Physicochemical Properties
| Molecular Formula | C18H15N5O2 |
| Molecular Weight | 333.344002962112 |
| Exact Mass | 333.122574 |
| CAS # | 2762286-04-2 |
| PubChem CID | 168355665 |
| Appearance | Typically exists as Off-white to light yellow solids at room temperature |
| LogP | 1.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 25 |
| Complexity | 708 |
| Defined Atom Stereocenter Count | 0 |
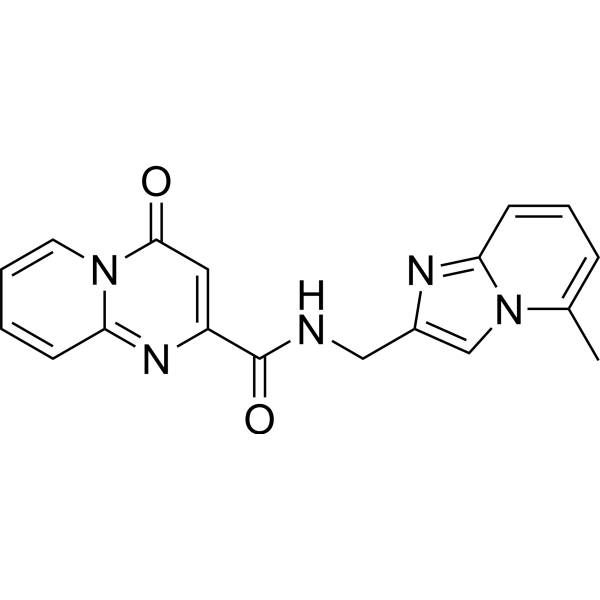
| SMILES | O=C(C1=CC(N2C=CC=CC2=N1)=O)NCC1=CN2C(C)=CC=CC2=N1 |
| InChi Key | BOTBQIXFHWFWHV-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H15N5O2/c1-12-5-4-7-15-20-13(11-23(12)15)10-19-18(25)14-9-17(24)22-8-3-2-6-16(22)21-14/h2-9,11H,10H2,1H3,(H,19,25) |
| Chemical Name | N-[(5-methylimidazo[1,2-a]pyridin-2-yl)methyl]-4-oxopyrido[1,2-a]pyrimidine-2-carboxamide |
| Synonyms | STM-2120; EX-A8309; DA-67811; STM2120; EXA8309; DA67811; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | IC50: 64.5 μM (METTL3-METTL14)[1] |
| ln Vitro | STM2120 (0.04-50 μM; 72 h) does not affect the proliferation of MOLM-13 cells. To investigate the therapeutic potential of targeting the enzymatic activity of METTL3 as an anti-leukaemic strategy, we developed the small molecule STM2457. A high throughput screen (HTS) of 250,000 diverse drug-like compounds was carried out. STM1760 [half maximum inhibitory concentration (IC50) = 51.7 μM] was one of only two non-S-adenosyl methionine (SAM) related hits arising from the HTS (Extended Data Fig. 1a, b). After optimisation of potency, in vitro ADME and in vivo pharmacokinetic properties, we identified STM2457 (Fig. 1a). STM2120 (IC50 = 64.5 μM) was identified as structurally related, but 1,000 fold less active compared to STM2457 (Fig. 1b and Extended Data Fig. 1a)[1]. |
| Enzyme Assay |
METTL3/14 RF/MS methyltransferase assay[1] The enzymatic assay was established to determine IC50 values for the inhibition of RNA methyltransferase activity. The enzyme used was full-length his-tagged METTL3 co-expressed with full length FLAG-tagged METTL14 in a baculovirus expression system. The enzyme complex was purified using standard affinity chromatography. Enzymatic reactions were performed at room temperature in 384-well plates using a final reaction volume of 20 μL containing 20 mM TrisCl pH 7.6, 1 mM DTT, 0.01% Tween-20. 5 nM final concentration of METTL3/14 was pre-incubated with various compound concentrations for 10 minutes, followed by addition of 0.2 μM final concentration synthetic RNA substrate (5’P-uacacucgaucuggacuaaagcugcuc-3’) and 0.5 μM final concentration S-adenosyl methionine (SAM). The reaction was incubated for further 60 minutes at room temperature, and then quenched by the addition of 40 μL 7.5% TCA with internal standard. After termination, plates were sealed, centrifuged and stored at 4°C until analysis. METTL3 activity was assessed using the RapidFire™ mass spectrometry (RF/MS) platform to measure the S-adenosyl homocysteine (SAH) product. Stopped and stable assay plates were analyzed on the Agilent RF300 integrated autosampler/solid-phase extraction (SPE) system coupled to an ABSciex 4000 mass spectrometer for the quantification of the SAH and normalized to the ratio of signal of two internal standards. The mass transition for the product (SAH) was 384.9/135.9 Da. Transitions of the internal standard were used for normalization of matrix effects.[2] |
| Cell Assay |
Drug and Proliferation Assays[1] All suspension cells were plated in 96-well plates in triplicate at 5,000–10,000 cells per well and treated for 72 hours with vehicle or the indicated concentrations of STM2457 and STM2120 (0.04-50 μM). On day 4, an equal volume for all wells was split using fresh media and compound, such that the resulting cell density in each well matched the initial seeding density. Plates were measured on day 6 using CellTiter 96 AQueous Non-Radioactive Cell Proliferation Assay in order to calculate the relative cell proliferation. All the compounds were dissolved in DMSO. |
| References |
[1]. Small-molecule inhibition of METTL3 as a strategy against myeloid leukaemia. Nature. 2021 May;593(7860):597-601. |
| Additional Infomation | N6-methyladenosine (m6A) is an abundant internal RNA modification1,2 that is catalysed predominantly by the METTL3-METTL14 methyltransferase complex3,4. The m6A methyltransferase METTL3 has been linked to the initiation and maintenance of acute myeloid leukaemia (AML), but the potential of therapeutic applications targeting this enzyme remains unknown5-7. Here we present the identification and characterization of STM2457, a highly potent and selective first-in-class catalytic inhibitor of METTL3, and a crystal structure of STM2457 in complex with METTL3-METTL14. Treatment of tumours with STM2457 leads to reduced AML growth and an increase in differentiation and apoptosis. These cellular effects are accompanied by selective reduction of m6A levels on known leukaemogenic mRNAs and a decrease in their expression consistent with a translational defect. We demonstrate that pharmacological inhibition of METTL3 in vivo leads to impaired engraftment and prolonged survival in various mouse models of AML, specifically targeting key stem cell subpopulations of AML. Collectively, these results reveal the inhibition of METTL3 as a potential therapeutic strategy against AML, and provide proof of concept that the targeting of RNA-modifying enzymes represents a promising avenue for anticancer therapy.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9999 mL | 14.9997 mL | 29.9994 mL | |
| 5 mM | 0.6000 mL | 2.9999 mL | 5.9999 mL | |
| 10 mM | 0.3000 mL | 1.5000 mL | 2.9999 mL |