Physicochemical Properties
| Molecular Formula | C15H22O6 |
| Molecular Weight | 298.33158 |
| Exact Mass | 298.141 |
| CAS # | 34114-99-3 |
| PubChem CID | 9904331 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 515.2±50.0 °C at 760 mmHg |
| Melting Point | 125°C- 135 °C |
| Flash Point | 265.4±30.1 °C |
| Vapour Pressure | 0.0±3.0 mmHg at 25°C |
| Index of Refraction | 1.644 |
| LogP | -0.77 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 21 |
| Complexity | 520 |
| Defined Atom Stereocenter Count | 8 |
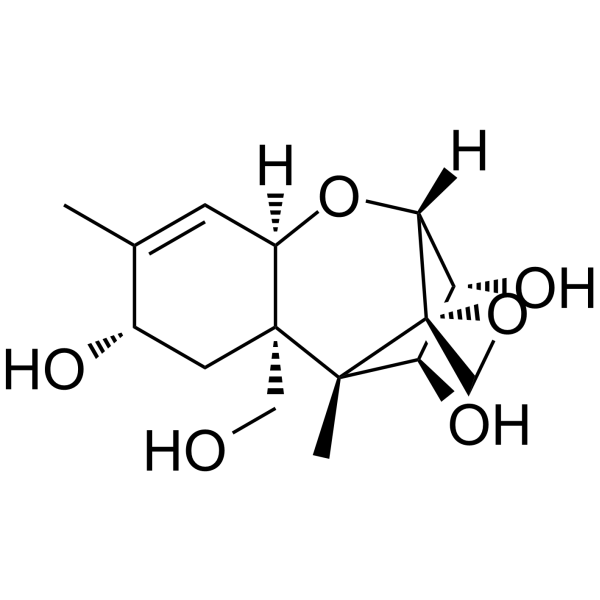
| SMILES | CC1=C[C@@H]2[C@@](C[C@@H]1O)(CO)[C@@]3(C)[C@@H]([C@H]([C@H](C43CO4)O2)O)O |
| InChi Key | ZAXZBJSXSOISTF-LYFQSNBGSA-N |
| InChi Code | InChI=1S/C15H22O6/c1-7-3-9-14(5-16,4-8(7)17)13(2)11(19)10(18)12(21-9)15(13)6-20-15/h3,8-12,16-19H,4-6H2,1-2H3/t8-,9+,10+,11+,12+,13+,14+,15-/m0/s1 |
| Chemical Name | (1S,2R,4S,7R,9R,10R,11S,12S)-2-(hydroxymethyl)-1,5-dimethylspiro[8-oxatricyclo[7.2.1.02,7]dodec-5-ene-12,2'-oxirane]-4,10,11-triol |
| Synonyms | T-2 Tetraol; 34114-99-3; T-2 toxin tetraol; T-?2 Tetraol; Z00UFS2AP1; Toxin T 2 tetraol; (1S,2R,4S,7R,9R,10R,11S,12S)-2-(hydroxymethyl)-1,5-dimethylspiro[8-oxatricyclo[7.2.1.02,7]dodec-5-ene-12,2'-oxirane]-4,10,11-triol; (2R,2'S,3R,4S,5S,5AR,7S,9aR)-5a-(hydroxymethyl)-5,8-dimethyl-2,3,4,5,5a,6,7,9a-octahydrospiro[2,5-methanobenzo[b]oxepine-10,2'-oxirane]-3,4,7-triol; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Metabolite of T-2 toxin |
| ln Vitro | Researchers tested a novel colorimetric toxicity test, based on inhibition of beta-galactosidase activity in the yeast Kluyveromyces marxianus, for sensitivity to a range of mycotoxins. A variety of trichothecene mycotoxins could be detected. The order of toxicity established with this bioassay was verrucarin A > roridin A > T-2 toxin > diacetoxyscirpenol > HT-2 toxin > acetyl T-2 toxin > neosolaniol > fusarenon X > T-2 triol > scirpentriol > nivalenol > deoxynivalenol > T-2 tetraol. The sensitivity of detection was high, with the most potent trichothecene tested, verrucarin A, having a 50% effective concentration (concentration of toxin causing 50% inhibition) of 2 ng/ml. Other mycotoxins (cyclopiazonic acid, fumonisin B1, ochratoxin A, patulin, sterigmatocystin, tenuazonic acid, and zearalenone) could not be detected at up to 10 micrograms/ml, nor could aflatoxins B1 and M1 be detected at concentrations up to 25 micrograms/ml. This test should be useful for trichothecene detection and for studies of relevant interactions-both between trichothecenes themselves and between trichothecenes and other food constituents [1]. |
| ln Vivo | Recently researchers found that a single administration of T-2 toxin (T-2), a trichothecene mycotoxin, into mice induced DNA fragmentation, a biochemical hallmark of apoptosis, in the thymus. In this study, we investigated the effective chemical structure(s) of T-2-derived metabolites capable of inducing thymic apoptosis in vivo in mice. Metabolic conversion of T-2 to 3'-hydroxy-T-2 toxin (3'-OH-T-2) did not diminish the apoptosis-inducing activity, since essentially the same level of fragmented DNA was detected in the thymus taken from mice injected with either T-2 or 3'-OH-T-2. In contrast, hydrolysis of T-2 and 3'-OH-T-2 at the carbon-4 (C-4) position to HT-2 toxin (HT-2) and 3'-hydroxy-HT-2 toxin (3'-OH-HT-2), respectively, greatly decreased the level of DNA fragmentation. Similarly, hydrolysis of T-2 at the carbon-8 (C-8) position to neosolaniol strongly diminished its ability to induce DNA fragmentation. T-2 tetraol, having no ester groups, was unable to induce apoptosis. Based on the data presented in this study, researchers concluded that both the acetyl group at the C-4 position and the isovaleryl or 3'-hydroxyisovaleryl group at the C-8 position of the T-2 molecule are important for inducing cell death through apoptosis in the thymus [2]. |
| Toxicity/Toxicokinetics |
9904331 rat LD50 oral 36900 ug/kg Developments in Food Science., 4(135), 1983 9904331 rat LD50 subcutaneous 1300 ug/kg Toxicon., 26(923), 1988 [PMID:3201481] 9904331 rat LD50 intracrebral 184 ug/kg BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD; BEHAVIORAL: FLUID INTAKE; BEHAVIORAL: ATAXIA Toxicon., 26(923), 1988 [PMID:3201481] 9904331 guinea pig LD50 oral 4 mg/kg Developments in Food Science., 4(135), 1983 9904331 chicken LD50 oral 10 mg/kg Developments in Food Science., 4(135), 1983 |
| References |
[1]. A colorimetric technique for detecting trichothecenes and assessing relative potencies. Appl Environ Microbiol. 1999 May;65(5):1854-7. [2]. Structure-function relationship of T-2 toxin and its metabolites in inducing thymic apoptosis in vivo in mice. Biosci Biotechnol Biochem. 1998 Aug;62(8):1492-7. |
| Additional Infomation | T-2 Tetraol has been reported in Fusarium heterosporum, Fusarium acuminatum, and other organisms with data available. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3520 mL | 16.7600 mL | 33.5199 mL | |
| 5 mM | 0.6704 mL | 3.3520 mL | 6.7040 mL | |
| 10 mM | 0.3352 mL | 1.6760 mL | 3.3520 mL |