SU-5408, formerly known as VEGFR2 Kinase Inhibitor I, is a potent, cell-permeable inhibitor of mouse VEGFR2 kinase (IC50 = 70 nM)
Physicochemical Properties
| Molecular Formula | C18H18N2O3 |
| Molecular Weight | 310.34712 |
| Exact Mass | 310.132 |
| Elemental Analysis | C, 69.66; H, 5.85; N, 9.03; O, 15.47 |
| CAS # | 15966-93-5 |
| Related CAS # | 15966-93-5 |
| PubChem CID | 6419834 |
| Appearance | Yellow to orange solid powder |
| LogP | 3.438 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 23 |
| Complexity | 516 |
| Defined Atom Stereocenter Count | 0 |
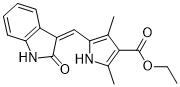
| SMILES | O=C(C1=C(C)NC(/C=C2C(NC3=C\2C=CC=C3)=O)=C1C)OCC |
| InChi Key | PMUJUSJUVIXDQC-LCYFTJDESA-N |
| InChi Code | InChI=1S/C18H18N2O3/c1-4-23-18(22)16-10(2)15(19-11(16)3)9-13-12-7-5-6-8-14(12)20-17(13)21/h5-9,19H,4H2,1-3H3,(H,20,21)/b13-9- |
| Chemical Name | ethyl 2,4-dimethyl-5-[(Z)-(2-oxo-1H-indol-3-ylidene)methyl]-1H-pyrrole-3-carboxylate |
| Synonyms | SU5408; SU 5408; VEGF Receptor 2 Kinase Inhibitor I; 15966-93-5; VEGFR2 Kinase Inhibitor I; SU5408; Vegfr 2 kinase inhibitor I; Vegfr-2 inhi; CHEMBL86943; ethyl 2,4-dimethyl-5-[(Z)-(2-oxo-1H-indol-3-ylidene)methyl]-1H-pyrrole-3-carboxylate; SU-5408; VEGFR2 Kinase Inhibitor I |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | VEGFR2 (IC50 = 70 nM) |
| ln Vitro | 3-substituted dihydrogen derivatives (indolin-2-ones)are a novel class of tyrosine coupling agents that exhibit selectivity towards distinct tyrosine coupling agents (RTKs). According to evaluations, SU5408 (VEGFR2 Multiplier I) is the most effective and selective VEGFR2 multiplier among these substances. The receptors for fold-inducing growth factors, epidermal growth factors, and insulin-like growth factors (IC50: >100 μM) are not significantly affected by SU5408 (VEGFR2 Multiplier I) [1]. |
| Enzyme Assay | Structure−activity relationships (SAR) for inhibition of ligand-dependent tyrosine phosphorylation by the indolin-2-ones were determined in a panel of NIH3T3 mouse fibroblast lines engineered to overexpress various RTKs including the human PDGF receptor β, the murine VEGF receptor (Flk-1), the human EGF receptor, and the human insulin-like growth factor-1 (IGF-1) receptor. To assess homology to the EGF receptor family-2 (Her-2) (p185erbB2) kinase activity, the cytoplasmic domain of the human Her-2 was engineered to contain the extracellular and transmembrane domain of the human EGF receptor. The chimeric receptor was overexpressed in NIH3T3 cells, and ligand-dependent Her-2 kinase activity was assessed following stimulation of the receptor with EGF as described.19 IC50 values were defined as the concentration of a compound required to achieve 50% inhibition of tyrosine phosphorylation on various RTKs compared to ligand-stimulated control reactions in the presence of vehicle alone (dimethyl sulfoxide). Compounds with IC50 values greater than 100 μΜ were considered inactive. The relative selectivity of a given inhibitor was assessed using calculated ratios of the IC50 values corresponding to specific RTKs. [1] |
| Cell Assay |
Effects of Zeaxanthin on GBM can be blocked by VEGFR2 kinase inhibitor SU5408. U87, U251, HEB and HUVEC cells in logarithmic growth phase were inoculated in 96-well plates at a density of 9 × 104 cells/mL and incubated overnight until reaching 90% confluence. Subsequently, various concentrations (0, 5, 10, 20, 40, 80 μM) of Zea were treated, and 5 replicate wells established for each concentration. The cells were then incubated for further culture. Cell viability was measured at 24 or 48 h post-treatment with Zea using the MTT assay. The morphology of cells after Zea treatment for 24 h were photographed by a microscope (IX73, Olympus Corporation, Tokyo, Japan). For the SU5408 blocking assay, SU5408, a classical VEGFR2 kinase inhibitor, was employed. U251 cells were pretreated with or without 100 nM SU5408 prior different concentrations of Zea (0, 10, 20, 40 μM) for 48 h, then the cell viability was detected by MTT assay. To test SU5408's effects on GBM cells only, U87 and U251 cells were treated with different doses of SU5408 (0, 10, 20, 40, 80, 160 nM) for 24 or 48 h, followed by cell viability assessment using the MTT assay[2]. |
| References |
[1]. Synthesis and biological evaluations of 3-substituted indolin-2-ones: a novel class of tyrosine kinase inhibitors that exhibit selectivity towardparticular receptor tyrosine kinases. J Med Chem. 1998 Jul 2;41(14):2588-603. |
| Additional Infomation |
In summary of the SAR studies: (1) Proton at the N-1 position of the indolin-2-ones is essential for the inhibitory activity against the PDGF and VEGF (Flk-1) RTKs. (2) Vinyl proton is critical for 3-substituted indolin-2-ones to inhibit the PDGF and VEGF (Flk-1) RTKs. (3) Z Isomeric form is required for the inhibitory activity against the PDGF and VEGF (Flk-1) RTKs. (4) 3-[(Substituted pyrrolyl)methylidenyl]indolin-2-ones are the most potent and selective inhibitors of the PDGF and VEGF (Flk-1) RTKs. (5) Electron-donating substitution is preferred for the inhibitory potency of these 3-substituted indolin-2-ones against the PDGF and VEGF (Flk-1) RTKs. (6) Bulky lipophilic groups in the phenyl ring at the C-3 position of 3-(substituted benzylidenyl)indolin-2-ones are the key determinants for the selective inhibitory activity of these inhibitors against the EGF and Her-2 RTKs. In conclusion, we described a chemical series that shows promise in the development of RTK inhibitors with the capacity to be specific to particular enzymes. PTK function has been associated with a wide variety of human diseases including cancers, metastasis, arterial restenosis, various inflammatory diseases including psoriasis and rheumatoid arthritis, pulmonary fibrosis, liver cirrhosis, kidney sclerosis, and other diseases. The potential to design and synthesize 3-substituted indolin-2-ones with inhibitory properties for various PTK subtypes supports the use of compounds of this class to treat a wide variety of human diseases.[1] |
Solubility Data
| Solubility (In Vitro) | DMSO: ~6 mg/mL (~19.3 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 0.77 mg/mL (2.48 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 7.7 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 0.77 mg/mL (2.48 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 7.7 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2222 mL | 16.1108 mL | 32.2217 mL | |
| 5 mM | 0.6444 mL | 3.2222 mL | 6.4443 mL | |
| 10 mM | 0.3222 mL | 1.6111 mL | 3.2222 mL |