Physicochemical Properties
| Molecular Formula | C12H20N2O3S |
| Molecular Weight | 272.3638 |
| Exact Mass | 272.119 |
| CAS # | 3930-20-9 |
| Related CAS # | Sotalol hydrochloride;959-24-0;Sotalol-d6;1246912-17-3 |
| PubChem CID | 5253 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.239 g/cm3 |
| Boiling Point | 443.3ºC at 760 mmHg |
| Flash Point | 221.9ºC |
| Index of Refraction | 1.57 |
| LogP | 2.634 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 18 |
| Complexity | 330 |
| Defined Atom Stereocenter Count | 0 |
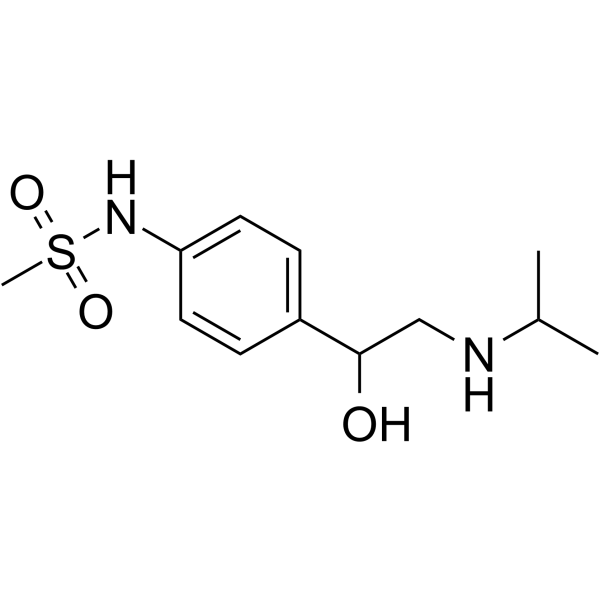
| SMILES | CS(=O)(NC1=CC=C(C(O)CNC(C)C)C=C1)=O |
| InChi Key | ZBMZVLHSJCTVON-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C12H20N2O3S/c1-9(2)13-8-12(15)10-4-6-11(7-5-10)14-18(3,16)17/h4-7,9,12-15H,8H2,1-3H3 |
| Chemical Name | N-[4-[1-hydroxy-2-(propan-2-ylamino)ethyl]phenyl]methanesulfonamide |
| Synonyms | beta-Cardone; DL-Sotalol; Sotalolum; Darob mite; Sotalolum [INN-Latin]; N-(4-(1-Hydroxy-2-(isopropylamino)ethyl)phenyl)methanesulfonamide; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | β-adrenoceptor |
| ln Vitro | In vitro activity: Sotalol hydrochloride is a strong and non-specific β-adrenergic receptor antagonist. With an IC50 value of about 1.2 mM in HEK cell lines, sotalol is also a potassium channel inhibitor. |
| ln Vivo | One antiarrhythmic medication is sotalol.Up to 100 mg/kg of sotalol has no effect on the electroconvulsive threshold. Sotalol does not interfere with the antielectroshock effects of lamotrigine, pregabalin, topiramate, or oxcarbazepine when administered at doses of 80–100 mg/kg. Neither long-term memory nor motor function are hampered by sotalol, either by alone or in combination with antiepileptic medications. Lamotrigine's brain concentration is greatly reduced by sotalol (100 mg/kg), but topiramate's and oxcarbazepine's are increased. Pregabalin levels are unaffected[3]. |
| Cell Assay | Class II antiarrhythmics or β-blockers are antisympathetic nervous system agents that act by blocking β-adrenoceptors. Despite their common clinical use, little is known about the effects of β-blockers on free intracellular calcium (Ca2+ i), an important cytosolic second messenger and a key regulator of cell function. We investigated the role of four chemical analogs, commonly prescribed β-blockers (atenolol, metoprolol, propranolol, and sotalol), on Ca2+ i release and whole-cell currents in mammalian cancer cells (PC3 prostate cancer and MCF7 breast cancer cell lines). We discovered that only propranolol activated free Ca2+ i release with distinct kinetics, whereas atenolol, metoprolol, and sotalol did not. The propranolol-induced Ca2+ i release was significantly inhibited by the chelation of extracellular calcium with ethylene glycol tetraacetic acid (EGTA) and by dantrolene, an inhibitor of the endoplasmic reticulum (ER) ryanodine receptor channels, and it was completely abolished by 2-aminoethoxydiphenyl borate, an inhibitor of the ER inositol-1,4,5-trisphosphate (IP3) receptor channels. Exhaustion of ER stores with 4-chloro-m-cresol, a ryanodine receptor activator, or thapsigargin, a sarco/ER Ca2+ ATPase inhibitor, precluded the propranolol-induced Ca2+ i release. Finally, preincubation of cells with sotalol or timolol, nonselective blockers of β-adrenoceptors, also reduced the Ca2+ i release activated by propranolol. Our results show that different β-blockers have differential effects on whole-cell currents and free Ca2+ i release and that propranolol activates store-operated Ca2+ i release via a mechanism that involves calcium-induced calcium release and putative downstream transducers such as IP3 The differential action of class II antiarrhythmics on Ca2+ i release may have implications on the pharmacology of these drugs[1]. |
| Animal Protocol |
