Physicochemical Properties
| Molecular Formula | C17H25CLN2O2S |
| Molecular Weight | 356.9106 |
| Exact Mass | 356.132 |
| Elemental Analysis | C, 57.21; H, 7.06; Cl, 9.93; N, 7.85; O, 8.97; S, 8.98 |
| CAS # | 1818252-53-7 |
| Related CAS # | 1818252-52-6; 1818252-53-7 (HCl); |
| PubChem CID | 118432651 |
| Appearance | White to yellow solid-liquid Mixture |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 23 |
| Complexity | 410 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | Cl.S1C=C(C(NC2CCOCC2)=O)C=C1[C@@H]1C[C@H]1NCC1CC1 |
| InChi Key | LEEWMGOHGNRDKC-NUNOUFIPSA-N |
| InChi Code | InChI=1S/C17H24N2O2S.ClH/c20-17(19-13-3-5-21-6-4-13)12-7-16(22-10-12)14-8-15(14)18-9-11-1-2-11/h7,10-11,13-15,18H,1-6,8-9H2,(H,19,20)1H/t14?,15-/m1./s1 |
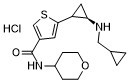
| Chemical Name | 5-((2R)-2-((cyclopropylmethyl)amino)cyclopropyl)-N-(tetrahydro-2H-pyran-4-yl)thiophene-3-carboxamide hydrochloride |
| Synonyms | TAK418; TAK-418 HCl; TAK-418; 1818252-53-7; LVM0PK6IHG; 5-((1R,2R)-2-(cyclopropylmethylamino)cyclopropyl)-N-(oxan-4-yl)thiophene-3-carboxamide; RefChem:187188; TAK 418 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
TAK-418 is a potent and selective inhibitor of lysine-specific demethylase 1 (LSD1/KDM1A) enzyme activity. It inhibits LSD1 with an IC₅₀ of 2.9 nM and a \(k_{\text{inact}}/K_{\text{I}}\) value of \(3.8 \times 10^5 \pm 3.8 \times 10^4\) M⁻¹ s⁻¹ [1]. |
| ln Vitro |
In primary cultured rat neurons, treatment with TAK-418 (0.0001–10 µM) for 3 days concentration-dependently increases H3K4me2 and H3K9me2 levels at the Ucp2 gene and induces Ucp2 mRNA expression, with significant effects observed at 0.01 µM [1]. TAK-418 has minimal impact on the interaction between LSD1 and its cofactor GFI1B in TF-1a cells, unlike the conventional LSD1 inhibitor T-711. It forms a compact formylated adduct with the FAD cofactor in LSD1, avoiding steric interference with GFI1B binding [1]. |
| ln Vivo |
TAK-418 (1 mg/kg; oral; once daily for 14 days) improves certain autism spectrum disorder (ASD)-like symptoms in animals caught in models of chronic neurodevelopmental abnormalities [1]. TAK-418 elevates H3K4me1/2/3 and H3K9me2 levels of the Ucp2 gene and stimulates Ucp2 mRNA expression in primary cultured neurons. TAK-418 also elevates H3K4me1/2/3 of the Bdnf gene. TAK-418 avoids steric hindrance of GFI1B in the binding pocket by creating a tightly formylated adduct form of coenzyme flavin adenine dinucleate (FAD). TAK-418 demonstrates excellent pharmacokinetic characteristics in omnidids and inhibits the LSD1 enzyme in the brain. A single injection of 1 or 3 mg of TAK-418/kg enhanced H3K4me2 levels of the Ucp2 gene in the mouse brain [ 1]. TAK-418 can be active without generating hematological toxicity in hungry animals [1]. Improves neurological difficulties in terms of cells, molecules, and gene expression, and functional and functional levels of KS mouse model (Kmt2d+/βGeo mice) [2].
