Sodium selenite is an inorganic substance commonly used as a source for selenium in studies of cell proliferation and cancer research. Sodium selenite has been used to alter gene expression in HepG2 cells as analyzed by cDNA microarrays. It can inhibit zinc finger protein/DNA interactions. Selenium is an essential trace element normally provided by serum and is present in selenoproteins such as glutathione peroxidase and thioredoxin reductase, which contain the selenium analog of cysteine, selenocysteine, glutathione peroxidase has a role in detoxification in vivo as a scavenger of peroxides.
Physicochemical Properties
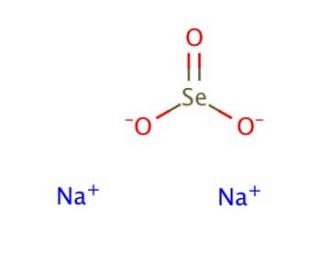
| Molecular Formula | NA2O3SE |
| Molecular Weight | 172.94 |
| Exact Mass | 173.88 |
| CAS # | 10102-18-8 |
| Related CAS # | 14013-56-0 (hydrochloride salt (2:1);15498-87-0 (hydrochloride salt);26970-82-1 (pentahydrate);7782-82-3 (mono-hydrochloride salt);10102-18-8 (Parent) |
| PubChem CID | 24934 |
| Appearance |
Tetragonal prisms White tetragonal crystals White powder |
| Melting Point | 350 °C |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 6 |
| Complexity | 18.8 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | [Na+].[Na+].O=[Se]([O-])[O-].O.O.O.O.O |
| InChi Key | BVTBRVFYZUCAKH-UHFFFAOYSA-L |
| InChi Code | InChI=1S/2Na.H2O3Se/c;;1-4(2)3/h;;(H2,1,2,3)/q2*+1;/p-2 |
| Chemical Name | disodium;selenite |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion ... Rats were depleted of endogenous natural abundance selenium by feeding a single selenium stable isotope ((82)Se-selenite) and then administered (76)Se-selenite and (77)Se-selenomethionine ((77)Se-SeMet)simultaneously. Biological samples were subjected to quantification and speciation analysis by HPLC-ICPMS. Metabolites of the labeled (76)Se and (77)Se and interaction with endogenous selenium were traced and examined without interference from the corresponding endogenous natural abundance isotopes. Differences in the distribution and metabolism among organs and between the two nutritional selenocompounds were compared under exactly identical biological and analytical conditions: (1) selenite was distributed more efficiently than SeMet in organs and body fluids except the pancreas. (2) SeMet was taken up by organs in its intact form. (3) Selenium of SeMet origin was distributed selectively in the pancreas and mostly bound to a protein together with intact SeMet. (4) Selenosugars A and B but not trimethylselenonium (TMSe) were detected in the liver. (5) Selenosugar B and TMSe were detected in the kidneys. Rats were injected intraperitoneally with (75)Se sodium selenite (5 mg/kg), and it was concluded that selenium in the form of selenite accumulated in the anterior pituitary gland. The maximum selenium content was observed after 2 hours, at which time the anterior pituitary gland contained 2.9 mg/g wet weight. The selenium contact in pituitary glands from untreated rats was 0.48 mg/g wet weight. With an adequate supplementation of the diet (1.0 mg/kg), 67% of a tracer dose of selenite was excreted in the urine, whereas in a state of deficiency only 6% of the same dose was excreted. /Selenite/ A tracer dose of selenite accumulated in red blood cells, translocated to plasma proteins, and then to the liver. Selenoprotein P seems to be involved in the transport of Se from the liver to other tissues, even though other transporters may exist and different organs may have different preferences for selenium sources. /Selenite/ For more Absorption, Distribution and Excretion (Complete) data for SODIUM SELENITE (16 total), please visit the HSDB record page. Metabolism / Metabolites Most dietary selenium is in the form of selenomethionine (the major dietary form of selenium) or selenocysteine, both of which are well absorbed. Other forms of selenium include selenate and selenite, which are not major dietary constituents, but are commonly used in fortified foods and dietary supplements. Two pools of reserve selenium are present in the body. The first is as selenomethionine, which is not known to have a physiological function separate from that of methionine. The second reserve pool is the selenium found in liver glutathione peroxidase. Ingested selenite, selenate, and selenocysteine are all metabolized directly to selenide, the reduced form of selenium. Selenomethionine