Physicochemical Properties
| Molecular Formula | C19H23CL2N3O |
| Molecular Weight | 380.311422586441 |
| Exact Mass | 379.121 |
| CAS # | 1639220-19-1 |
| PubChem CID | 101910737 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 5.1 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 25 |
| Complexity | 387 |
| Defined Atom Stereocenter Count | 0 |
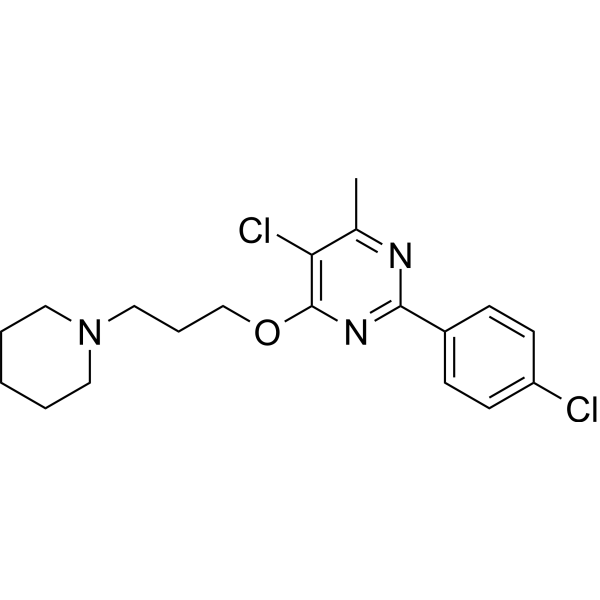
| SMILES | ClC1=C(C)N=C(C2C=CC(=CC=2)Cl)N=C1OCCCN1CCCCC1 |
| InChi Key | MSFUOAXHQLXDAO-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C19H23Cl2N3O/c1-14-17(21)19(25-13-5-12-24-10-3-2-4-11-24)23-18(22-14)15-6-8-16(20)9-7-15/h6-9H,2-5,10-13H2,1H3 |
| Chemical Name | 5-chloro-2-(4-chlorophenyl)-4-methyl-6-(3-piperidin-1-ylpropoxy)pyrimidine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | The σ1R receptor is highly bound to sigma1 receptor antagonist 1 (Ki = 1.06 nM), and it has good σ-1/2 selectivity (1344-fold) [1]. |
| ln Vitro |
The σ1R receptor is highly bound to sigma1 receptor antagonist 1 (Ki = 1.06 nM), and it has good σ-1/2 selectivity (1344-fold) [1]. 137 exhibited high binding affinity for the sigma-1 receptor (Ki σ₁ = 1.06 nM) and excellent selectivity over the sigma-2 receptor (Ki σ₂ = 1425 nM, selectivity σ₂/σ₁ = 1344-fold). [1] In a functional phenytoin-shift assay, 137 showed a Ki ratio (without/with phenytoin) of 0.87, confirming its antagonist profile at the sigma-1 receptor. [1] 137 showed weak affinity for the hERG potassium channel (IC₅₀ = 6.98 µM), indicating a low potential for cardiac toxicity. [1] In a broad selectivity panel against receptors and ion channels implicated in pain (e.g., μ-opioid, serotonin, NMDA, Nav1.7, TRPV1), 137 showed no significant affinity (% inhibition < 50% at 1 µM). [1] |
| ln Vivo |
Sigma?In both the rat CCI neuropathic pain model and the mouse formalin model, 1 receptor antagonist 1 exhibits dose-dependent analgesic effects [1]. In the mouse formalin test, pretreatment with 137 (80 mg/kg, ip) significantly reduced pain responses in both Phase I (licking time: 12.65 ± 5.07 s) and Phase II (licking time: 37.41 ± 8.89 s), with effects comparable to the reference sigma-1 antagonist S1RA. The antinociceptive effect was dose-dependent (ED₅₀ values: 48.36 ± 5.11 mg/kg for Phase I and 42.15 ± 3.96 mg/kg for Phase II). [1] In the rat chronic constriction injury (CCI) model of neuropathic pain, single and repeated oral administration of 137 (20, 40, 80 mg/kg) dose-dependently inhibited both mechanical allodynia (von Frey test) and thermal hyperalgesia (plantar test). The ED₅₀ values for single-dose treatment were 58.25 ± 6.04 mg/kg (mechanical) and 47.23 ± 3.87 mg/kg (thermal). [1] In the rotarod test, 137 at analgesic doses did not impair motor coordination in mice, unlike the positive control pregabalin. [1] |
| Enzyme Assay |
The sigma-1 receptor binding affinity of 137 was determined using guinea pig brain membranes. Membranes were incubated with the radioligand [³H]-(+)-pentazocine and various concentrations of the test compound (10⁻⁵ to 10⁻¹⁰ M) in Tris-HCl buffer (pH 8.0) at 25°C for 180 minutes. Nonspecific binding was defined using haloperidol (10 µM). The incubation was terminated by rapid vacuum filtration through GF/B filters, followed by washing and scintillation counting. Ki values were calculated using the Cheng-Prusoff equation. [1] The sigma-2 receptor binding assay was conducted similarly using guinea pig brain membranes incubated with [³H]-DTG in the presence of (+)-SKF-10047 (400 nM) to block sigma-1 sites. Incubation proceeded at 25°C for 120 minutes. Nonspecific binding was defined with DTG (10 µM). [1] |
