Sch-42495, the orally bioactive ethylester prodrug of SCH 42354, is a novel and potent inhibitor of neutral metalloendopeptidase (NEP). Neutral metalloendopeptidase (NEP) inhibitors delay atrial natriuretic factor (ANF) catabolism and potentiate biological responses to ANF. SCH 42354 selectively inhibited hydrolysis of leu-enkephalin and ANF (IC50 of 8.3 and 10.0 nmol/L, respectively) in vitro. Plasma levels of exogenous ANF were augmented and ANF clearance from plasma was delayed by oral SCH 42495 (3 to 30 mg/kg) in normotensive rats. Plasma ANF levels in volume expanded rats were higher in SCH 42495-treated rats. Diuretic and natriuretic effects of ANF were increased in rats treated with SCH 42495. Oral doses of 1, 3, or 10 mg/kg of SCH 42495 produced significant reductions in blood pressure in DOCA-Na hypertensive rats of 22 +/- 6, 43 +/- 7, and 62 +/- 12 mm Hg, respectively, which were not associated with increases in heart rate. These doses did not alter urine flow, salt excretion, or plasma ANF. SCH 42495 produced significant elevation of urinary excretion of ANF and cGMP. In Dahl-S hypertensive rats, SCH 42495 (1 to 10 mg/kg orally) produced falls in blood pressure of a magnitude similar to that observed in DOCA-Na hypertensive rats. Significant hypotensive activity was observed 18 h after a single 10 mg/kg oral dose in Dahl-S hypertensive rats. In DOCA-Na hypertensive rats, a single dose of SCH 42495 significantly decreased cardiac output and did not lower systemic vascular resistance, a profile similar to that of ANF. The hypotensive response to SCH 42495 was not ascribable to ACE inhibition. Pithed rat preparations revealed no interaction of the drug with autonomic cardiovascular function. The antihypertensive effect of SCH 42495 likely results from potentiation of endogenous ANF via NEP inhibition.
Physicochemical Properties
| Molecular Formula | C20H29NO4S2 |
| Molecular Weight | 411.57856 |
| Exact Mass | 411.154 |
| CAS # | 136511-43-8 |
| Related CAS # | SCH 42495 racemate;145841-10-7;(R)-SCH 42495;156792-25-5 |
| PubChem CID | 131965 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 3.625 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 13 |
| Heavy Atom Count | 27 |
| Complexity | 487 |
| Defined Atom Stereocenter Count | 2 |
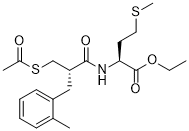
| SMILES | CSCCC(NC(C(CC1=CC=CC=C1C)CSC(=O)C)=O)C(OCC)=O |
| InChi Key | ZVQXPUMRSJGLSF-MSOLQXFVSA-N |
| InChi Code | InChI=1S/C20H29NO4S2/c1-5-25-20(24)18(10-11-26-4)21-19(23)17(13-27-15(3)22)12-16-9-7-6-8-14(16)2/h6-9,17-18H,5,10-13H2,1-4H3,(H,21,23)/t17-,18+/m1/s1 |
| Chemical Name | N-(2(S)-(Acetylthiomethyl)-3-(2-methylphenyl)-1-oxopropyl)-L-methionine ethyl ester |
| Synonyms | Sch 42495; Sch-42495; Sch42495. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Leucine enkephalin and ANF hydrolysis are specifically inhibited by SCH 42354 in vitro (IC50: 8.3 and 10.0 nM, respectively) [1]. |
| ln Vivo | After 10 days, SCH 42495 (30 mg/kg; oral gavage; twice daily) dramatically decreased ventricular hypertrophy and pulmonary vascular remodeling in hypoxic rats [2]. Rats treated with SCH 42495 (30 mg/kg; oral gavage; twice daily) have decreased cardiovascular remodeling as a result of chronic hypoxia [2]. SCH 42495 (oral dosages of 1, 3 or 10 mg/kg) significantly decreased blood pressure in DOCA-N hypertensive rats by 22±6, 43±7 and 62±12 mm Hg correspondingly [1]. |
| Animal Protocol |
Animal/Disease Models: hypoxic rats [2] Doses: 30 mg/kg Route of Administration: po (oral gavage); twice a day for 10 days Experimental Results: Caused a significant reduction in pulmonary vascular remodeling and ventricular hypertrophy. Resulting in diminished cardiovascular remodeling secondary to chronic hypoxia. |
| References |
[1]. Atrial natriuretic factor potentiating and hemodynamic effects of SCH 42495, a new, neutral metalloendopeptidase inhibitor. Am J Hypertens. 1993 May;6(5 Pt 1):357-68. [2]. Effects of the neutral endopeptidase inhibitor, SCH 42495, on the cardiovascular remodelling secondary to chronic hypoxia in rats. Clin Sci (Lond). 1994 Jul;87(1):109-14. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4297 mL | 12.1483 mL | 24.2966 mL | |
| 5 mM | 0.4859 mL | 2.4297 mL | 4.8593 mL | |
| 10 mM | 0.2430 mL | 1.2148 mL | 2.4297 mL |