can also be metabolized to selenide. /Selenite/ The addition of sodium selenite (Na2SeO3) to the drinking water of mice for 14 days resulted in the formation of dimethylselenide and also dimethyldiselenide in the breath. Exhalation seems to be a minor form of selenium elimination. Selenate and selenite injected intravenously into rats were speciated by the HPLC-ICP MS method with use of an enriched stable isotope as the tracer. In dose-relation experiments, 82(Se)-enriched selenate or selenite was injected intravenously into male Wistar rats of 8 weeks of age (three rats/group) at single doses of 10, 25, 50, 100 and 200 ug/kg body weight for the selenate group, and 2, 5, 10, 25 and 50 ug/kg body weight for the selenite group. The animals were sacrificed 1 or 24 hr later, and the concentrations and distributions of selenium-82 in the liver, kidneys, serum, and urine remaining in the bladder or 24-hr urine were determined. In time-course experiments, (82)Se-enriched selenate and selenite were injected at doses of 50 and 10 ug/kg body weight, respectively, and the animals were sacrificed 5, 15, 30, 60 and 180 min later. It was suggested that selenate is directly taken up by the liver with an efficiency of approximately 1/2 compared with selenite, the latter being taken up by the liver after being metabolized to selenide in red blood cells. Although selenate and selenite were metabolized differently in the bloodstream, and also a part of only selenate was excreted directly into the urine, the selenium-82 taken up by the liver was shown to be metabolized in a manner indistinguishable between selenate and selenite. Selenium-82 of selenite origin but not of selenate origin was suggested to undergo redox reaction in the bloodstream. These results suggest that although parenteral selenate is utilized less efficiently by the body, it is utilized in the liver in a similar manner to selenite much more safely. /Selenite/ Metabolic pathways for Se in the body were studied for selenite and selenate, with the use of enriched (82)Se ... . The concentrations of (82)Se in organs and body fluids and the distributions of their constituents depending on the dose and time after the intravenous administration of (82)Se-selenite and -selenate to rats were determined. Selenite was taken up by red blood cells within several minutes, reduced to selenide by glutathione, and then transported to the plasma, bound selectively to albumin and transferred to the liver. Contrary to selenite, intact selenate was either taken up directly by the liver or excreted into the urine. The (82)Se of selenite origin and that of selenate origin were detected in the forms of the two Se peak materials in the liver, A and B. The former one was methylated to the latter in vivo and in vitro. The latter one was identical with the major urinary metabolite and it was identified as Se-methyl-N-acetyl-selenohexosamine (selenosugar). The chemical species-specific metabolic pathway for Se was explained by the metabolic regulation through selenide as the assumed common intermediate for the inorganic and organic Se sources and as the checkpoint metabolite between utilization for the selenoprotein synthesis and methylation for the excretion of Se. /Selenite/ For more Metabolism/Metabolites (Complete) data for SODIUM SELENITE (6 total), please visit the HSDB record page. Selenium may be absorbed through inhalation and ingestion, while some selenium compounds may also be absorbed dermally. Once in the body, selenium is distributed mainly to the liver and kidney. Selenium is an essential micronutrient and is a component of glutathione peroxidase, iodothyronine 5'-deiodinases, and thioredoxin reductase. Organic selenium is first metabolized into inorganic selenium. Inorganic selenium is reduced stepwise to the intermediate hydrogen selenide, which is either incorporated into selenoproteins after being transformed to selenophosphate and selenocysteinyl tRNA or excreted into the urine after being transformed into methylated metabolites of selenide. Elemental selenium is also methylated before excretion. Selenium is primarily eliminated in the urine and feces, but certain selenium compounds may also be exhaled. (L619) Biological Half-Life In humans, whole body retention studies following oral administration of sodium selenite have