Physicochemical Properties
| Molecular Formula | C44H71N13O13 |
| Molecular Weight | 990.113849878311 |
| Exact Mass | 989.529 |
| CAS # | 39698-78-7 |
| Related CAS # | Saralasin TFA;Saralasin;34273-10-4 |
| PubChem CID | 11954381 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 14 |
| Hydrogen Bond Acceptor Count | 16 |
| Rotatable Bond Count | 25 |
| Heavy Atom Count | 70 |
| Complexity | 1700 |
| Defined Atom Stereocenter Count | 7 |
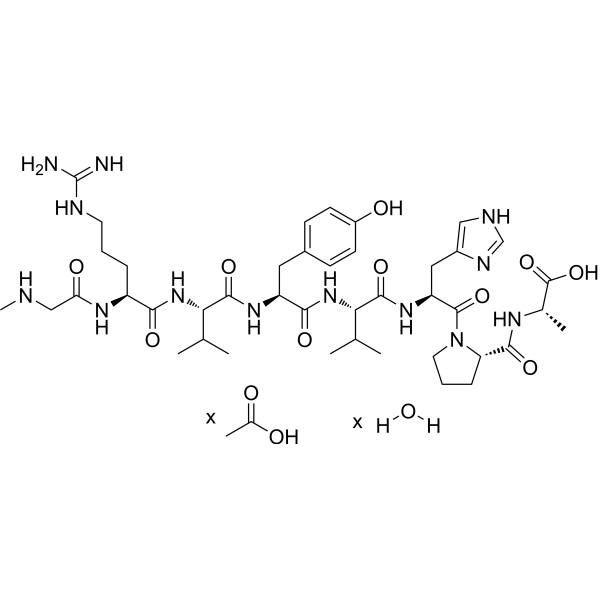
| SMILES | C[C@@H](C(=O)O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CC2=CN=CN2)NC(=O)[C@H](C(C)C)NC(=O)[C@H](CC3=CC=C(C=C3)O)NC(=O)[C@H](C(C)C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)CNC.CC(=O)O.O |
| InChi Key | YBZYNINTWCLDQA-UHKVWXOHSA-N |
| InChi Code | InChI=1S/C42H65N13O10.C2H4O2.H2O/c1-22(2)33(53-35(58)28(50-32(57)20-45-6)9-7-15-47-42(43)44)38(61)51-29(17-25-11-13-27(56)14-12-25)36(59)54-34(23(3)4)39(62)52-30(18-26-19-46-21-48-26)40(63)55-16-8-10-31(55)37(60)49-24(5)41(64)65;1-2(3)4;/h11-14,19,21-24,28-31,33-34,45,56H,7-10,15-18,20H2,1-6H3,(H,46,48)(H,49,60)(H,50,57)(H,51,61)(H,52,62)(H,53,58)(H,54,59)(H,64,65)(H4,43,44,47);1H3,(H,3,4);1H2/t24-,28-,29-,30-,31-,33-,34-;;/m0../s1 |
| Chemical Name | acetic acid;(2S)-2-[[(2S)-1-[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-5-(diaminomethylideneamino)-2-[[2-(methylamino)acetyl]amino]pentanoyl]amino]-3-methylbutanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-3-methylbutanoyl]amino]-3-(1H-imidazol-5-yl)propanoyl]pyrrolidine-2-carbonyl]amino]propanoic acid;hydrate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Cell proliferation of 3T3 and SV3T3 cells is inhibited by saralisin acetate hydrate (1 nM, 48 or 72 hours) [1]. Ito (a transient outward K+ current in rapidly inactivating mouse ventricles) and slow IK (a transient outward K+ current in slowly inactivating mouse ventricles) and Control levels in muscle cells are promptly restored by saralazine acetate hydrate (5 μM, 2 hours)[2]. The binding of FITC-Ang II to a rat liver membrane preparation (used as a source of angiotensin receptors) is inhibited by saralasin acetate hydrate (0.1–10 nM, 40 min), with a Ki value of 0.32 for 74% of the binding sites nM and a Ki value of 2.7 nM for the remaining binding sites [3]. When compared to a control group, prostaglandin E2 and 6-keto prostaglandin F1α levels are decreased and the ovulation rate is inhibited by 1 μM of salamelan acetate hydrate perfused in rat ovaries in vitro [4]. |
| ln Vivo | Saracin acetate hydrate (intravenous injection, 5-50 μg/kg, single dosage) reduces oxidative stress and tissue damage in cerulein-induced pancreatitis [5]. Saralasin acetate hydrate (subcutaneous injection, 10 and 30 mg/kg, single dose) increases serum renin activity (SRA) in normal, awake rats without significantly affecting blood pressure or heart rate [6]. |
| Cell Assay |
Cell proliferation assay [1] Cell Types: 3T3 and SV3T3 Cell Tested Concentrations: 1 nM Incubation Duration: 48 hrs (hours), 72 hrs (hours) Experimental Results: Inhibited cell growth of 3T3 and SV3T3 cells and caused an increase in cellular renin concentration. |
| Animal Protocol |
Animal/Disease Models: Cerulein-induced acute pancreatitis rat model [5] Doses: 5, 10, 20, 50 μg/kg, single dose. Mode of Route of Administration: intravenous (iv) (iv)injection. Experimental Results: The morphological characteristics of the pancreas returned to control levels. Reduces pancreatic damage and inhibits azure-induced glutathione depletion. Animal/Disease Models: Male SD (SD (Sprague-Dawley)) rat [6] Doses: 10 and 30 mg/kg, single dose. Route of Administration: subcutaneous injection Experimental Results: Stimulates renin release without changing blood pressure or heart rate when serum renin levels are measured 20 minutes after injection. |
| References |
[1]. Effects of angiotensin II and angiotensin II antagonist saralasin on cell growth and renin in 3T3 and SV3T3 cells. J Cell Physiol. 1979 Mar;98(3):503-13. [2]. Pressure-overload-induced angiotensin-mediated early remodeling in mouse heart. PLoS One. 2017 May 2;12(5):e0176713. [3]. A non-radioactive method for angiotensin II receptor binding studies using the rat liver. J Pharmacol Toxicol Methods. 2006 May-Jun;53(3):206-14. [4]. Saralasin-induced inhibition of ovulation in the in vitro perfused rat ovary is not replicated by the angiotensin II type-2 receptor antagonist PD123319. Am J Obstet Gynecol. 1998 Jul;179(1):35-40. [5]. Saralasin, a nonspecific angiotensin II receptor antagonist, attenuates oxidative stress and tissue injury in cerulein-induced acute pancreatitis. Pancreas. 2003 Apr;26(3):224-9. 6/. [6]. Saralasin-induced renin release: its blockade by prostaglandin synthesis inhibitors in the conscious rat. Hypertension. 1979;1(6):637‐642. |
| Additional Infomation |
An octapeptide analog of angiotensin II (bovine) with amino acids 1 and 8 replaced with sarcosine and alanine, respectively. It is a highly specific competitive inhibitor of angiotensin II that is used in the diagnosis of HYPERTENSION. See also: Saralasin Acetate (annotation moved to). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.0100 mL | 5.0499 mL | 10.0999 mL | |
| 5 mM | 0.2020 mL | 1.0100 mL | 2.0200 mL | |
| 10 mM | 0.1010 mL | 0.5050 mL | 1.0100 mL |