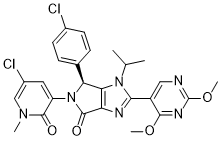
Siremadlin (formerly also known as NVP-HDM201; NVP-HDM-201; HDM-201; HDM201) is a novel, potent, highly specific and orally bioavailable inhibitor of MDM-2/p53 with potential antitumor activity. It has potential antineoplastic activity as a human double minute 2 homolog (HDM2) inhibitor. Siremadlin prevents the HDM2 protein from attaching to the p53 tumor suppressor protein's transcriptional activation domain. Cancer cells frequently overexpress HDM2, a zinc finger protein that is a negative regulator of the p53 pathway and has been linked to cancer cell survival and proliferatio.
Physicochemical Properties
| Molecular Formula | C26H24CL2N6O4 |
| Molecular Weight | 555.41 |
| Exact Mass | 554.123 |
| Elemental Analysis | C, 56.23; H, 4.36; Cl, 12.77; N, 15.13; O, 11.52 |
| CAS # | 1448867-41-1 |
| Related CAS # | Siremadlin (R Enantiomer);1448867-42-2 |
| PubChem CID | 71678098 |
| Appearance | White to off-white solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 736.5±70.0 °C at 760 mmHg |
| Flash Point | 399.2±35.7 °C |
| Vapour Pressure | 0.0±2.4 mmHg at 25°C |
| Index of Refraction | 1.688 |
| LogP | 2.01 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 38 |
| Complexity | 987 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | O=C1N(C2=CC(Cl)=CN(C)C2=O)[C@@H](C3=CC=C(Cl)C=C3)C4=C1N=C(C5=CN=C(OC)N=C5OC)N4C(C)C |
| InChi Key | AGBSXNCBIWWLHD-FQEVSTJZSA-N |
| InChi Code | InChI=1S/C26H24Cl2N6O4/c1-13(2)33-21-19(30-22(33)17-11-29-26(38-5)31-23(17)37-4)25(36)34(18-10-16(28)12-32(3)24(18)35)20(21)14-6-8-15(27)9-7-14/h6-13,20H,1-5H3/t20-/m0/s1 |
| Chemical Name | (4S)-5-(5-chloro-1-methyl-2-oxopyridin-3-yl)-4-(4-chlorophenyl)-2-(2,4-dimethoxypyrimidin-5-yl)-3-propan-2-yl-4H-pyrrolo[3,4-d]imidazol-6-one |
| Synonyms | HDM201; NVP-HDM201; NVP-HDM-201; HDM 201; HDM-201; NVP HDM 201; NVP-HDM 201 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | p53-MDM2 interaction | ||
| ln Vitro |
HDM201 binds to the p53 binding-site of the Mdm2 protein, disrupting the interaction of the two proteins and leading to the activation of the p53 pathway[1]. In human p53 wild-type tumor cells, it causes a robust cell cycle arrest and apoptosis. A variety of cancer cell lines show that HDM201 is highly selective[2]. |
||
| ln Vivo |
|
||
| Cell Assay | At Horizon Discovery (Cambridge, MA), cancer cell lines were subjected to an in vitro combination screen, and data analysis was carried out as previously mentioned. Here, the data analysis was concentrated on combinations involving CGM097, a member of an earlier generation of selective TP53-MDM2 inhibitors. CGM097 and 25 additional compounds were applied to a total of 485 cancer cell lines at various concentrations. We combined the data regarding the presence or absence of TP53 mutations and distinguished between cell lines with and without TP53 mutations. By combining HDM201 and A-1155463 as previously mentioned, we evaluated the synergistic effect. | ||
| Animal Protocol |
|
||
| References |
[1]. Cancer Res (2016) 76 (14_Supplement): 1239. [2]. Cancer Res (2016) 76 (14_Supplement): 4855. [3]. Cancers (Basel) . 2021 Dec 21;14(1):14. [4]. Eur J Cancer . 2020 Feb;126:93-103. |
||
| Additional Infomation | Siremadlin is an orally bioavailable human double minute 2 homolog (HDM2) inhibitor with potential antineoplastic activity. Siremadlin inhibits the binding of the HDM2 protein to the transcriptional activation domain of the tumor suppressor protein p53. By preventing this HDM2-p53 interaction, the proteasome-mediated enzymatic degradation of p53 is inhibited, which may result in the restoration of both p53 signaling and p53-mediated induction of tumor cell apoptosis. HDM2, a zinc finger protein and negative regulator of the p53 pathway, is often overexpressed in cancer cells and has been implicated in cancer cell proliferation and survival. |
Solubility Data
| Solubility (In Vitro) |
DMSO: 56.75~100 mg/mL (102.2~180.1 mM) Ethanol: ~3 mg/mL (~5.4 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.50 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.50 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 3: ≥ 2.5 mg/mL (4.50 mM) (saturation unknown) in 5% DMSO + 40% PEG300 + 5% Tween80 + 50% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8005 mL | 9.0024 mL | 18.0047 mL | |
| 5 mM | 0.3601 mL | 1.8005 mL | 3.6009 mL | |
| 10 mM | 0.1800 mL | 0.9002 mL | 1.8005 mL |