Physicochemical Properties
| Molecular Formula | C12H8O4 |
| Molecular Weight | 216.1895 |
| Exact Mass | 216.042 |
| CAS # | 483-66-9 |
| PubChem CID | 108104 |
| Appearance | Off-white to light yellow solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 413.0±45.0 °C at 760 mmHg |
| Flash Point | 203.6±28.7 °C |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.635 |
| LogP | 1.83 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 16 |
| Complexity | 325 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | DLCJNIBLOSKIQW-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C12H8O4/c1-14-9-6-7-2-3-10(13)16-11(7)8-4-5-15-12(8)9/h2-6H,1H3 |
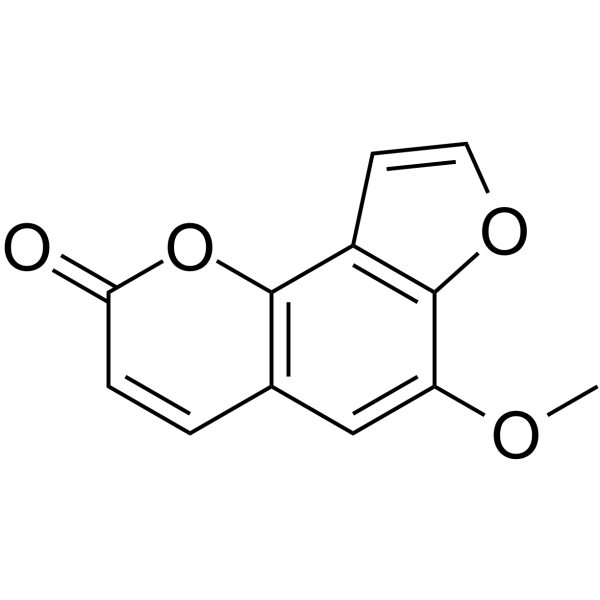
| Chemical Name | 6-methoxyfuro[2,3-h]chromen-2-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
The target of Sphondin is cyclooxygenase-2 (COX-2). The literature focuses on the regulatory effect of Sphondin on IL-1beta-induced COX-2 expression, but no specific IC50, Ki, or EC50 values for Sphondin targeting COX-2 are mentioned in the publicly available abstract and title information [1] |
| ln Vitro |
Sphondin (10–50 μM) pretreatment of A549 cells decreased COX-2 protein expression and PGE2 release caused by IL-1β in a concentration-dependent manner [1]. 1. The in vitro experiment was conducted using human pulmonary epithelial cells. After the cells were treated with IL-1beta (a pro-inflammatory cytokine) to induce COX-2 expression, Sphondin (isolated from Heracleum Laciniatum) was added for intervention. The results showed that Sphondin could inhibit IL-1beta-induced COX-2 expression in human pulmonary epithelial cells. However, the specific concentration of Sphondin used and the exact inhibition rate (e.g., the degree of reduction in COX-2 mRNA or protein levels) were not clearly stated in the publicly available information [1] 2. The experiment further detected the expression level of COX-2 at the molecular level (likely including mRNA and protein levels), and the regulatory effect of Sphondin on COX-2 was confirmed through corresponding molecular biology experiments (such as PCR and Western blot) [1] |
| Enzyme Assay |
1. For the detection of COX-2 mRNA expression: Human pulmonary epithelial cells were first cultured under appropriate conditions. After reaching a certain confluency, the cells were divided into different groups, including a control group, an IL-1beta-induced model group, and Sphondin intervention groups (with different concentrations possibly set, but specific concentrations not specified). After treatment with IL-1beta and Sphondin for a certain period (treatment time not specified in public information), total RNA was extracted from the cells. Then, reverse transcription was performed to synthesize cDNA, and the relative expression level of COX-2 mRNA was detected by polymerase chain reaction (PCR) technology. The expression level of a housekeeping gene (e.g., GAPDH) was used as an internal reference to normalize the COX-2 mRNA expression data [1] 2. For the detection of COX-2 protein expression: After the same cell treatment process as the mRNA detection experiment, total protein was extracted from the human pulmonary epithelial cells. The protein concentration was determined by a protein quantification method (e.g., BCA assay). Then, equal amounts of protein samples were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) for separation, followed by transferring the separated proteins to a membrane (e.g., PVDF membrane). After blocking the membrane to reduce non-specific binding, the membrane was incubated with a primary antibody specific for COX-2 and a primary antibody for a housekeeping protein (e.g., beta-actin) as an internal reference. Subsequently, the membrane was incubated with a corresponding secondary antibody labeled with a detection reagent. Finally, the signal of the COX-2 protein band was detected by a chemiluminescence or color development method, and the relative expression level of COX-2 protein was quantified by image analysis software [1] |
| Cell Assay |
1. Human pulmonary epithelial cell culture and treatment: Human pulmonary epithelial cells were cultured in a suitable medium containing essential nutrients (e.g., fetal bovine serum, antibiotics) in an incubator with a controlled environment (37°C, 5% CO2). When the cells grew to the logarithmic growth phase or a specific confluency (e.g., 70%-80% confluency), they were seeded into culture plates (e.g., 6-well plates, 96-well plates) for subsequent experiments. The cells were divided into experimental groups: a blank control group (without IL-1beta and Sphondin treatment), an IL-1beta stimulation group (treated with IL-1beta alone to induce COX-2 expression), and multiple Sphondin intervention groups (treated with IL-1beta and different concentrations of Sphondin). The treatment duration was set according to the experimental design (specific duration not mentioned in public information), and during the treatment period, the medium was replaced if necessary to maintain cell viability [1] 2. Detection of cell response to Sphondin: After the treatment period, the cells were collected for subsequent detection of COX-2 expression (as described in the Enzyme Assay section, including mRNA and protein level detection). No information about other cell functional detections (e.g., cell viability, apoptosis) related to Sphondin was found in the publicly available content [1] |
| ADME/Pharmacokinetics |