indicated that selenium elimination is triphasic. During the initial phase, which lasted about 1 week, elimination of selenium was rapid, with a half-life of approximately 1 day. In the second phase, which also lasted approximately 1 week, selenium elimination was slower, with a half-life of 8-9 days. In the third phase, selenium elimination was much slower, with a half-life estimated to be 115-116 days. The first two elimination phases correspond to the fecal elimination of nonabsorbed selenium and the urinary excretion of absorbed but unutilized selenium Following a single, oral, 200 ug dose of sodium selenite in six male and female volunteers, the half-time was 200 to 285 hours for terminal plasma elimination and 115 to 285 days for tissues. The excretion pattern of a single exposure to selenite appears to have at least two phases: a rapid initial phase with as much as 15 to 40 percent of the absorbed dose excreted in the urine the first week. There is expotential excretion of the remainder of the dose with a half life of 103 days. /Selenite/ |
| Toxicity/Toxicokinetics |
Toxicity Summary Selenium readily substitutes for sulfur in biomolecules and in many biochemical reactions, especially when the concentration of selenium is high and the concentration of sulfur is low. Inactivation of the sulfhydryl enzymes necessary for oxidative reactions in cellular respiration, through effects on mitochondrial and microsomal electron transport, might contribute to acute selenium toxicity. Selenomethionine (a common organic selenium compound) also appears to randomly substitute for methionine in protein synthesis. This substitution may affect the structure and functionability of the protein, for example, by altering disulfide bridges. Inorganic forms of selenium appear to react with tissue thiols by redox catalysis, resulting in formation of reactive oxygen species and causing damage by oxidative stress. (L619) Toxicity Data LD50: 7 mg/kg (Oral, Rat) (L738) LD50: 3 mg/kg (Intravenous, Rat) (L738) Interactions The highest lead concn (1 mM) reduced the percentage of selenite absorbed from the in situ ligated duodenal loop, but did not influence the retention of the orally admin cmpd. /Selenite/ ... Groups of 5 mice received either 2 daily ip injections each of 2 mg/kg sodium selenite pentahydrate (Na2SeO3.5H2O) on successive days or 2 daily sc injections of 0.1 mL saline. Forty eight hr after the last Se injection, the Se treated group and 1 of the control groups received 20 mg/kg silver lactate ip and the other control group received a sodium lactate injection. Livers were analyzed 3 hr after the injection of silver. In a control experiment, 1 group of mice received sodium selinite and another group saline and livers were analyzed 51 hr after the last Se injection. Pretreatment of mice with Se resulted in a significant increase of silver induced lipid peroxidation in liver, when compared with animals exposed to silver alone (p< 0.001) or with controls (p< 0.001). The doses of selenium used in the experiment did not increase or decrease lipid peroxidation. /Sodium selenite pentahydrate/ Acute treatment with sodium selenite effectively reduces bromobenzene hepatotoxicity in male rats. Male Porton Wistar rats were tested to determine the protection exerted by selenite, selenomethionine, selenium, or biological selenium (in the form of the liver soluble fraction from animals treated with sodium selenite) against the toxic effects of mercury. Mercury was given as mercuric chloride, injected subcutaneously in doses of 2.5, or 7.5 umoles/kg. The animals received equimolar doses of selenium compounds at the time of treatment with mercury. A reduction in the level of mercury in the urine was recorded 48 hours after treatment with both biological selenium and selenomethionine. Treatment with selenite induced a greater decrease in the levels of urinary mercury, together with a significant reduction of the levels of mercury in the kidney. The activity of alkaline phosphatase in urine and the levels of plasma urea nitrogen recorded in animals treated with mercury in doses of 2.5 or 5 umoles/kg were lowest in the rats treated with selenium, followed by biological selenium, and selenite in that order. The degree of necrosis seen in the proximal kidney tubules of animals treated with 5 or 7.5 umoles/kg mercury showed the same pattern, while in animals treated with the lowest dose of mercury necrotic damage was slight. These results support the hypothesis that the protective effect of selenium against mercury renal toxicity is due to the formation of mercury selenide. For more Interactions (Complete) data for SODIUM SELENITE (23 total), please visit the HSDB record page. Non-Human Toxicity Values LD50 Rat female oral 0.0147 g/kg bw LD50 Rat male oral 0.0171 g/kg bw LD50 Lamb oral 119 mg/kg LD50 Rat oral 7mg/kg For more Non-Human Toxicity Values (Complete) data for SODIUM SELENITE (11 total), please visit the HSDB record page. |
