Physicochemical Properties
| Molecular Formula | C25H22O10 |
| Molecular Weight | 482.43618 |
| Exact Mass | 482.121 |
| Elemental Analysis | C, 62.24; H, 4.60; O, 33.16 |
| CAS # | 142797-34-0 |
| Related CAS # | Silybin A;22888-70-6;Isosilybin;72581-71-6;Silybin;802918-57-6;Silymarin;65666-07-1 |
| PubChem CID | 1548994 |
| Appearance | White to off-white solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 793.0±60.0 °C at 760 mmHg |
| Melting Point | 158-160℃ (methanol water ) |
| Flash Point | 274.5±26.4 °C |
| Vapour Pressure | 0.0±2.9 mmHg at 25°C |
| Index of Refraction | 1.684 |
| LogP | 2.59 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 35 |
| Complexity | 750 |
| Defined Atom Stereocenter Count | 4 |
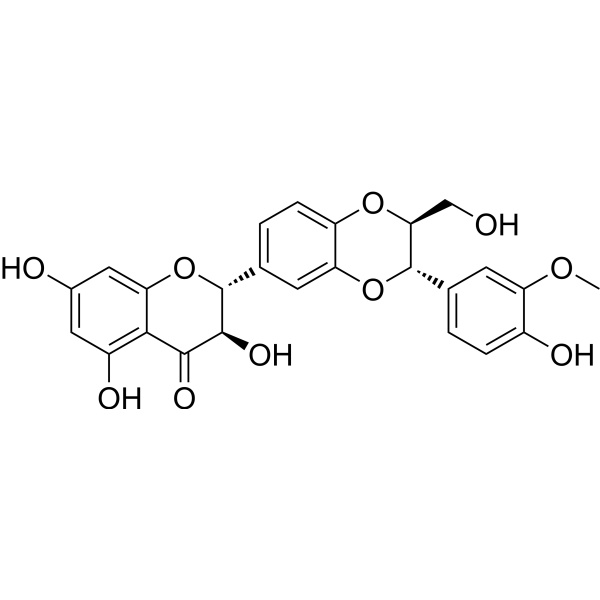
| SMILES | COC1=C(C=CC(=C1)[C@H]2[C@@H](OC3=C(O2)C=C(C=C3)[C@@H]4[C@H](C(=O)C5=C(C=C(C=C5O4)O)O)O)CO)O |
| InChi Key | SEBFKMXJBCUCAI-WAABAYLZSA-N |
| InChi Code | InChI=1S/C25H22O10/c1-32-17-6-11(2-4-14(17)28)24-20(10-26)33-16-5-3-12(7-18(16)34-24)25-23(31)22(30)21-15(29)8-13(27)9-19(21)35-25/h2-9,20,23-29,31H,10H2,1H3/t20-,23-,24-,25+/m0/s1 |
| Chemical Name | (2R,3R)-3,5,7-trihydroxy-2-[(2S,3S)-3-(4-hydroxy-3-methoxyphenyl)-2-(hydroxymethyl)-2,3-dihydro-1,4-benzodioxin-6-yl]-2,3-dihydrochromen-4-one |
| Synonyms | Silibinin B; 142797-34-0; UNII-853OHH1429; DTXSID30858697; 4H-1-BENZOPYRAN-4-ONE, 2-((2S,3S)-2,3-DIHYDRO-3-(4-HYDROXY-3-METHOXYPHENYL)-2-(HYDROXYMETHYL)-1,4-BENZODIOXIN-6-YL)-2,3-DIHYDRO-3,5,7-TRIHYDROXY-, (2R,3R)-; 4H-1-Benzopyran-4-one, 2-[(2S,3S)-2,3-dihydro-3-(4-hydroxy-3-methoxyphenyl)-2-(hydroxymethyl)-1,4-benzodioxin-6-yl]-2,3-dihydro-3,5,7-trihydroxy-, (2R,3R)-; DTXCID703580; SILYBIN B (CONSTITUENT OF MILK THISTLE); |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Natural flavonolignan from Silybum marianum; anti-tumor |
| ln Vitro |
Silybins A and B (4 and 5) are the major components in the active extract of milk thistle (silymarin) and have been linked previously to skin cancer prevention effects via anti-inflammatory, antioxidant, and immunomodulatory mechanisms (Katiyar, Citation2005). Our study results now reveal that all seven major compounds in silymarin show good EBV-EA inhibition activities. In our testing model, silychristins A and B (1 and 2) and isosilybins A and B (6 and 7) had better inhibition effects compared with β.-carotene, and silybins A and B (4 and 5) showed lower activity compared with the other flavonolignans in silymarin. Further in vivo. testing on mouse skin papillomas is ongoing.
