Physicochemical Properties
| Molecular Formula | C17H9CL2F3N2O2 |
| Molecular Weight | 401.166772603989 |
| Exact Mass | 399.999 |
| CAS # | 136186-07-7 |
| PubChem CID | 54735328 |
| Appearance | White to off-white solid powder |
| LogP | 5.8 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 26 |
| Complexity | 597 |
| Defined Atom Stereocenter Count | 0 |
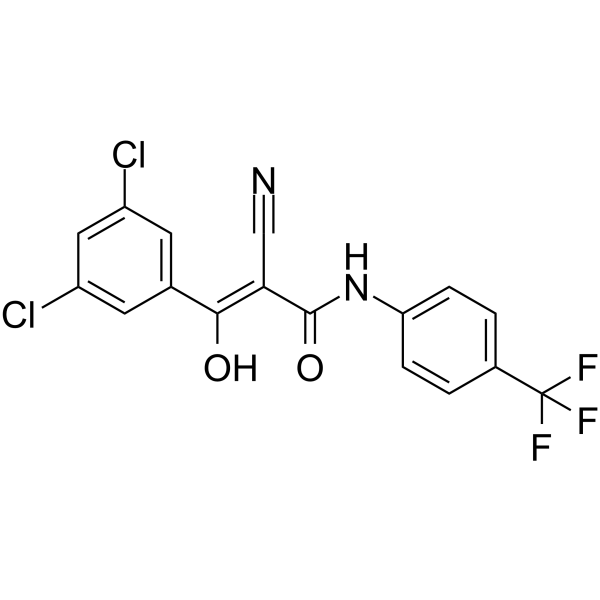
| SMILES | ClC1C=C(C=C(C=1)/C(=C(\C#N)/C(NC1C=CC(C(F)(F)F)=CC=1)=O)/O)Cl |
| InChi Key | FVJSEJNZNQVVDZ-PFONDFGASA-N |
| InChi Code | InChI=1S/C17H9Cl2F3N2O2/c18-11-5-9(6-12(19)7-11)15(25)14(8-23)16(26)24-13-3-1-10(2-4-13)17(20,21)22/h1-7,25H,(H,24,26)/b15-14- |
| Chemical Name | (Z)-2-cyano-3-(3,5-dichlorophenyl)-3-hydroxy-N-[4-(trifluoromethyl)phenyl]prop-2-enamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | EC50: 10.5 μM (Ca2+ current response from TPC2)[1] |
| ln Vitro | Ancient members of the voltage-gated ion channel superfamily are known as two-pore channels (TPC1-3). The endo-lysosomal system expresses TPCs, which control the trafficking of different payloads. Depending on how it is triggered, TPC2 can mediate various physiological and potentially pathological effects. TPC2's ion selectivity is agonist-dependent rather than fixed. One special type of ion channel that responds to various activating ligands by conducting distinct ions is TPC2. Ca2+ signals are consistently evoked by TPC2-A1-N (10 μM), and the response reaches its plateau more quickly in TPC2-A1-N than in TPC2-A1-P. TPC2-A1-N has an EC50 of 7.8 μM in full concentration-effect relationships for the plateau response in a cell line that is stable in expressing TPC2L11A/L12A. Cells expressing TPC2L11A/L12A are activated by TPC2-A1-N (10 μM) to induce Ca2+ influx through the TPC2 pore, but not TPC2L11A/L12A/L265P. Furthermore, by removing extracellular Ca2+, the known TPC2 blockers Tetrandrine (Tet), Raloxifene (Ral), and Fluphenazine (Flu) can specifically prevent the reactions to TPC2-A1-N[1]. TPC2-A1-N (30 μM) elicits currents using Na+ as the main permeation in endo-lysosomal patch-clamp tests conducted on vacuolin-enlarged endo-lysosomes derived from HEK293 cells transiently expressing human TPC2 (hTPC2), but not in cells expressing TPC1[1]. TPC2-A1-N (30 μM) causes more currents in endo-lysosomes isolated from cells expressing a gain-of-function variation of TPC2 (TPC2M484L) than in the wild-type isoform in endo-lysosomal patch-clamp experiments. TPC2-A1-N has an EC50 value of 0.6 μM[1]. |
| References |
[1]. Agonist-mediated switching of ion selectivity in TPC2 differentially promotes lysosomal function. Elife. [2]. Targeting Two-Pore Channels: Current Progress and Future Challenges. Trends Pharmacol Sci. 2020 Aug;41(8):582-594. |
Solubility Data
| Solubility (In Vitro) | DMSO: 250 mg/mL (623.18 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (5.18 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (5.18 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (5.18 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4927 mL | 12.4635 mL | 24.9271 mL | |
| 5 mM | 0.4985 mL | 2.4927 mL | 4.9854 mL | |
| 10 mM | 0.2493 mL | 1.2464 mL | 2.4927 mL |