| Additional Infomation |
Sodium selenite appears as a white colored crystalline solid. Soluble in water and more dense than water. Contact may irritate skin, eyes and mucous membranes. Toxic by ingestion, inhalation and skin absorption. Disodium selenite is an inorganic sodium salt composed of sodium and selenite ions in a 2:1 ratio. It has a role as a nutraceutical. It is a selenite salt and an inorganic sodium salt. Sodium Selenite is an inorganic form of the trace element selenium with potential antineoplastic activity. Selenium, administered in the form of sodium selenite, is reduced to hydrogen selenide (H2Se) in the presence of glutathione (GSH) and subsequently generates superoxide radicals upon reaction with oxygen. This may inhibit the expression and activity of the transcription factor Sp1; in turn Sp1 down-regulates androgen receptor (AR) expression and blocks AR signaling. Eventually, selenium may induce apoptosis in prostate cancer cells and inhibit tumor cell proliferation. Sodium selenite is a chemical compound of sodium and selenium. It is the most common water-soluble form of selenium. Together with the related barium and zinc salts, sodium selenite is mainly used in the manufacture of colorless glass. Its pink coloration cancels out the green color imparted by iron impurities. Selenium is a nonmetal element with the atomic number 34 and the chemical symbol Se. Selenium rarely occurs in its elemental state in nature and is usually found in sulfide ores such as pyrite, partially replacing the sulfur in the ore matrix. It may also be found in silver, copper, lead, and nickel minerals. Though selenium salts are toxic in large amounts, trace amounts of the element are necessary for cellular function in most animals, forming the active center of the enzymes glutathione peroxidase, thioredoxin reductase, and three known deiodinase enzymes. (L620, L737) The disodium salt of selenious acid. It is used therapeutically to supply the trace element selenium and is prepared by the reaction of SELENIUM DIOXIDE with SODIUM HYDROXIDE. See also: Selenium (has active moiety); Selenite ion (has active moiety); Sodium Selenite; Vitamin E (component of) ... View More ... Therapeutic Uses Used therapeutically to supply the trace element selenium. Medication (Vet): Prevention and treatment of white muscle disease (selenium-tocopherol deficiency syndrome) in cattle, sheep, and swine /L-SE and BO-SE/ Medication (Vet): As an aid in alleviating and controlling inflammation, pain, and lameness associated with certain arthropathies in dogs. /Seletoc Minicaps and Caps, Seletoc Injection/ Medication (Vet): For the prevention and treatment of (selenium-tocopherol deficiency syndrome) in cattle. /Mu-Se, Velenium/ For more Therapeutic Uses (Complete) data for SODIUM SELENITE (9 total), please visit the HSDB record page. Drug Warnings The most frequently reported adverse reactions of selenosis or chronic selenium toxicity are hair and nail brittleness and loss. Other symptoms include skin rash, garlic-like breath odor, fatigue, irritability and nausea and vomiting. /Selenium containing preparations/ Intakes of selenium less than 900 ug daily (for adults) are unlikely to cause adverse reactions. Prolonged intakes of selenium of doses of 1,000 ug (or 1 mg) or greater daily may cause adverse reactions. /Selenium countaining preparations/ Pregnant women and nursing mothers should avoid selenium intakes greater than Recommended Dietary Allowance amounts. /Selenium countaining preparations/ |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.7824 mL | 28.9118 mL | 57.8235 mL | |
| 5 mM | 1.1565 mL | 5.7824 mL | 11.5647 mL | |
| 10 mM | 0.5782 mL | 2.8912 mL | 5.7824 mL |