Physicochemical Properties
| Molecular Formula | C17H10F4N6 |
| Molecular Weight | 374.30 |
| Exact Mass | 370.308 |
| CAS # | 2072051-04-6 |
| PubChem CID | 7641 |
| Appearance |
COLORLESS OR VERY PALE AMBER LIQ Light colored, oily liquid. COLORLESS OR VERY PALE AMBER LIQUID Clear liquid |
| Melting Point |
-90 °F (NTP, 1992) -67.8 °C 7.49 °C (280.65 K) -67.8 °C |
| LogP | 6.8 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 19 |
| Heavy Atom Count | 26 |
| Complexity | 320 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | SAOKZLXYCUGLFA-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C22H42O4/c1-5-9-13-19(7-3)17-25-21(23)15-11-12-16-22(24)26-18-20(8-4)14-10-6-2/h19-20H,5-18H2,1-4H3 |
| Chemical Name | bis(2-ethylhexyl) hexanedioate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
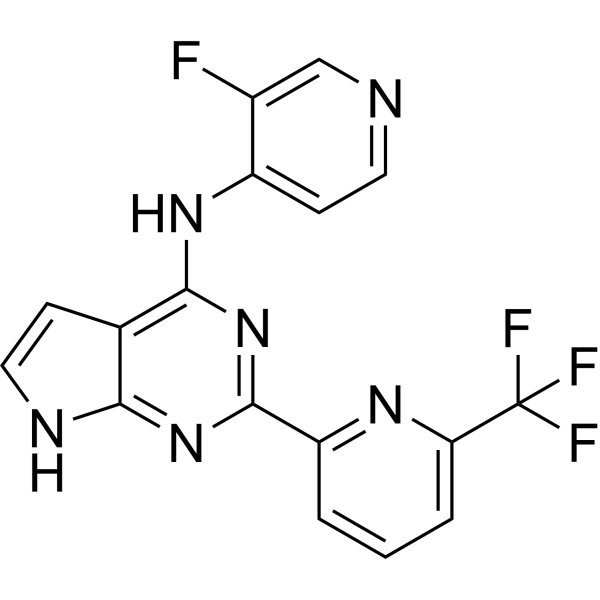
| ln Vitro | TGFβRI-IN-6 (compound 22) has a Ki value of 0.22 nM and inhibits TGFβRI[1]. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion The absorption, distribution, and elimination of DEHA were studied in mice and rats. Male Sprague Dawley rats, male NMRI mice, and pregnant female NMRI mice on day 17 of gestation were administered (14)C labeled DEHA dissolved in dimethyl sulfoxide or corn oil iv or intragastrically. The DEHA was labeled on the carbonyl or alcohol moiety. Animals were killed 5 min to 4 days after dosing, and the tissue distribution of (14)C activity was determined by whole body autoradiography. The tissue distribution of (14)C activity from carbonyl labeled DEHA was similar in all animals. Highest levels of radioactivity were observed in the body fat, liver, and kidney after intragastrically or iv administration. (14)C activity from alcohol labeled DEHA was found in the bronchi of male mice. In pregnant mice, (14)C activity was observed in the fetal liver, intestine, and bone marrow during the first 24 hr after carbonyl labeled DEHA was given. Very little radiolabel was found in fetuses of mice given alcohol labeled DEHA. No DEHA derived radioactivity was found in mice 4 days after dosing. Blood DEHA concn in rats increased faster and were two or three times higher when the dose was given in DMSO rather than corn oil. Significant amounts of DEHA were excreted in the bile of rats treated with DEHA in DMSO. Very little biliary elimination of radiolabel occurred in animals given carbonyl labeled DEHA. DEHA was excreted in the urine, the amounts being smaller in animals used in the bile collection experiments. The vehicle had very little effect on the amount excreted. DEHA is poorly absorbed from an oil solution. Metabolism / Metabolites In vivo and in vitro metabolism of the plasticizer DEHA was examined in the rat to determine the different steps involved in the hepatic concn of peroxisomal proliferators. In the in vivo studies, different doses of DEHA and mono-(2-ethylhexyl)-adipate were administered by gavage to Wistar rats for 5 days. In the in vitro studies, hepatocytes were isolated by in situ perfusion. No DEHA was recovered in rat urine 24 hr after administration; adipic acid was the main metabolite. Only the 2-ethylhexanol pathway showed further metabolites, mainly 2-ethylhexanoic acid which was either conjugated or submitted to other pathways. While 2-ethylhexanoic acid glucuronidation appeared to be dose and time dependent, 2-ethylhexanol glucuronidation was more stable. In vitro, the first hydrolysis of DEHA appeared to be a rate limiting step. When mono-(2-ethylhexyl) adipate was added directly to the culture medium, all the metabolites identified in the in vivo study were found. Glucuronidation of both 2-ethylhexanol and 2-ethylhexanoic acid was dose and time dependent. |
| Toxicity/Toxicokinetics |
Non-Human Toxicity Values LD50 Rat iv 0.90 ml/kg LD50 Rat oral 5.6 g/kg LD50 Rat ip 47 ml/kg LD50 Rat (male, F344) oral, gavage 45 g/kg For more Non-Human Toxicity Values (Complete) data for BIS(2-ETHYLHEXYL) ADIPATE (10 total), please visit the HSDB record page. LD50 Rat oral 9110 mg/kg LD50 Rat iv 900 mg/kg LD50 Mouse oral 15 g/kg LD50 Rabbit iv 540 mg/kg |
| References |
[1]. Heterobicyclic inhibitors of transforming growth factor beta receptor I (TGFβRI). Bioorg Med Chem. 2018 Mar 1;26(5):1026-1034. |
| Additional Infomation |
Bis(2-ethylhexyl) adipate is a colorless to straw-colored liquid with a mild odor. Floats on water. (USCG, 1999) Bis(2-ethylhexyl) adipate is a diester resulting from the formal condensation of the carboxy groups of adipic acid with 2-ethylhexan-1-ol. It is used as a plasticiser in the preparation of various polymers. It has a role as a plasticiser. It is a diester, a carboxylic ester and a member of dicarboxylic acids and O-substituted derivatives. It is functionally related to a 2-ethylhexan-1-ol and an adipic acid. Bis(2-ethylhexyl) adipate has been reported in Panax ginseng with data available. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6717 mL | 13.3583 mL | 26.7165 mL | |
| 5 mM | 0.5343 mL | 2.6717 mL | 5.3433 mL | |
| 10 mM | 0.2672 mL | 1.3358 mL | 2.6717 mL |