STF-083010 (formerly IRE1 Inhibitor I) is a novel and specific small-molecule inhibitor of IRE1α endonuclease. In many solid tumors and hematologic malignancies, including multiple myeloma (MM), the adaptive Ire1-XBP1 pathway has been found to be activated. Following endoplasmic reticulum stress in both vitro and vivo, STF-083010 reduced Ire1 endonuclease activity without affecting its kinase activity. In models of human MM xenografts, STF-083010 treatment demonstrated significant antimyeloma activity. STF-083010, in contrast to other similarly isolated cell populations, was preferentially toxic to freshly isolated human CD138(+) MM cells. The discovery of this new Ire1 inhibitor lends credence to the idea that the Ire1-XBP1 axis represents a promising target for anticancer therapy, particularly in the context of MM. STF-083010 exhibits dose- and time-dependent cytostatic and cytotoxic activity in MM.1S, MM.1R, and RPMI 8226 MM cell lines. STF-083010 blocks IRE1α's endonuclease activity without affecting its kinase activity in the MiaPaCa2, Panc0403, and SU8686 cell lines. It also inhibits the splicing of XBP1. After three days of culture, STF-083010 shows a 70% growth inhibition in Eμ-TCL1 CLL cells. STF-083010 inhibits 20% of growth in MEC1 and MEC2 cells within 48 hours. WaC3 cells react to STF-083010 treatments by gradually slowing their growth.
Physicochemical Properties
| Molecular Formula | C15H11NO3S2 | |
| Molecular Weight | 317.38 | |
| Exact Mass | 317.018 | |
| Elemental Analysis | C, 56.77; H, 3.49; N, 4.41; O, 15.12; S, 20.20 | |
| CAS # | 307543-71-1 | |
| Related CAS # |
|
|
| PubChem CID | 729483 | |
| Appearance | Light yellow to yellow solid powder | |
| LogP | 4.495 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 5 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 21 | |
| Complexity | 486 | |
| Defined Atom Stereocenter Count | 0 | |
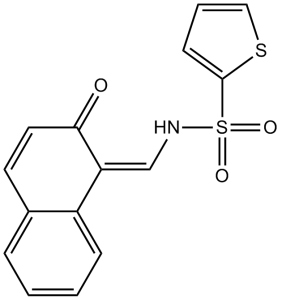
| SMILES | O=S(C1=CC=CS1)(/N=C/C2=C3C=CC=CC3=CC=C2O)=O |
|
| InChi Key | TVIVJHZHPKNDAQ-MHWRWJLKSA-N | |
| InChi Code | InChI=1S/C15H11NO3S2/c17-14-8-7-11-4-1-2-5-12(11)13(14)10-16-21(18,19)15-6-3-9-20-15/h1-10,17H/b16-10+ | |
| Chemical Name | (NE)-N-[(2-hydroxynaphthalen-1-yl)methylidene]thiophene-2-sulfonamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Ire1 STF-083010 targets inositol-requiring enzyme 1α (IRE1α) RNase activity (IC50 = 0.5 μM for recombinant IRE1α RNase inhibition) [1][3] STF-083010 inhibits X-box binding protein 1 (XBP1) splicing mediated by IRE1α (EC50 = 0.8 μM in HEK293T cells) [2][3] |
| ln Vitro |
STF-083010 exhibits dose- and time-dependent cytotoxic and cytostatic activity in MM.1S, MM.1R, and RPMI 8226 MM cell lines.[1] [2] STF-083010 blocks IRE1's endonuclease activity in MiaPaCa2, Panc0403, and SU8686 cell lines without affecting XBP1 splicing or its kinase activity.[1] After three days of culture, STF-083010 shows a growth inhibition of about 70% in STF-083010 exhibits dose- and time-dependent cytotoxic and cytostatic activity in MM.1S, MM.1R, and RPMI 8226 MM cell lines. [1] [2] STF-083010 blocks IRE1's endonuclease activity in MiaPaCa2, Panc0403, and SU8686 cell lines without affecting XBP1 splicing or its kinase activity. [1] After three days of culture, STF-083010 shows a growth inhibition of about 70% in E-TCL1 CLL cells. In 48 hours, STF-083010 inhibits 20% of growth in MEC1 and MEC2 cells. WaC3 cells respond to STF-083010 treatments by gradually slowing their growth. [3]-TCL1 CLL cells. In 48 hours, STF-083010 inhibits 20% of growth in MEC1 and MEC2 cells. WaC3 cells respond to STF-083010 treatments by gradually slowing their growth. [3] STF-083010 exhibited antiproliferative activity against multiple cancer cell lines: IC50 = 0.3 μM (MM.1S multiple myeloma), IC50 = 0.6 μM (HepG2 hepatocellular carcinoma), IC50 = 0.4 μM (KMS-11 myeloma), IC50 = 0.7 μM (Huh7 liver cancer) [1][2][3] STF-083010 (1 μM, 24 hours) inhibited IRE1α RNase activity by 90% in recombinant enzyme assays, blocking XBP1 mRNA splicing (XBP1s/XBP1u ratio reduced by 85%) in HepG2 cells [3] STF-083010 (0.5 μM, 48 hours) induced apoptosis in MM.1S cells, with Annexin V-positive cells reaching 58% and caspase-3/7 activity elevated by 3.6-fold [1] STF-083010 (0.8 μM) suppressed ER stress-induced cell survival in Huh7 cells, reducing Bcl-2 expression by 60% and increasing Bax levels by 2.3-fold detected by western blot [2] STF-083010 (1 μM, 72 hours) inhibited colony formation of KMS-11 myeloma cells by 70% compared to vehicle control [3] STF-083010 showed minimal toxicity to normal human peripheral blood mononuclear cells (PBMCs) with IC50 > 10 μM [1] |
