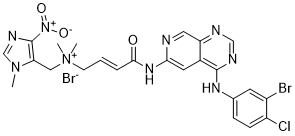
Tarloxotinib bromide (formerly TH-4000; PR-610) is a novel quarternary amine-based and irreversible EGFR/HER2 inhibitor designed as a prodrug to selectively release a covalent (irreversible (covalent)) EGFR tyrosine kinase inhibitor under severe hypoxia, a feature of many solid tumors. Tarloxotinib may be able to reduce or eliminate the systemic side effects of EGFR tyrosine kinase inhibitors that are currently on the market by effectively blocking aberrant EGFR signaling in a tumor-specific manner.
Physicochemical Properties
| Molecular Formula | C24H24BR2CLN9O3 |
| Molecular Weight | 681.766860961914 |
| Exact Mass | 679.005 |
| Elemental Analysis | C, 42.28; H, 3.55; Br, 23.44; Cl, 5.20; N, 18.49; O, 7.04 |
| CAS # | 1636180-98-7 |
| Related CAS # | 1636938-13-0 (cation);1636180-98-7 (bromide); |
| PubChem CID | 51038315 |
| Appearance | Light yellow to yellow solid powder |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 39 |
| Complexity | 863 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | BrC1=C(C=CC(=C1)NC1C2C(=CN=C(C=2)NC(/C=C/C[N+](C)(C)CC2=C([N+](=O)[O-])N=CN2C)=O)N=CN=1)Cl.[Br-] |
| InChi Key | WAKIMVYUBWMMHJ-FXRZFVDSSA-N |
| InChi Code | InChI=1S/C24H23BrClN9O3.BrH/c1-33-14-30-24(34(37)38)20(33)12-35(2,3)8-4-5-22(36)32-21-10-16-19(11-27-21)28-13-29-23(16)31-15-6-7-18(26)17(25)9-15;/h4-7,9-11,13-14H,8,12H2,1-3H3,(H-,27,28,29,31,32,36);1H/b5-4+; |
| Chemical Name | [(E)-4-[[4-(3-bromo-4-chloroanilino)pyrido[3,4-d]pyrimidin-6-yl]amino]-4-oxobut-2-enyl]-dimethyl-[(3-methyl-5-nitroimidazol-4-yl)methyl]azanium;bromide |
| Synonyms | TH-4000; TH 4000; TH4000; PR-610; PR610; PR 610; Tarloxotinib bromide; Hypoxin; Tarloxotinib |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | EGFR/HER2 | |
| ln Vitro |
|
|
| ln Vivo |
|
|
| References |
[1]. Abstract A67: Preclinical efficacy of tarloxotinib bromide (TH-4000), a hypoxia-activated EGFR/HER2 inhibitor: rationale for clinical evaluation in EGFR mutant, T790M-negative NSCLC following progression on EGFR-TKI therapy. Abstracts: AACR- [2]. Abstract 5358: The hypoxia-activated EGFR-TKI TH-4000 overcomes erlotinib-resistance in preclinical NSCLC models at plasma levels achieved in a Phase 1 clinical trial. AACR 106th Annual Meeting 2015; April 18-22, 2015; Philadelphia, PA. |
Solubility Data
| Solubility (In Vitro) | DMSO: ≥ 33 mg/mL (~48.4 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (3.67 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (3.67 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4668 mL | 7.3339 mL | 14.6677 mL | |
| 5 mM | 0.2934 mL | 1.4668 mL | 2.9335 mL | |
| 10 mM | 0.1467 mL | 0.7334 mL | 1.4668 mL |