Physicochemical Properties
| Molecular Formula | C14H26CL2N2O7 |
| Molecular Weight | 405.2714 |
| Exact Mass | 404.111 |
| CAS # | 21736-83-4 |
| Related CAS # | Spectinomycin dihydrochloride pentahydrate;22189-32-8;Spectinomycin;1695-77-8 |
| PubChem CID | 64740 |
| Appearance | White to off-white solid powder |
| Density | 1.43g/cm3 |
| Boiling Point | 583.1ºC at 760 mmHg |
| Melting Point | 194ºC |
| Flash Point | 306.4ºC |
| Vapour Pressure | 5.05E-16mmHg at 25°C |
| Index of Refraction | 1.586 |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 25 |
| Complexity | 478 |
| Defined Atom Stereocenter Count | 9 |
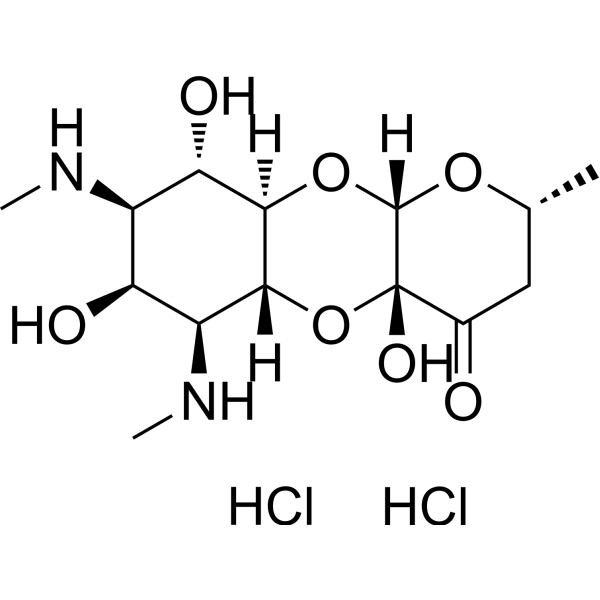
| SMILES | C[C@@H]1CC(=O)[C@]2([C@@H](O1)O[C@@H]3[C@H]([C@@H]([C@@H]([C@@H]([C@H]3O2)NC)O)NC)O)O.Cl.Cl |
| InChi Key | FOGDPGQSHLLYIK-XYQGXRRISA-N |
| InChi Code | InChI=1S/C14H24N2O7.2ClH/c1-5-4-6(17)14(20)13(21-5)22-12-10(19)7(15-2)9(18)8(16-3)11(12)23-14;;/h5,7-13,15-16,18-20H,4H2,1-3H3;2*1H/t5-,7-,8+,9+,10+,11-,12-,13+,14+;;/m1../s1 |
| Chemical Name | (1R,3S,5R,8R,10R,11S,12S,13R,14S)-8,12,14-trihydroxy-5-methyl-11,13-bis(methylamino)-2,4,9-trioxatricyclo[8.4.0.03,8]tetradecan-7-one;dihydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Spectinomycin dihydrochloride targets bacterial 16S rRNA (specifically residues G1064 and C1192) [2] Spectinomycin dihydrochloride targets group 1 intron RNA [3] |
| ln Vitro |
Protein synthesis in cells and E is specifically inhibited by spectinomycin dihydrochloride. Coli extracts: 50 μg/mL of streptomycin dihydrochloride quickly and reversibly inhibits E. coli development and stops the incorporation of amino acids right away [1]. With a maximal inhibition rate of 70-80% in E, spectinomycin diHClide (1 μg/mL or 3 μM) inhibits peptide synthesis directed by endogenous messenger RNA or MS-2 phage RNA. extracted from Coli[1][1]. Spectinomycin dihydrochloride prevents elongation factor G from attaching to the ribosome, hence preventing peptidyl tRNA from translocating from the A site to the P site [2]. Reagents G1064 and 01192 in 16S rRNA are particularly interacting with and may be rendered inactive by streptinomycin diHClide [2]. Due to its dependence on pH variations and Mg2+ concentration, spectinomycin diHClide shows inhibition of splicing, which may indicate electrostatic interactions with intronic RNA [3]. Spectinomycin dihydrochloride (10 μg/mL–100 μg/mL) dose-dependently inhibited protein synthesis in Escherichia coli (E. coli) cell-free extracts: 50 μg/mL reduced [¹⁴C]-leucine incorporation into proteins by 78% [1] Spectinomycin dihydrochloride (20 μM–100 μM) specifically bound to 16S rRNA residues G1064 and C1192 in E. coli ribosomes [2] Spectinomycin dihydrochloride (50 μM–200 μM) inhibited the self-splicing of group 1 intron RNA from Tetrahymena thermophila: 150 μM reduced splicing efficiency by 65% after 60 minutes of incubation at 37°C [3] |
| ln Vivo | It has been demonstrated that in healthy chicks, spectinomycin dihydrochloride (20 mg/kg; IM; 20-100 mg/kg; 9 days) is safe [4]. The majority of the dose of specinomycin dihydrochloride (10 mg/kg; intravenous injection; single dose) is excreted unchanged into the urine by the kidneys [5]. Pharmacokinetics of spectinomycin hydrochloride in rats [5] Parameter C0 (μg/mL) AUC0-∞ (μg·h/mL) Vd (L/kg) CL (L/h/kg) MRT (h) T1/2α (h) T1/2β (h) fe CLrenal (L/h/kg) Eratio non-compartment analysis 44.3 16.8 0.756 0.602 0.757 / / 0.553 0.359 1.00 Three-compartment model 37.8 15.7 0.747 0.649 1.11 / 0.237 0.754 19.5 / / |
| Enzyme Assay |