| Cell Assay | The potential of 137 to block the hERG potassium channel was assessed using the whole-cell patch-clamp technique on HEK293 cells stably expressing hERG channels. Cells were voltage-clamped, and tail currents were measured at -50 mV following a depolarizing pulse to +50 mV. The effect of 137 was tested at concentrations of 0.3, 1, 3, and 10 µM. The IC₅₀ value was determined from the concentration-response curve of the tail current inhibition. [1] |
| Animal Protocol |
Formalin Test: Mice were administered 137 (dissolved in 30% PEG 400) or vehicle intraperitoneally (ip) 15 minutes before an intraplantar injection of 2.5% formalin solution (20 µL) into the hind paw. The time spent licking or biting the injected paw was recorded during Phase I (0-5 min) and Phase II (15-45 min) post-injection. [1] Chronic Constriction Injury (CCI) Model: Under anesthesia, the right sciatic nerve of rats was exposed and loosely ligated with four silk sutures. Sham-operated rats underwent exposure without ligation. Starting on day 14 post-surgery, rats were orally administered 137 (suspended in 0.5% methylcellulose) or vehicle twice daily for 4 consecutive days. Mechanical allodynia was assessed using von Frey filaments (up-down method), and thermal hyperalgesia was assessed using a plantar test apparatus (radiant heat) on day 15 (single dose) and day 18 (repeated dose). [1] Rotarod Test: Mice were trained to stay on a rotating rod (10 rpm). After ip administration of 137 or vehicle, the latency to fall from the rod was recorded at 30, 60, 90, and 120 minutes. [1] Acute Toxicity Study: Mice were orally administered a single dose of 137 (suspended in 0.5% methylcellulose) at doses of 200, 500, 1000, 1500, and 2000 mg/kg. Mortality was recorded over 24 hours, and the LD₅₀ was calculated. [1] |
| ADME/Pharmacokinetics |
In rats following a single intravenous (iv) dose (16 mg/kg), 137 had a half-life (t₁/₂) of 10.57 hours and an AUC₀‑∞ of 3686.17 ng·h/mL. [1] Following a single oral (po) dose (320 mg/kg), 137 had a t₁/₂ of 3.81 hours, a Tmax of 0.5 hours, a Cmax of 17394.90 ng/mL, and an AUC₀‑∞ of 55393.45 ng·h/mL. [1] The oral bioavailability (F) of 137 in rats was calculated to be 75.1%. [1] |
| Toxicity/Toxicokinetics |
The median lethal dose (LD₅₀) of 137 in mice following oral administration was greater than 2000 mg/kg. [1] No treatment-related adverse effects or motor impairments were observed in behavioral tests at efficacious doses. [1] |
| References |
[1]. Synthesis and biological evaluation of novel sigma-1 receptor antagonists based on pyrimidine scaffold as agents for treating neuropathic pain. J Med Chem. 2014 Dec 26;57(24):10404-23. |
| Additional Infomation |
137 (5-Chloro-2-(4-chlorophenyl)-4-methyl-6-(3-(piperidin-1-yl)propoxy)pyrimidine) is a novel, potent, and selective sigma-1 receptor antagonist identified from a pyrimidine-based scaffold. [1] It is proposed as a potential therapeutic agent for the treatment of neuropathic pain, based on its efficacy in preclinical pain models, favorable pharmacokinetic profile, and good safety margin. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~6.25 mg/mL (~16.43 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6294 mL | 13.1472 mL | 26.2943 mL | |
| 5 mM | 0.5259 mL | 2.6294 mL | 5.2589 mL | |
| 10 mM | 0.2629 mL | 1.3147 mL | 2.6294 mL |