In conclusion, seven pure compounds silychristin A (1), silychristin B (2), silydianin (3), silybin A (4), Silybin B (5), isosilybin A (6), and isosilybin B (7) were isolated from an extract of milk thistle. Evaluation with an in vitro. EBV-EA activation assay showed that silychristin B (2) was the most active compound with 94.9% EBV-EA inhibition at 1000 mol ratio/TPA. As it also showed low cytotoxicity, 2 could be valuable as an antitumor promoter or as a lead compound for new cancer preventive drug development. [1] Here, we combine biophysical (ThT assays, TEM and AFM imaging), biochemical (WB and ESI-MS), and computational (all-atom molecular dynamics) techniques to investigate the capacity of four optically pure components of the natural product silymarin (silybin A, Silybin B, 2,3-dehydrosilybin A, 2,3-dehydrosilybin B) to inhibit Aβ aggregation. Despite TEM analysis demonstrated that all the four investigated flavonoids prevent the formation of mature fibrils, ThT assays, WB and AFM investigations showed that only Silybin B was able to halt the growth of small-sized protofibrils thus promoting the formation of large, amorphous aggregates. Molecular dynamics (MD) simulations indicated that silybin B interacts mainly with the C-terminal hydrophobic segment 35MVGGVV40 of Aβ40. Consequently to silybin B binding, the peptide conformation remains predominantly unstructured along all the simulations. By contrast, silybin A interacts preferentially with the segments 17LVFF20 and 27NKGAII32 of Aβ40 which shows a high tendency to form bend, turn, and β-sheet conformation in and around these two domains. Both 2,3-dehydrosilybin enantiomers bind preferentially the segment 17LVFF20 but lead to the formation of different small-sized, ThT-positive Aβ aggregates. [2] |
| ln Vivo | Finally, in vivo studies in a transgenic Caenorhabditis elegans strain expressing human Aβ indicated that Silybin B is the most effective of the four compounds in counteracting Aβ proteotoxicity. This study underscores the pivotal role of stereochemistry in determining the neuroprotective potential of silybins and points to Silybin B as a promising lead compound for further development in anti-AD therapeutics [2]. |
| Toxicity/Toxicokinetics | mouse LD50 oral >1600 mg/kg German Offenlegungsschrift Patent Document., #2423725 |
| References |
[1]. Cancer Preventive Agents. 7. Antitumor-Promoting Effects of Seven Active Flavonolignans from Milk Thistle (Silybum marianum.) on Epstein-Barr Virus Activation, Pharmaceutical Biology, 45:10, 735-738. [2]. Inhibition of Aβ Amyloid Growth and Toxicity by Silybins: The Crucial Role of Stereochemistry. ACS Chem Neurosci. 2017 Aug 16;8(8):1767-1778. |
| Additional Infomation |
Silybin B is a flavonolignan. Silibinin B has been reported in Aspergillus iizukae, Anastatica hierochuntica, and other organisms with data available. Silymarin is a mixture of flavonolignans isolated from the milk thistle plant Silybum marianum. Silymarin may act as an antioxidant, protecting hepatic cells from chemotherapy-related free radical damage. This agent may also promote the growth of new hepatic cells. (NCI04) The major active component of silymarin flavonoids extracted from seeds of the MILK THISTLE, Silybum marianum; it is used in the treatment of HEPATITIS; LIVER CIRRHOSIS; and CHEMICAL AND DRUG INDUCED LIVER INJURY, and has antineoplastic activity; silybins A and B are diastereomers. See also: Milk Thistle (part of); Silybin (annotation moved to). |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~518.20 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.31 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.31 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.31 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0728 mL | 10.3640 mL | 20.7280 mL | |
| 5 mM | 0.4146 mL | 2.0728 mL | 4.1456 mL | |
| 10 mM | 0.2073 mL | 1.0364 mL | 2.0728 mL |