Protein synthesis inhibition assay: E. coli cell-free extracts were mixed with Spectinomycin dihydrochloride (10 μg/mL–100 μg/mL) in reaction buffer containing [¹⁴C]-leucine, ATP, GTP, and aminoacyl-tRNAs. The mixture was incubated at 37°C for 90 minutes, and [¹⁴C]-leucine incorporation into TCA-precipitable proteins was measured by liquid scintillation counting [1] 16S rRNA binding assay: Purified E. coli 16S rRNA was incubated with Spectinomycin dihydrochloride (20 μM–100 μM) in binding buffer at 25°C for 30 minutes. RNA footprinting analysis with chemical probes identified specific binding residues [2] Group 1 intron self-splicing inhibition assay: Group 1 intron RNA from Tetrahymena thermophila was incubated with Spectinomycin dihydrochloride (50 μM–200 μM) in splicing buffer (containing Mg²⁺ and GTP) at 37°C. Reaction products were separated by PAGE, and splicing efficiency was quantified by densitometry [3] |
| Animal Protocol |
Animal/Disease Models: Arbor Acres plus broiler chicken (15 days old) [4] Doses: 20 mg/kg, 60 mg/kg, 100 mg/kg Route of Administration: intramuscularinjection (chest muscle); 9 Day Experimental Results: Complete blood count, biochemical parameters, histopathology, clinical signs, weight gain and feed conversion ratio (FCR) demonstrated biosafety of 20 mg/kg. Produces mild toxicity at 60 mg/kg. |
| ADME/Pharmacokinetics |
In Sprague-Dawley rats, intramuscular administration of Spectinomycin dihydrochloride (20 mg/kg) showed a Cmax of 112 μg/mL, Tmax of 1.5 hours, and elimination half-life (t1/2) of 2.8 hours [5] |
| Toxicity/Toxicokinetics |
In 1-day-old chicks, intramuscular administration of Spectinomycin dihydrochloride (10 mg/kg, 20 mg/kg, 40 mg/kg) once daily for 7 days: no mortality was observed; the 40 mg/kg group showed no significant changes in body weight, hematological parameters (WBC, RBC, platelets), or biochemical parameters (ALT, AST, BUN, creatinine) [4] Histopathological examination of liver, kidney, heart, and lung from treated chicks showed no drug-related lesions [4] |
| References |
[1]. Inhibition of protein synthesis by spectinomycin. Science. 1965 Sep 3;149(3688):1096-8. [2]. Spectinomycin interacts specifically with the residues G1064 and C1192 in 16S rRNA, thereby potentially freezing this molecule into an inactive conformation. Nucleic Acids Res. 1994 Feb 11;22(3):325-31. [3]. Spectinomycin inhibits the self-splicing of the group 1 intron RNA. Biochem Biophys Res Commun. 2000 Mar 16;269(2):574-9. [4]. Safety evaluation study of lincomycin and spectinomycin hydrochloride intramuscular injection in chickens. Toxicol Rep. 2022 Jan 29;9:204-209. [5]. Pharmacokinetic profile of spectinomycin in rats. Pharmazie. 2013 Aug;68(8):675-6. |
| Additional Infomation |
Spectinomycin dihydrochloride is a hydrochloride obtained by combining spectinomycin with two molar equivalents of hydrochloric acid. An antibiotic that is active against gram-negative bacteria and used (as its pentahydrate) to treat gonorrhea. It has a role as an antibacterial drug and an antimicrobial agent. It contains a spectinomycin(2+). Spectinomycin Hydrochloride is the hydrochloride salt form of spectinomycin, an aminocyclitol antibiotic derived from Streptomyces spectabilis with antibacterial property. Spectinomycin hydrochloride binds to the bacterial 30S ribosomal subunit, thereby inhibiting peptide elongation and protein synthesis, consequently leading to bacterial cell death. An antibiotic produced by Streptomyces spectabilis. It is active against gram-negative bacteria and used for the treatment of gonorrhea. See also: Spectinomycin Hydrochloride (annotation moved to). Spectinomycin dihydrochloride is an aminocyclitol antibiotic that inhibits bacterial protein synthesis by blocking the translocation step [1,2] Its binding to 16S rRNA residues G1064 and C1192 stabilizes the ribosome in an inactive conformation [2] It selectively inhibits group 1 intron RNA self-splicing without affecting eukaryotic pre-mRNA splicing [3] |
Solubility Data
| Solubility (In Vitro) | H2O : ≥ 100 mg/mL (~246.75 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 50 mg/mL (123.37 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4675 mL | 12.3375 mL | 24.6749 mL | |
| 5 mM | 0.4935 mL | 2.4675 mL | 4.9350 mL | |
| 10 mM | 0.2467 mL | 1.2337 mL | 2.4675 mL |