Physicochemical Properties
| Molecular Formula | C6H12NO2P |
| Molecular Weight | 161.13878 |
| Exact Mass | 161.061 |
| CAS # | 182485-36-5 |
| PubChem CID | 5521 |
| Appearance | White to off-white solid powder |
| LogP | 1.092 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 10 |
| Complexity | 200 |
| Defined Atom Stereocenter Count | 0 |
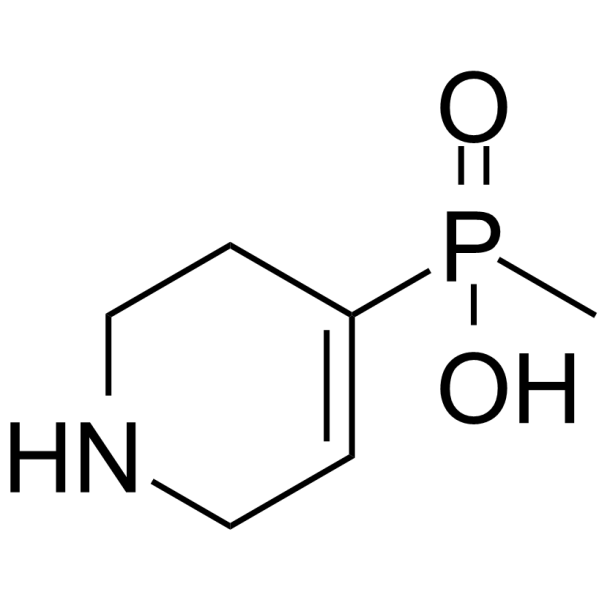
| SMILES | CP(C1=CCNCC1)(=O)O |
| InChi Key | MFUKVPOVVKKLRQ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C6H12NO2P/c1-10(8,9)6-2-4-7-5-3-6/h2,7H,3-5H2,1H3,(H,8,9) |
| Chemical Name | methyl(1,2,3,6-tetrahydropyridin-4-yl)phosphinic acid |
| Synonyms | TPMPA; 182485-36-5; methyl(1,2,3,6-tetrahydropyridin-4-yl)phosphinic acid; Tpmpa [MI]; Phosphinic acid, P-methyl-P-(1,2,3,6-tetrahydro-4-pyridinyl)-; UNII-TR7I0800L2; TR7I0800L2; CHEMBL397209; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | GABAC receptor (KB = 2.1 μM) |
| ln Vitro |
The GABA currents of ρ1 receptors (IC50 = 1.6 μM) and the chimeric ρ1/α1 receptors are antagonized by TPMPA with a potency of roughly 1.3 μM[1]. The KB values of TPMPA against rho-1 and rho-2 receptors are 2.0 and 15.6 μM, respectively, indicating modest activity[2].
Furthermore, the specific GABAC antagonist (1,2,5,6-tetrahydropyridine-4-yl)methylphosphinic acid (TPMPA) antagonized the GABA currents of ρ1 receptors (IC50 = 1.6 μM) and those of the chimeric ρ1/α1 receptors with approximately the same potency (IC50 = 1.3 μM) (Fig. 3B), whereas GABAA receptors were only slightly affected. Actually, at high concentrations of either zinc or TPMPA, the application of GABA elicited an outward current, instead of the usual inward current (Fig. 3). This is probably because at high concentrations, zinc and TPMPA block poorly characterized, inward-resting currents, and this block became evident after the action of GABA on the chimeric receptors was fully blocked [1]. |
| Cell Assay |
Electrophysiological Recordings in Xenopus Oocytes.[1] Isolation of oocytes and recordings were essentially as described previously. Briefly, ovaries from female Xenopus laevis were dissected out, follicles were isolated manually, and, to remove the enveloping follicular cells, the follicles were treated with 0.5 mg/ml collagenase type 1 for 1 h and finally maintained at 16°C in Barth's medium containing gentamycin (0.1 mg/ml). One day later, 5–10 nl of plasmid pcDNAρ1, pcDNAρ1/α1, or pcDNAρ1[α1TM2] at 0.5 mg/ml was injected into the nucleus of Xenopus oocytes. A combination of GABAA receptor subunits α1β2γ2L (2:2:1) was injected at the same concentration (cf. ref. 24). Electrophysiological records were similar to those we have described previously. Dose-response relations were fitted with the Hill equation (cf. ref. 19). To estimate the rate of desensitization, the GABA currents were fitted with one or two exponential decay functions: τs and τf are the time constants for the slow- and fast-decay components respectively. |
| References |
[1]. GABArho 1/GABAAalpha 1 receptor chimeras to study receptor desensitization. Proc Natl Acad Sci U S A. 2000;97(7):3562-3566. [2]. Neurologically-active compounds. WO1998058939A1 [3]. GABAc receptors: relatively simple transmitter -gated ion channels?. Trends Pharmacol Sci. 1996;17(9):319-323. |
| Additional Infomation |
