Physicochemical Properties
| Molecular Formula | C25H22O10 |
| Molecular Weight | 482.4362 |
| Exact Mass | 482.121 |
| CAS # | 33889-69-9 |
| PubChem CID | 441764 |
| Appearance | White to off-white solid powder |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 782.0±60.0 °C at 760 mmHg |
| Flash Point | 270.5±26.4 °C |
| Vapour Pressure | 0.0±2.8 mmHg at 25°C |
| Index of Refraction | 1.716 |
| LogP | 2.2 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 35 |
| Complexity | 765 |
| Defined Atom Stereocenter Count | 4 |
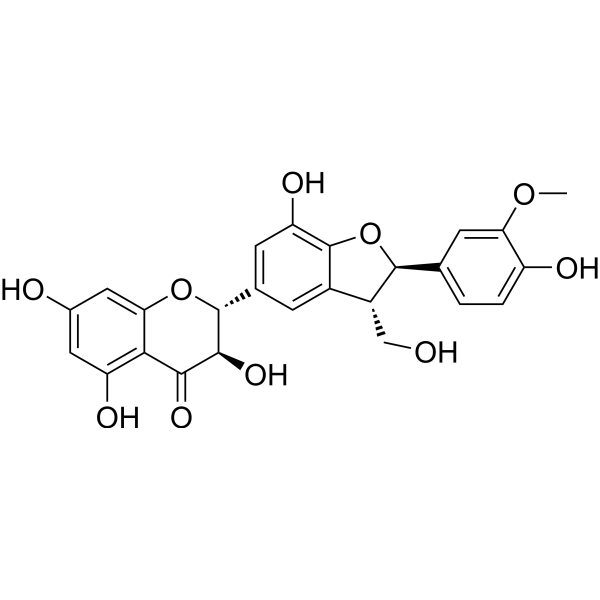
| SMILES | COC1=C(C=CC(=C1)[C@H]2[C@@H](C3=C(O2)C(=CC(=C3)[C@@H]4[C@H](C(=O)C5=C(C=C(C=C5O4)O)O)O)O)CO)O |
| InChi Key | BMLIIPOXVWESJG-LMBCONBSSA-N |
| InChi Code | InChI=1S/C25H22O10/c1-33-18-6-10(2-3-15(18)28)23-14(9-26)13-4-11(5-17(30)25(13)35-23)24-22(32)21(31)20-16(29)7-12(27)8-19(20)34-24/h2-8,14,22-24,26-30,32H,9H2,1H3/t14-,22+,23+,24-/m1/s1 |
| Chemical Name | (2R,3R)-3,5,7-trihydroxy-2-[(2R,3S)-7-hydroxy-2-(4-hydroxy-3-methoxyphenyl)-3-(hydroxymethyl)-2,3-dihydro-1-benzofuran-5-yl]-2,3-dihydrochromen-4-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Monocarboxylate Transporter 8 (MCT8) (IC50 = 0.45 μM for T3 transport inhibition) [2] |
| ln Vitro |
In MCT8-overexpressing MDCK1 cells, silychristin has a potent inhibitory effect on MCT8-mediated T3 uptake, with an IC50 of 110 nM [2]. Fibroblasts are not cytotoxically affected by silymarin [3]. Silymarin (6.5-75 μM; 24 hours) has dose-dependent protective effects against UVA toxicity and ROS generation [3]. Cells' levels of metalloproteinase-1 (MMP-1) are decreased by silychristin (12.5μM, 25μM) [3]. - Silychristin is a potent inhibitor of MCT8-mediated thyroid hormone (T3) transport. In HEK293 cells overexpressing human MCT8, it dose-dependently inhibited [¹²⁵I]-T3 uptake with an IC50 of 0.45 μM (0.1-10 μM concentration range). It did not affect the activity of other thyroid hormone transporters (e.g., OATP1C1, LAT1) at concentrations up to 10 μM (radioligand uptake assay) [2] - In human dermal fibroblasts exposed to UVA radiation (3 J/cm²), Silychristin (1, 5, 10 μM) exhibited UVA-photoprotective effects. It dose-dependently reduced reactive oxygen species (ROS) production (DCFH-DA staining), inhibited UVA-induced DNA damage (comet assay, 8-oxo-dG immunostaining), and improved cell viability (MTT assay) with a 35% viability increase at 10 μM compared to UVA-only group. It also upregulated the expression of antioxidant proteins Nrf2 and HO-1 (Western blot) [3] - Silychristin (1-20 μM) modulated osteoblast-related biological activities in vitro. It promoted the proliferation of MC3T3-E1 pre-osteoblasts (CCK-8 assay) and enhanced alkaline phosphatase (ALP) activity (colorimetric assay) at 5-10 μM, indicating potential effects on osteogenic differentiation [1] |
| Enzyme Assay | - MCT8 transport activity assay: HEK293 cells stably expressing human MCT8 were seeded in 24-well plates and incubated with Silychristin (0.1-10 μM) for 30 minutes. [¹²⁵I]-T3 was added to the medium, and cells were incubated for another 15 minutes at 37°C. The reaction was terminated by washing with ice-cold buffer, and cell-associated radioactivity was measured by a gamma counter. The inhibition rate of T3 uptake was calculated, and IC50 value was derived from dose-response curves [2] |
| Cell Assay |