Animal/Disease Models: 20-25 g female Swiss mice[3] Doses: 100 mg/kg Route of Administration: Administered intraperitoneally (ip) Experimental Results: Dramatically diminished the brain concentrations of Lamotrigine and increased those of Oxcarbazepine and Topiramate. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Sotalol is 90-100% bioavailable. When taken with a meal, adsorption is lowered by 18%. In patients with a creatinine clearance >80mL/min, the maximum concentration is 6.25±2.19. 80-90% of a given dose is excreted in the urine as unchanged sotalol. A small fraction of the doses is excreted in the feces as unchanged sotalol. The apparent volume of distribution is 1.2-2.4L/kg. In patients with a creatinine clearance >80mL/min, the plasma clearance is 6.78±2.72L/h and the renal clearance is 4.99±1.43L/h. In patients with a creatinine clearance 30-80mL/min, the plasma clearance is 2.74±0.53L/h and the renal clearance is 2.00±0.67L/h. In patients with a creatinine clearance 10-30mL/min, the plasma clearance is 1.56±0.44L/h and the renal clearance is 0.65±0.31L/h. In patients with a creatinine clearance <10mL/min, the plasma clearance is 0.65±0.20L/h and the renal clearance is 0.27±0.13L/h. Metabolism / Metabolites Sotalol is not metabolized. Biological Half-Life The terminal elimination half life is 10-20 hours in healthy patients. In patients with a creatinine clearance >80mL/min, the half life is 17.5±0.97h. In patients with a creatinine clearance 30-80mL/min, the half life is 22.7±6.4h. In patients with a creatinine clearance 10-30mL/min, the half life is 64±27.2h. In patients with a creatinine clearance <10mL/min, the half life is 97.9±57.3h. |
| Toxicity/Toxicokinetics |
Hepatotoxicity Mild-to-moderate elevations in serum aminotransferase levels occur in less than 2% of patients on sotalol and are usually transient and asymptomatic, resolving even with continuation of therapy. Sotalol has been linked to a single case of clinically apparent liver injury, with onset of an acute hepatitis-like syndrome with prolonged jaundice 12 weeks after sotalol was initiated. The injury improved but did not resolve with discontinuation of sotalol. In large case series of drug induced liver disease and acute liver failure, sotalol has not been listed as a cause. Other beta-blockers have been linked to rare instances of acute clinically apparent liver injury with a latency to onset ranging from 4 to 24 weeks, a hepatocellular pattern of serum enzyme elevations and a mild, self-limiting course without evidence of hypersensitivity or autoimmune reactions. Likelihood score: E (unlikely cause of clinically apparent liver injury). Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Because of its extensive excretion into breastmilk and its renal excretion, other beta-adrenergic blocking drugs are preferred to sotalol, especially while nursing a newborn or preterm infant. Some authors recommend using sotalol during breastfeeding only while monitoring the infant closely for signs of beta-blockade. Infant exposure is predicted to less after the infant is 4 weeks of age or older. ◉ Effects in Breastfed Infants A study of mothers taking beta-blockers during nursing found a numerically, but not statistically significant increased number of adverse reactions in those taking any beta-blocker. Although the ages of infants were matched to control infants, the ages of the affected infants were not stated. One of the mothers was taking sotalol. Bradycardia was not seen in one 12-day-old infant who was breastfed from birth during maternal use of 600 mg of sotalol daily. In another breastfed infant whose mother was taking 80 mg 2 to 3 times daily for more than 3 months, no bradycardia was seen and developmental milestones were achieved normally. Beta-adrenergic blocking drugs with similar breastmilk excretion characteristics and renal elimination have caused adverse effects in breastfed newborns. ◉ Effects on Lactation and Breastmilk Relevant published information on the effects of beta-blockade or sotalol during normal lactation was not found as of the revision date. A study in 6 patients with hyperprolactinemia and galactorrhea found no changes in serum prolactin levels following beta-adrenergic blockade with propranolol. Protein Binding 0%. |
| References |
[1]. Marta Reyes-Corral, et al. Differential Free Intracellular Calcium Release by Class II Antiarrhythmics in Cancer Cell Lines. J Pharmacol Exp Ther. 2019 Apr;369(1):152-162. [2]. Xiaomei Li, et al. Pediatric Dosing of Intravenous Sotalol Based on Body Surface Area in Patients with Arrhythmia. Pediatr Cardiol. 2017 Oct;38(7):1450-1455. [3]. Kinga K Borowicz-Reutt, et al. Sotalol does not interfere with the antielectroshock action of selected second-generation antiepileptic drugs in mice.Pharmacol Rep. 2021 Apr;73(2):516-524. |
| Additional Infomation |
