Physicochemical Properties
| Molecular Formula | C17H27CLN2O4S |
| Molecular Weight | 390.92 |
| Exact Mass | 390.138 |
| CAS # | 23694-17-9 |
| Related CAS # | Sultopride;53583-79-2 |
| PubChem CID | 3082221 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.16g/cm3 |
| Boiling Point | 530ºC |
| Melting Point | 181-182ºC |
| Vapour Pressure | 2.57E-11mmHg at 25°C |
| LogP | 3.914 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 25 |
| Complexity | 517 |
| Defined Atom Stereocenter Count | 0 |
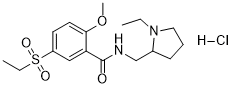
| SMILES | O=C(NCC1N(CC)CCC1)C2=CC(S(=O)(CC)=O)=CC=C2OC.Cl |
| InChi Key | IGOWMQPOGQYFFM-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H26N2O4S.ClH/c1-4-19-10-6-7-13(19)12-18-17(20)15-11-14(24(21,22)5-2)8-9-16(15)23-3/h8-9,11,13H,4-7,10,12H2,1-3H3,(H,18,20)1H |
| Chemical Name | N-((1-Ethylpyrrolidin-2-yl)methyl)-5-(ethylsulfonyl)-2-methoxybenzamide hydrochloride |
| Synonyms | Sultopride Hydrochloride Sultopride HCl |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vivo | Sultopride Hydrochloride (LIN-1418 HydroHClide) is a selective antagonist of dopamine D2 receptors. Rats treated with both sultopride hydrochloride and sulpiride showed higher levels of DOPAC and HVA in the striatum, nucleus accumbens, and medial prefrontal cortex when compared to controls. In the striatum, sulpiride hydrochloride-treated rats had higher levels of DOPAC and HVA than sulpiride-treated rats (p<0.05). In the nucleus accumbens, sulpiride hydrochloride-treated rats had higher DOPAC and HVA levels than sulpiride-treated rats (p<0.05). In rats treated with sultopride hydrochloride and sulpiride, DOPAC and HVA levels were higher in the striatum or nucleus accumbens than in the medial prefrontal cortex (p<0.05) [1]. |
| References | [1]. Moriuchi K, et al. Differences in effects of sultopride and sulpiride on dopamine turnover in rat brain. Neurochem Res. 1995 Jan;20(1):95-9 |
| Additional Infomation | A benzamide derivative that is used as an antipsychotic agent for the treatment of schizophrenia. It is also used as an antidepressive agent. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~50 mg/mL (~127.90 mM) H2O : ~50 mg/mL (~127.90 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 140 mg/mL (358.12 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5581 mL | 12.7903 mL | 25.5807 mL | |
| 5 mM | 0.5116 mL | 2.5581 mL | 5.1161 mL | |
| 10 mM | 0.2558 mL | 1.2790 mL | 2.5581 mL |