Physicochemical Properties
| Molecular Formula | C18H14FN3O5 |
| Molecular Weight | 371.319267749786 |
| Exact Mass | 371.091 |
| CAS # | 2245693-15-4 |
| PubChem CID | 146313868 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 1.9 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 27 |
| Complexity | 659 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | ACSDSFPAGHERCC-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H14FN3O5/c1-20-12-7-9(19)6-11-14(12)22-17(25)13(15(11)23)16(24)21-10-4-2-8(3-5-10)18(26)27/h2-7,20H,1H3,(H,21,24)(H,26,27)(H2,22,23,25) |
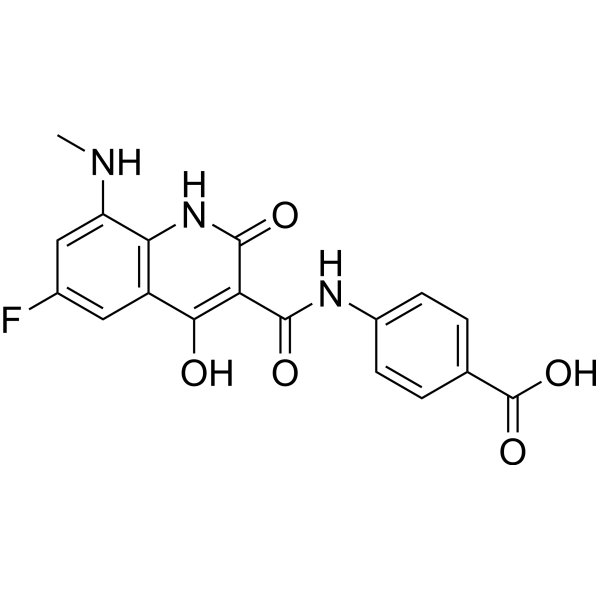
| Chemical Name | 4-[[6-fluoro-4-hydroxy-8-(methylamino)-2-oxo-1H-quinoline-3-carbonyl]amino]benzoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | TP0480066 (Compound 32) (0-2048 μg/mL; 18-24 hours), exhibits strong antibacterial activity against a range of bacteria, including some drug-resistant strains of MRSA (n = 24), gPRSP (n = 30), and VRE (n = 34) that have been clinically isolated[1]. The antibacterial activity of TP0480066 (0-2048 μg/mL; 18-24 h) against Clostridium difficile is good [1]. Strong antimicrobial activity against Neisseria gonorrhoeae, including strains less susceptible to or resistant to current antimicrobial agents, is exhibited by TP0480066 (0–2048 μg/mL; 24-48 hours) [2]. Neisseria gonorrhoeae ATCC 49226 MIC (5×10-4 µg/mL) was reached (or exceeded) at concentrations of TP0480066 (1.25×10-4, 5×10-4, and 2×10-3 µg/mL; 24 hours) [2]. Determination of cell viability [1] |
| ln Vivo | The Cmax, Tmax, t1/2, and AUC0-24 h values of TP0480066 (100 mg/kg; subcutaneous injection; once) are 12400 ng/mL, 0.250 h, 6.79 h, and 16000 h·ng/mL, respectively [2]. Neisseria gonorrhoeae ATCC 49226 and NCTC 13479 are inhibited by TP0480066 (1, 3, 10, 30, 100 mg/kg; subcutaneous injection; single dose) in a dose-dependent manner over a 24-hour period [2]. |
| Cell Assay |
Cell viability determination [1] Cell Types: clinically isolated methicillin-resistant Staphylococcus aureus (24 strains), vancomycin-resistant enterococci (Enterococcus faecium (29 strains) and Enterococcus faecalis (5 strains), vanA positive (18 strains), vanB positive (14 strains) and vanA/vanB negative (2 strains)), genotype penicillin-resistant Streptococcus pneumoniae (30 strains) Tested Concentrations: 0-2048 μg/mL Incubation Duration: 18-24 hrs (hours) Experimental Results: The drug-resistant strains demonstrated good antibacterial activity with MIC ranges of 0.03-0.5, 0.015-0.25 and 0.002-0.015 μg/mL for MRSA (n=24), gPRSP (n=30) and VRE (n=34), respectively. Cell viability assay [2] Cell Types: Neisseria gonorrhoeae ATCC (49226, 43069, BAA-1846, 700717, 700825), Neisseria gonorrhoeae NCTC (13477, 13478, 13479, 13480, 13481, 13482, 13483, 13818, 13821) Tested Concentrations: 0-2048 μg/mL Incubation Duration: 24-48 h Experimental Results: It demonstrated significant antibacterial activity against Neisseria gonorrhoeae and drug-resistant Neisseria gon |
| Animal Protocol |
Animal/Disease Models: Female Slc:ICR mice [1] Doses: 100 mg/kg Route of Administration: subcutaneous injection, once. Experimental Results: 1.19 pharmacokinetic/PK/PK parameters of TP0480066 in female Slc:ICR mice (n=3)[1]. Tmax (h) Cmax (ng/mL) AUC0-24 (ng/mL·h) t1/2 (h) SC (100 mg/kg) 0.250 12400 16000 6.79 Animal/Disease Models: Female balb/c (Bagg ALBino) mouse (6 weeks- Elderly; reproductive tract infection model) [2] Doses: 1, 3, 10, 30 and 100 mg/kg Route of Administration: subcutaneous injection; single. Experimental Results: The mean viable cell count of Neisseria gonorrhoeae ATCC 49226 and NCTC 13479 was Dramatically diminished at doses of 30 and 100 mg/kg. |
| References |
[1]. Lead optimization of 8-(methylamino)-2-oxo-1,2-dihydroquinolines as bacterial type II topoisomerase inhibitors. Bioorg Med Chem. 2020 Nov 15;28(22):115776. [2]. In Vitro and In Vivo Activities of TP0480066, a Novel Topoisomerase Inhibitor, against Neisseria gonorrhoeae. Antimicrob Agents Chemother. 2021 Mar 18;65(4):e02145-20. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6931 mL | 13.4655 mL | 26.9309 mL | |
| 5 mM | 0.5386 mL | 2.6931 mL | 5.3862 mL | |
| 10 mM | 0.2693 mL | 1.3465 mL | 2.6931 mL |