Physicochemical Properties
| Molecular Formula | C48H59NO16 |
| Exact Mass | 905.383 |
| CAS # | 1234218-62-2 |
| PubChem CID | 161734498 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 1.8 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 16 |
| Rotatable Bond Count | 18 |
| Heavy Atom Count | 65 |
| Complexity | 1910 |
| Defined Atom Stereocenter Count | 11 |
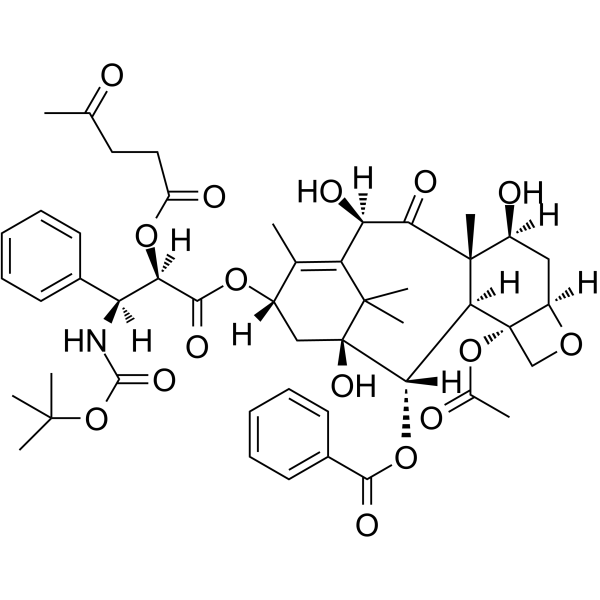
| SMILES | CC1=C2[C@H](C(=O)[C@@]3([C@H](C[C@@H]4[C@]([C@H]3[C@@H]([C@@](C2(C)C)(C[C@@H]1OC(=O)[C@@H]([C@H](C5=CC=CC=C5)NC(=O)OC(C)(C)C)OC(=O)CCC(=O)C)O)OC(=O)C6=CC=CC=C6)(CO4)OC(=O)C)O)C)O |
| InChi Key | XKHDVNYIFGGXRE-LEPVCIIKSA-N |
| InChi Code | InChI=1S/C48H59NO16/c1-25(50)20-21-33(53)62-37(35(28-16-12-10-13-17-28)49-43(58)65-44(4,5)6)42(57)61-30-23-48(59)40(63-41(56)29-18-14-11-15-19-29)38-46(9,39(55)36(54)34(26(30)2)45(48,7)8)31(52)22-32-47(38,24-60-32)64-27(3)51/h10-19,30-32,35-38,40,52,54,59H,20-24H2,1-9H3,(H,49,58)/t30-,31-,32+,35-,36+,37+,38-,40-,46+,47-,48+/m0/s1 |
| Chemical Name | [(1S,2S,3R,4S,7R,9S,10S,12R,15S)-4-acetyloxy-1,9,12-trihydroxy-10,14,17,17-tetramethyl-15-[(2R,3S)-3-[(2-methylpropan-2-yl)oxycarbonylamino]-2-(4-oxopentanoyloxy)-3-phenylpropanoyl]oxy-11-oxo-6-oxatetracyclo[11.3.1.03,10.04,7]heptadec-13-en-2-yl] benzoate |
| Synonyms | Sudocetaxel; 1234218-62-2; [(1S,2S,3R,4S,7R,9S,10S,12R,15S)-4-Acetyloxy-1,9,12-trihydroxy-10,14,17,17-tetramethyl-15-[(2R,3S)-3-[(2-methylpropan-2-yl)oxycarbonylamino]-2-(4-oxopentanoyloxy)-3-phenylpropanoyl]oxy-11-oxo-6-oxatetracyclo[11.3.1.03,10.04,7]heptadec-13-en-2-yl] benzoate; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Tubublin; microtubule depolymerization |
| References |
[1]. Polymeric complex micelles based on the double-hydrazone linkage and dual drug-loading strategy for pH-sensitive docetaxel delivery. J Mater Chem B. 2016 Feb 14;4(6):1122-1133. |
| Additional Infomation | Stimuli-triggered drug release and improved drug loading are two key prerequisites in the preparation of nano-drug delivery systems. Herein, we constructed the first pluronic P123-double (d)-hydrazone bond (hyd)-docetaxel (DTX)/DTX complex micelles (P123-d-hyd-DTX/DTX) which integrated the highly pH-sensitive strategy and the dual drug-loading pattern in one platform. We synthesized pluronic P123-double (d)-hydrazone bond (hyd)-docetaxel (DTX) conjugates (P123-d-hyd-DTX) by conjugating DTX to the PEO chains of P123 via two hydrazone linkages in the backbone for the first time. An increase in the number of hydrazone linkages within the unimolecular conjugate was conducive to improve the pH sensitivity of conjugated-based micelles. Besides, aiming at improving drug loading, the dual drug-loading strategy (chemical conjugation and physical encapsulation) was adopted. Namely, pH-sensitive P123-d-hyd-DTX was used as a vehicle to further encapsulate DTX for preparing P123-d-hyd-DTX/DTX complex micelles. The resulting P123-d-hyd-DTX/DTX micelles exhibited intact spherical shape, uniform particle size distribution (110.37 nm) and higher drug loading (12.64%). The release study in vitro confirmed that P123-d-hyd-DTX/DTX micelles and P123-d-hyd-DTX micelles (as a control) showed highly pH-responsive release properties. In particular at pH 6.5, almost 90.9% and 75% of DTX was released within 48 h, respectively. Compared to P123-d-hyd-DTX micelles, P123-d-hyd-DTX/DTX complex micelles exhibited higher proliferation inhibition effects on B16F10 cells (p < 0.01), while the cytotoxicity of P123-d-hyd-DTX/DTX complex micelles was slightly inferior to that of free DTX (p < 0.05). In the xenograft B16F10 melanoma model, P123-d-hyd-DTX/DTX complex micelles suppressed tumor growth more effectively than Duopafei® (p < 0.01) and P123-d-hyd-DTX micelles (p < 0.05), without causing obvious adverse effects. Overall, the novel polymeric complex micelle based on double-hydrazone bond and dual drug-loading strategies was a promising delivery platform to improve therapeutic efficiency and decrease side effects in cancer treatments.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |