | 8.2408 mL | 16.4815 mL | |
| 5 mM | 0.3296 mL | 1.6482 mL | 3.2963 mL | |
| 10 mM | 0.1648 mL | 0.8241 mL | 1.6482 mL |