Sotalol is a sulfonamide that is N-phenylmethanesulfonamide in which the phenyl group is substituted at position 4 by a 1-hydroxy-2-(isopropylamino)ethyl group. It has both beta-adrenoreceptor blocking (Vaughan Williams Class II) and cardiac action potential duration prolongation (Vaughan Williams Class III) antiarrhythmic properties. It is used (usually as the hydrochloride salt) for the management of ventricular and supraventricular arrhythmias. It has a role as a beta-adrenergic antagonist, an anti-arrhythmia drug, an environmental contaminant and a xenobiotic. It is a member of ethanolamines, a secondary amino compound, a secondary alcohol and a sulfonamide. It is a conjugate base of a sotalol(1+). Sotalol is a methanesulfonanilide developed in 1960. It was the first of the class III anti arrhythmic drugs. Sotalol was first approved as an oral tablet on 30 October 1992. A racemic mixture of sotalol is currently formulated as a tablet, oral solution, and intravenous injection indicated for life threatening ventricular arrhythmias and maintaining normal sinus rhythm in atrial fibrillation or flutter. Sotalol is an Antiarrhythmic. The mechanism of action of sotalol is as an Adrenergic beta-Antagonist. The physiologic effect of sotalol is by means of Cardiac Rhythm Alteration. Sotalol is a nonselective beta-adrenergic blocker used largely in the therapy cardiac arrhythmias. Sotalol has been linked to at least one instance of clinically apparent liver injury. Sotalol is an ethanolamine derivative with Class III antiarrhythmic and antihypertensive properties. Sotalol is a nonselective beta-adrenergic receptor and potassium channel antagonist. In the heart, this agent inhibits chronotropic and inotropic effects thereby slowing the heart rate and decreasing myocardial contractility. This agent also reduces sinus rate, slows conduction in the atria and in the atrioventricular (AV) node and increases the functional refractory period of the AV node. An adrenergic beta-antagonist that is used in the treatment of life-threatening arrhythmias. See also: Sotalol Hydrochloride (has salt form). Drug Indication Sotalol is indicated to treat life threatening ventricular arrhytmias and maintain normal sinus rhythm in patients with atrial fibrillation or flutter. There are also oral solutions and intravenous injections indicated for patients requiring sotalol, but for whom a tablet would not be appropriate. FDA Label Mechanism of Action Sotalol inhibits beta-1 adrenoceptors in the myocardium as well as rapid potassium channels to slow repolarization, lengthen the QT interval, and slow and shorten conduction of action potentials through the atria. The action of sotalol on beta adrenergic receptors lengthens the sinus node cycle, conduction time through the atrioventricular node, refractory period, and duration of action potentials. Pharmacodynamics Sotalol is a competitive inhibitor of the rapid potassium channel. This inhibition lengthens the duration of action potentials and the refractory period in the atria and ventricles. The inhibition of rapid potassium channels is increases as heart rate decreases, which is why adverse effects like torsades de points is more likely to be seen at lower heart rates. L-sotalol also has beta adrenergic receptor blocking activity seen above plasma concentrations of 800ng/L. The beta blocking ability of sotalol further prolongs action potentials. D-sotalol does not have beta blocking activity but also reduces a patient's heart rate while standing or exercising. These actions combine to produce a negative inotropic effect that reduces the strength of contractility of muscle cells in the heart. Extension of the QT interval is also adversely associated with the induction of arrhythmia in patients. Hyperglycemia is a greater risk for non insulin dependant diabetics than insulin dependant diabetics. Beta blockers inhibit insulin secretion which may cause hyperglycemia in type II diabetes mellitus. The risk of hypoglycemia is higher in insulin dependant diabetes than non insulin dependant diabetics. Beta blockers decrease secretion of insulin, which may mask hypoglycemia in an insulin dependant patient. Beta blockers also increase glucose uptake into cells which may prolong or potentiate hypoglycemia. Further information regarding adverse reactions can be found here. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6716 mL | 18.3581 mL | 36.7161 mL | |
| 5 mM | 0.7343 mL | 3.6716 mL | 7.3432 mL | |
| 10 mM | 0.3672 mL | 1.8358 mL | 3.6716 mL |