Cell viability assay [3] Cell Types: NHDF Tested Concentrations: 6.5 μM, 12.5 μM, 25 μM, 50 μM, 75 μM Incubation Duration: 24 hrs (hours) Experimental Results: UVA toxicity is diminished and ROS generation is diminished in a dose-dependent manner. Cell viability assay[3] Cell Types: NHDF Tested Concentrations: 12.5 μM, 25 μM Incubation Duration: Experimental Results: diminished metalloproteinase-1 (MMP-1) levels in cells. - MCT8 inhibition cell assay: HEK293 cells were transfected with human MCT8 expression plasmid or empty vector. After 48 hours, cells were treated with Silychristin (0.1-10 μM) and [¹²⁵I]-T3. Radioactivity was quantified to evaluate MCT8-specific transport inhibition. The selectivity was assessed by testing the compound on cells expressing OATP1C1 or LAT1 [2] - UVA photoprotection assay: Human dermal fibroblasts were seeded in 96-well plates or coverslips and pre-treated with Silychristin (1-10 μM) for 24 hours. Cells were exposed to UVA radiation (3 J/cm²) and incubated for another 24 hours. ROS production was detected by DCFH-DA staining; DNA damage by comet assay and 8-oxo-dG immunofluorescence; cell viability by MTT assay; Nrf2/HO-1 expression by Western blot [3] - Osteoblast activity assay: MC3T3-E1 pre-osteoblasts were seeded in 96-well plates or 6-well plates and treated with Silychristin (1-20 μM) for 3-7 days. Cell proliferation was measured by CCK-8 assay; ALP activity was detected by colorimetric assay using an ALP substrate kit; osteogenic differentiation-related gene expression was analyzed by qPCR (collagen I, osteocalcin) [1] |
| References |
[1]. Silychristin: Skeletal Alterations and Biological Activities. J Nat Prod. 2016 Dec 23;79(12):3086-3092. [2]. Silychristin, a Flavonolignan Derived From the Milk Thistle, Is a Potent Inhibitor of the Thyroid Hormone Transporter MCT8. Endocrinology. 2016 Apr;157(4):1694-701. [3]. A pilot study of the UVA-photoprotective potential of dehydrosilybin, isosilybin, silychristin, and silydianin on human dermal fibroblasts. Arch Dermatol Res. 2019 Aug;311(6):477-490. |
| Additional Infomation |
Silychristin is a flavonolignan isolated from Silybum marianum and has been shown to exhibit inhibitory activities against lipoxygenase and prostaglandin synthetase. It has a role as a radical scavenger, a lipoxygenase inhibitor, a prostaglandin antagonist and a metabolite. It is a flavonolignan, a member of 1-benzofurans, a polyphenol, an aromatic ether and a secondary alpha-hydroxy ketone. Silicristin has been reported in Silybum eburneum, Anastatica hierochuntica, and other organisms with data available. - Silychristin is a natural flavonolignan compound isolated from the seeds of Silybum marianum (milk thistle) [1][2][3] - Its core biological mechanisms include: specific inhibition of MCT8-mediated thyroid hormone transport [2]; UVA-photoprotection via activating the Nrf2-HO-1 antioxidant pathway, reducing ROS and DNA damage [3]; modulation of osteoblast proliferation and differentiation related to skeletal biology [1] - Silychristin exhibits potential application value in thyroid hormone transport disorders, skin photoprotection, and skeletal-related research |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~207.28 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.18 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.18 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.18 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0728 mL | 10.3640 mL | 20.7280 mL | |
| 5 mM | 0.4146 mL | 2.0728 mL | 4.1456 mL | |
| 10 mM | 0.2073 mL | 1.0364 mL | 2.0728 mL |