TPMPA ((1,2,5,6-tetrahydropyridine-4-yl)methylphosphinic acid) is a selective antagonist of GABAC receptors (also known as GABA-ρ or GABAA-ρ receptors). GABAC receptors are found primarily in the retina, although they may be present in other tissues, including the hippocampus, spinal cord, superior colliculus, pituitary and the gut. TPMPA is used as a pharmacological probe for the investigation of GABAC receptor function. gamma-Aminobutyrate type C (GABA(C)) receptors are ligand-gated ion channels that are expressed preponderantly in the vertebrate retina and are characterized, among other things, by a very low rate of desensitization and resistance to the specific GABA(A) antagonist bicuculline. To examine which structural elements determine the nondesensitizing character of the human homomeric rho1 receptor, we used a combination of gene chimeras and electrophysiology of receptors expressed in Xenopus oocytes. Two chimeric genes were constructed, made up of portions of the rho1-subunit and of the alpha1-subunit of the GABA(A) receptor. When expressed in Xenopus oocytes, one chimeric gene (rho1/alpha1) formed functional homooligomeric receptors that were fully resistant to bicuculline and were blocked by the specific GABA(C) antagonist (1,2,5, 6-tetrahydropyridine-4-yl)methylphosphinic acid and by zinc. Moreover, these chimeric receptors had a fast-desensitizing component, even faster than that of heterooligomeric GABA(A) receptors, in striking contrast to the almost nil desensitization of wild-type rho1 (wt rho1) receptors. To see whether the fast-desensitizing characteristic of the chimera was determined by the amino acids forming the ion channels, we replaced the second transmembrane segment (TM2) of rho1 by that of the alpha1-subunit of GABA(A). Although the alpha1-subunit forms fast-desensitizing receptors when coexpressed with other GABA(A) subunits, the sole transfer of the alpha1TM2 segment to rho1 was not sufficient to form desensitizing receptors. All this suggests that the slow-desensitizing trait of rho1 receptors is determined by a combination of several interacting domains along the molecule. [1] The inhibitory neurotransmitter, GABA, activates a variety of receptors in all areas of the CNS. Two major subtypes of GABA receptors are well known: (1) GABAA receptors are ligand-gated Cl- channels that consist of a heteromeric mixture of protein subunits forming a pentameric structure, and (2) GABAB receptors couple to Ca2+ and K+ channels via G proteins and second messengers. Here, Graham Johnston discusses evidence for a third major subclass of GABA receptors. GABAC receptors appear to be relatively simple ligand-gated Cl- channels with a distinctive pharmacology, in that they are not blocked by bicuculline and not modulated by barbiturates, benzodiazepines or neuroactive steroids. Compared with GABAA receptors, GABAC receptors are activated at lower concentrations of GABA and are less liable to desensitization. In addition, their channels open for a longer time. The pharmacology of these novel subtypes of GABA receptors may yield important therapeutic agents.[2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 6.2058 mL | 31.0289 mL | 62.0578 mL | |
| 5 mM | 1.2412 mL | 6.2058 mL | 12.4116 mL | |
| 10 mM | 0.6206 mL | 3.1029 mL | 6.2058 mL |