In valproate (VPA)-exposed rat models of autism spectrum disorder (ASD), oral administration of TAK-418 (1 mg/kg/day for 2 weeks) significantly improves sociability deficits in both juvenile (5-week-old) and adult (15-week-old) rats, while lower doses (0.1, 0.3 mg/kg) are ineffective. A 1-week treatment also rescues sociability, but a single dose is ineffective. TAK-418 (1 mg/kg/day for 16–20 days) also ameliorates cognitive impairment in VPA rats in the novel object recognition test [1]. In poly I:C-induced maternal immune activation mouse models of ASD, TAK-418 (0.1, 0.3, 1 mg/kg/day for 2 weeks) dose-dependently rescues sociability deficits [1]. TAK-418 (1 mg/kg) inhibits LSD1 enzyme activity in the rat cortex by 95.7% at 2 hours post-dose and in mouse brain by 91.5%. It almost completely normalizes genome-wide dysregulated gene expression in the cortex of VPA rats and poly I:C mice, as shown by RNA-seq, and restores aberrant DNA methylation patterns [1]. In Kmt2d\(^{+/βGeo}\) mice (a model of Kabuki syndrome), oral TAK-418 (1 mg/kg/day for 2 weeks) rescues adult hippocampal neurogenesis deficits (increased DCX⁺ cells) in a dose-dependent manner (full effect at 1 mg/kg, partial at 0.5 and 2 mg/kg). It restores H3K4me1/2 levels and partially restores H3K4me3 in the hippocampus, and reverses abnormal gene expression (e.g., Fos, Fosb). TAK-418 also improves spatial learning and memory deficits in the Morris water maze and corrects splenomegaly after 8 weeks of treatment [2]. |
| Enzyme Assay |
LSD1 enzyme kinetics are measured using a peroxidase-coupled assay. Recombinant human LSD1/CoREST complex is incubated with various concentrations of TAK-418 in assay buffer containing monomethylated H3K4 peptide. Horseradish peroxidase and Amplex Red are added, and the hydrogen peroxide produced during the amine oxidase reaction is monitored fluorometrically (excitation 570 nm, emission 585 nm). Progress curves are fitted to obtain \(k_{\text{obs}}\), and \(k_{\text{inact}}/K_{\text{I}}\) values are calculated from replots of \(k_{\text{obs}}\) versus inhibitor concentration [1]. |
| Cell Assay |
Primary cultured rat neurons are treated with TAK-418 (0.0001–10 µM) or DMSO for 3 days. Chromatin immunoprecipitation (ChIP) is performed using antibodies against H3K4me2 and H3K9me2, followed by qPCR at the Ucp2 gene. Total RNA is extracted and Ucp2 mRNA levels are quantified by qRT-PCR [1]. For co-immunoprecipitation, TF-1a cells are treated with TAK-418 or T-711 for 6 h, then lysed. LSD1 is immunoprecipitated from chromatin fractions, and bound GFI1B is detected by Western blotting [1]. |
| Animal Protocol |
VPA rats are generated by injecting pregnant SD rats with sodium valproate (500 mg/kg i.p.) on gestational day 12.5. Poly I:C mice are generated by injecting pregnant C57BL/6J mice with poly I:C (5 mg/kg i.v.) on gestational day 15. Male offspring are used. TAK-418 is dissolved in 0.5% methylcellulose/0.5% citrate and administered orally once daily for 2 weeks (or 1 week, or single dose) at doses of 0.1, 0.3, or 1 mg/kg. Behavioral tests (three-chamber sociability, novel object recognition, Morris water maze) are performed after treatment. Tissues (cortex, hippocampus) are collected for RNA-seq, ChIP-seq, RRBS, Western blot, and immunohistochemistry [1, 2]. For Kabuki syndrome model, Kmt2d\(^{+/βGeo}\) mice on C57BL/6J background are orally gavaged daily with TAK-418 (1 mg/kg) or vehicle for 2 weeks (neurogenesis and molecular studies) or 8 weeks (splenomegaly study). Brains are processed for DCX immunofluorescence, histone extraction, ChIP-seq, RNA-seq; spleens are weighed [2]. |
| ADME/Pharmacokinetics |
TAK-418 exhibits a favorable pharmacokinetic profile in rodents. After oral administration, it is rapidly absorbed and shows good bioavailability. In mice, a single dose of 1 mg/kg increases H3K4me2 at the Ucp2 gene in the brain, indicating brain penetration. Specific parameters such as half-life, Cmax, and oral bioavailability are provided in supplementary figures [1]. |
| Toxicity/Toxicokinetics |
TAK-418 is well tolerated in rodents. No significant body weight loss or adverse clinical signs are observed in VPA rats, poly I:C mice, or Kmt2d\(^{+/βGeo}\) mice at therapeutic doses (up to 1 mg/kg/day for 8 weeks). Unlike the conventional LSD1 inhibitor T-711, TAK-418 does not cause thrombocytopenia or hematological toxicity [1, 2]. In the Kabuki syndrome mouse model, 8-week treatment with TAK-418 ameliorates splenomegaly without any obvious side effects [2]. |
| References |
[1]. LSD1 enzyme inhibitor TAK-418 unlocks aberrant epigenetic machinery and improves autism symptoms in neurodevelopmental disorder models. Sci Adv. 2021;7(11):eaba1187. [2]. Inhibition of KDM1A activity restores adult neurogenesis and improves hippocampal memory in a mouse model of Kabuki syndrome. Mol Ther Methods Clin Dev. 2021;20:779-791. |
| Additional Infomation |
TAK-418 is a novel, orally bioavailable LSD1 enzyme activity-specific inhibitor developed by Takeda Pharmaceutical Company. It is designed to avoid disruption of LSD1-cofactor interactions, thereby minimizing hematological toxicity. Preclinical studies demonstrate that TAK-418 normalizes aberrant epigenetic modifications and gene expression in multiple neurodevelopmental disorder models (autism spectrum disorder, Kabuki syndrome), leading to behavioral improvements. TAK-418 has completed Phase 1 clinical trials in healthy volunteers (NCT03228433, NCT03501069) and has received orphan drug designation from the EMA and FDA for the treatment of Kabuki syndrome [1, 2]. TAK-418 is a selective, orally active LSD1 (KDM1A) enzyme inhibitor. TAK-418 unlocks aberrant epigenetic machinery and improves autism symptoms in neurodevelopmental disorder models. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~55 mg/mL (~154.10 mM) H2O : ≥ 16.67 mg/mL (~46.71 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 5.5 mg/mL (15.41 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 55.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 5.5 mg/mL (15.41 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 55.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8018 mL | 14.0091 mL | 28.0183 mL | |
| 5 mM | 0.5604 mL | 2.8018 mL | 5.6037 mL | |
| 10 mM | 0.2802 mL | 1.4009 mL | 2.8018 mL |