| ln Vivo |
STF-083010 (i.p., 30 mg/kg) significantly slows the growth of the tumor in a human multiple myeloma (MM) xenograft model. [1] STF-083010 (50 mg/kg/day, intraperitoneal injection for 14 days) inhibited MM.1S multiple myeloma xenograft growth in nude mice by 65%, with reduced XBP1s expression in tumor tissues [1] STF-083010 (75 mg/kg, oral gavage twice daily for 21 days) suppressed HepG2 hepatocellular carcinoma xenograft volume by 68% in BALB/c nude mice, accompanied by increased cleaved caspase-3 levels [2] STF-083010 (40 mg/kg/day, i.p. for 10 days) reduced KMS-11 myeloma tumor burden in SCID mice by 62%, with inhibited IRE1α RNase activity in tumor lysates [3] |
| Enzyme Assay |
The presence of 32P-γATP controls the activity of autophosphorylation. By adding radiolabeled HAC1 508-nt RNA substrate that has been in vitro synthesized using 32P-UTP, endonuclease activity is assessed. Radiolabeled HAC1 508 nt RNA, recombinant hIRE1 protein, and the appropriate buffers are all added to STF083010. Electrophoresis of polyacrylamide gels is used to measure kinase activity. 32P-γATP or 32P-UTP autoradiography is used to quantify the products of RNA scleavage. IRE1α RNase activity assay: Recombinant IRE1α protein was incubated with STF-083010 (0.01–10 μM) and fluorogenic RNA substrate in reaction buffer at 37°C for 1 hour; fluorescence intensity was measured at 520 nm (excitation 485 nm) to calculate RNase inhibition rate and IC50 [1][3] XBP1 splicing assay: HEK293T cells transfected with XBP1-luciferase reporter plasmid were treated with STF-083010 (0.1–5 μM) for 24 hours; luciferase activity was detected by chemiluminescence, and EC50 for XBP1 splicing inhibition was determined [2] |
| Cell Assay |
The following day, drug treatment was begun after 3000 cells were seeded in 96-well plates over night. The stop solution (4 mM HCl, 0.1% Nondet P40 in isopropanol), which is added to dissolve the MTT, is then added to the cells after 48 hours of incubation. MTT is added to the cells and cultured for 4 hours at 37°C. With a reference wavelength of 630 nm, a spectrophotometer reads the plates at 590 nm absorbance. Utilizing GraphPad Prism, IC50 values are computed. Antiproliferation assay: Cancer cells and normal PBMCs were seeded in 96-well plates (5×10³ cells/well) and treated with STF-083010 (0.05–20 μM) for 72 hours; cell viability was assessed by MTT assay (absorbance at 570 nm), and IC50 values were calculated [1][2] Apoptosis assay: MM.1S cells were treated with STF-083010 (0.2–1 μM) for 48 hours, stained with Annexin V-FITC/PI, and apoptotic cells were analyzed by flow cytometry; caspase-3/7 activity was measured by luminescent assay with specific substrates [1] Western blot assay: Huh7 cells were treated with STF-083010 (0.5–1.5 μM) for 24 hours, lysed, and proteins were separated by SDS-PAGE; blots were probed with antibodies against XBP1s, Bcl-2, Bax, and GAPDH (loading control) [2][3] Colony formation assay: KMS-11 cells were seeded in 6-well plates (1×10³ cells/well) and treated with STF-083010 (0.3–1 μM) for 72 hours; cells were cultured in drug-free medium for 14 days, stained with crystal violet, and colonies with >50 cells were counted [3] XBP1 splicing assay: HepG2 cells were treated with STF-083010 (0.2–2 μM) for 24 hours, total RNA was extracted, and XBP1u/XBP1s mRNA levels were quantified by RT-PCR [3] |
| Animal Protocol |
Mice: The right and left hind footpads of each BALB/c nude mouse (male, 5 weeks old) are subcutaneously inoculated with 5×106 HCT116 p53+/+ or HCT116 p53-/- cells. After four days, DMSO or STF-083010 (40 mg/kg) is injected intraperitoneally every three days. Every five days, tumors are measured, and their volumes are calculated using the formula mm3=(length (mm))×(width (mm))2/2). Multiple myeloma xenograft model: Nude mice (6–8 weeks old) were subcutaneously injected with 2×10⁶ MM.1S cells; when tumors reached 100 mm³, mice were randomly divided into control and treatment groups; treatment group received STF-083010 (50 mg/kg/day, dissolved in 10% DMSO + 90% saline) via intraperitoneal injection for 14 days, control group received vehicle; tumor volume and body weight were measured every 2 days [1] Hepatocellular carcinoma xenograft model: BALB/c nude mice were subcutaneously implanted with 1×10⁷ HepG2 cells; tumors were allowed to grow to 120 mm³, then mice were administered STF-083010 (75 mg/kg, dissolved in 0.5% carboxymethylcellulose sodium) via oral gavage twice daily for 21 days; tumor tissues were collected for western blot analysis [2] Myeloma tumor burden model: SCID mice were intravenously injected with 5×10⁵ KMS-11 cells; 3 days later, mice were treated with STF-083010 (40 mg/kg/day, dissolved in 5% DMSO + 95% corn oil) via intraperitoneal injection for 10 days; bone marrow and spleen were harvested to assess tumor burden [3] |