Metabolism / Metabolites Papilio glaucus (tiger swallowtail) is a generalist that rarely encounters plants containing furanocoumarins yet is constitutively capable of metabolizing low levels of these highly toxic allelochemicals. In larvae of this species, metabolism of linear (xanthotoxin, bergapten), and angular (angelicin, sphondin), furanocoumarins can be induced up to 30-fold by the presence of xanthotoxin in their diet. Degenerate primers corresponding to conserved amino acid sequences in three insect P450s, Musca domestica (CYP6A1), Drosophila melanogaster (CYP6A2) and Papilio polyxenes (CYP6B1), were used to clone xanthotoxin-induced P450 transcripts from P. glaucus larvae by a reverse transcription-polymerase chain reaction (RT-PCR) strategy. Positive clones encoding the highly conserved F--G-R-C-G P450 signature motif were used to isolate a full-length CYP6B4v1 cDNA from a P. glaucus xanthotoxin-induced cDNA library. Sequence comparisons indicate the P. glaucus CYP6B4v1 protein sequence is 63% and 61% identical, respectively, to the P. polyxenes furanocoumarin-inducible CYP6B1v1 and CYP6B3v1 proteins. Northern analysis indicates that CYP6B4 and related transcripts are highly induced in response to xanthotoxin. Baculovirus-mediated expression of the CYP6B4v1 protein in lepidopteran cell lines demonstrates that this P450 isozyme metabolizes isopimpinellin, imperatorin, and bergapten at high rates, xanthotoxin and psoralen at intermediate rates and angelicin, sphondin, and trioxsalen only at very low rates. |
| Toxicity/Toxicokinetics |
Toxicity Summary IDENTIFICATION AND USE: Sphondin is a component of Heracleum maximum roots, these roots are commonly used by the indigenous peoples of North America for the treatment of respiratory ailments including tuberculosis. HUMAN STUDIES: Photoepicutaneous testing showed weak phototoxic effects from sphondin. Photocontact allergy to psoralens in Heracleum laciniatum occurred in two persons volunteering for investigations into phototoxicity of plant homogenates and purified psoralens. Photoallergy was noted following the fifth exposure in case 1, and the sixth in case 2. Testing with diluted solutions demonstrated allergy to sphondin, isobergapten and pimpinellin. ANIMAL STUDIES: Sphondin effectively inhibited mouse coumarin 7-hydroxylase (COH) activity. Sphondin showed anti-proliferative activity and caused G2/M arrest at concentrations of 0.05-15.0 uM when tested against B16F10 melanoma cells. Sphondin possesses an inhibitory effect on IL-1b-induced COX-2 protein expression and PGE2 release in human pulmonary epithelial cell line (A549). The mechanism of action many furocoumarins is based on their ability to form photoadducts with DNA and other cellular components such as RNA, proteins, and several proteins found in the membrane such as phospholipases A2 and C, Ca-dependent and cAMPdependent protein-kinase and epidermal growth factor. Furocoumarins intercalate between base pairs of DNA and after ultraviolet-A irradiation, giving cycloadducts. (L579). Interactions At concentrations up to 6.7 ppm, 8-methoxypsoralen, sphondin, and khellin are not toxic to first-instar larvae of the mosquito Aedes aegypti. The irradiation of sensitized larvae with long-wavelength ultraviolet light did not always produce any immediate toxicity enhancement, but delayed effects were clearly visible. These were observed over the development of the organisms from first-instar larvae to adults. No adverse effects were noted when larvae were irradiated in the absence of sensitizers, or when they were placed in solutions of sensitizers which had been previously irradiated with the same light sources. 8-Methoxypsoralen was slightly more phototoxic than its isomer sphondin. Khellin, recently reported to undergo photoinduced cyclization with DNA components, showed minimal phototoxicity in the concentration range used. |
| References |
[1]. Effects of Sphondin, Isolated From Heracleum Laciniatum, on IL-1beta-induced cyclooxygenase-2 Expression in Human Pulmonary Epithelial Cells. Life Sci. 2002 Nov 29;72(2):199-213. |
| Additional Infomation |
Sphondin is a furanocoumarin. Sphondin has been reported in Heracleum dissectum, Heracleum vicinum, and other organisms with data available. A furanocoumarin derivative isolated from Heracleum laciniatum (L579). Furocoumarins, are phototoxic and photocarcinogenic. They intercalate DNA and photochemically induce mutations. Furocoumarins are botanical phytoalexins found to varying extents in a variety of vegetables and fruits, notably citrus fruits. The levels of furocoumarins present in our diets, while normally well below that causing evident acute phototoxicity, do cause pharmacologically relevant drug interactions. Some are particularly active against cytochrome P450s. For example, in humans, bergamottin and dihydroxybergamottin are responsible for the 'grapefruit juice effect', in which these furanocoumarins affect the metabolism of certain drugs. 1. Sphondin is a natural compound isolated from the plant Heracleum Laciniatum [1] 2. The research background of this literature is related to inflammatory responses: IL-1beta is a key pro-inflammatory cytokine that can induce the expression of COX-2 (an enzyme closely related to the synthesis of inflammatory mediators such as prostaglandins), and the study explores the potential anti-inflammatory effect of Sphondin by regulating COX-2 expression [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~25 mg/mL (~115.64 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (11.56 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (11.56 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (11.56 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.6256 mL | 23.1278 mL | 46.2556 mL | |
| 5 mM | 0.9251 mL | 4.6256 mL | 9.2511 mL | |
| 10 mM | 0.4626 mL | 2.3128 mL | 4.6256 mL |