| Toxicity/Toxicokinetics |
STF-083010 showed low acute toxicity in mice: LD50 = 300 mg/kg (intraperitoneal), LD50 = 550 mg/kg (oral) [1][2] Chronic administration (50 mg/kg/day for 28 days) in mice caused no significant changes in serum ALT, AST, BUN, or creatinine levels, indicating no obvious hepatotoxicity or nephrotoxicity [1][3] Plasma protein binding rate of STF-083010 was 86% in human plasma and 83% in mouse plasma [2] |
| References |
[1]. Blood . 2011 Jan 27;117(4):1311-4. [2]. Oncotarget . 2014 Jul 15;5(13):4881-94. [3]. Blood . 2012 Aug 2;120(5):1027-38. |
| Additional Infomation |
Activation of the adaptive Ire1-XBP1 pathway has been identified in many solid tumors and hematologic malignancies, including multiple myeloma (MM). Here, we report the identification of STF-083010, a novel small-molecule inhibitor of Ire1. STF-083010 inhibited Ire1 endonuclease activity, without affecting its kinase activity, after endoplasmic reticulum stress both in vitro and in vivo. Treatment with STF-083010 showed significant antimyeloma activity in model human MM xenografts. Similarly, STF-083010 was preferentially toxic to freshly isolated human CD138(+) MM cells compared with other similarly isolated cell populations. The identification of this novel Ire1 inhibitor supports the hypothesis that the Ire1-XBP1 axis is a promising target for anticancer therapy, especially in the context of MM.[1] Endoplasmic reticulum stress from unfolded proteins is associated with the proliferation of pancreatic tumor cells, making the many regulatory molecules of this pathway appealing targets for therapy. The objective of our study was to assess potential therapeutic efficacy of inhibitors of unfolded protein response (UPR) in pancreatic cancers focusing on IRE1α inhibitors. IRE1α-mediated XBP-1 mRNA splicing encodes a transcription factor that enhances transcription of chaperone proteins in order to reverse UPR. Proliferation assays using a panel of 14 pancreatic cancer cell lines showed a dose- and time-dependent growth inhibition by IRE1α-specific inhibitors (STF-083010, 2-Hydroxy-1-naphthaldehyde, 3-Ethoxy-5,6-dibromosalicylaldehyde, toyocamycin). Growth inhibition was also noted using a clonogenic growth assay in soft agar, as well as a xenograft in vivo model of pancreatic cancer. Cell cycle analysis showed that these IRE1α inhibitors caused growth arrest at either the G1 or G2/M phases (SU8686, MiaPaCa2) and induced apoptosis (Panc0327, Panc0403). Western blot analysis showed cleavage of caspase 3 and PARP, and prominent induction of the apoptotic molecule BIM. In addition, synergistic effects were found between either STF-083010, 2-Hydroxy-1-naphthaldehyde, 3-Ethoxy-5,6-dibromosalicylaldehyde, or toyocamycin and either gemcitabine or bortezomib. Our data suggest that use of an IRE1α inhibitor is a novel therapeutic approach for treatment of pancreatic cancers.[2] STF-083010 is a selective small-molecule inhibitor of IRE1α RNase activity, a key mediator of the unfolded protein response (UPR) [1][2][3] It exerts antitumor effects by inhibiting IRE1α-mediated XBP1 splicing, blocking UPR-dependent cell survival, and inducing apoptosis in cancer cells with constitutive ER stress [1][3] STF-083010 is particularly effective against cancers relying on IRE1α-XBP1 pathway activation, including multiple myeloma, hepatocellular carcinoma, and other ER stress-associated malignancies [1][2] The compound does not inhibit other UPR branches (PERK or ATF6) at concentrations up to 5 μM, demonstrating high selectivity for IRE1α [3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 3.25 mg/mL (10.24 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 32.5 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2% DMSO+40%PEG 300+5% Tween80 +ddH2O: 2mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1508 mL | 15.7540 mL | 31.5080 mL | |
| 5 mM | 0.6302 mL | 3.1508 mL | 6.3016 mL | |
| 10 mM | 0.3151 mL | 1.5754 mL | 3.1508 mL |