Physicochemical Properties
| Molecular Formula | C18H21CLF3N3O |
| Molecular Weight | 387.827053785324 |
| Exact Mass | 387.132 |
| Elemental Analysis | C, 55.75; H, 5.46; Cl, 9.14; F, 14.70; N, 10.83; O, 4.13 |
| CAS # | 2319790-02-6 |
| Related CAS # | 2319790-02-6 |
| PubChem CID | 131839612 |
| Appearance | White to off-white solid powder |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 26 |
| Complexity | 407 |
| Defined Atom Stereocenter Count | 0 |
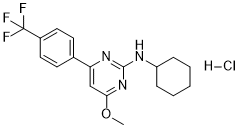
| SMILES | Cl.FC(C1C=CC(=CC=1)C1=CC(=NC(=N1)NC1CCCCC1)OC)(F)F |
| InChi Key | YXQSTPASDICGCA-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H20F3N3O.ClH/c1-25-16-11-15(12-7-9-13(10-8-12)18(19,20)21)23-17(24-16)22-14-5-3-2-4-6-14;/h7-11,14H,2-6H2,1H3,(H,22,23,24);1H |
| Chemical Name | C18H21ClF3N3OMolecular Weight |
| Synonyms | SSD114 HCl SSD114 SSD-114 SSD 114 SSD114 hydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
The target of SSD114 HCl is the γ-aminobutyric acid type B (GABAB) receptor, and it acts as a positive allosteric modulator (PAM) of this receptor. - In cAMP inhibition assay using CHO cells expressing human GABAB1b/GABAB2 receptors (in the presence of 1 μM GABA, a submaximal concentration): EC₅₀ = 0.32 μM [1] - In [³⁵S]GTPγS binding assay using rat brain membranes (in the presence of 1 μM GABA): EC₅₀ = 0.45 μM [1] - No significant binding to other receptors (e.g., GABA-A, 5-HT₁A, D₂, M₁ receptors): Ki > 10 μM for all tested non-target receptors [1] |
| ln Vitro |
The stimulation of [35S]GTPγS caused by GABA alone was greatly amplified (about 170% above basal levels) by 25 µM SSD114 hydrochloride in the presence of 10 µM GABA. The GABA concentration-response curve shifted leftward with the addition of 15 and 30 µM SSD114 hydrochloride, and the maximum GABA stimulation at the highest dose increased slightly. The EC50 of GABA was reduced by two and 2.5 times, respectively, in the presence of 15 µM and 30 µM SSD114 hydrochloride; however, the maximum stimulation (Emax) was only increased at 30 µM concentration, reaching a value of 161±5.09% of the basal value [1]. 1. Enhancement of GABAB receptor-mediated cAMP inhibition in CHO cells: CHO cells stably expressing human GABAB1b/GABAB2 receptors were treated with SSD114 HCl (0.01–10 μM) in the presence of 1 μM GABA (a submaximal agonist concentration). SSD114 HCl dose-dependently inhibited forskolin-induced cAMP production: 10 μM SSD114 HCl achieved an 85% inhibition rate, and the EC₅₀ was 0.32 μM. No cAMP inhibition was observed when SSD114 HCl was used alone (without GABA) [1] 2. Augmentation of [³⁵S]GTPγS binding to GABAB receptors in rat brain membranes: Rat brain membrane homogenates were incubated with SSD114 HCl (0.01–10 μM) and 1 μM GABA. SSD114 HCl dose-dependently increased [³⁵S]GTPγS binding (a marker of G protein activation): the maximum binding level was 60% higher than that in the GABA-only group, with an EC₅₀ of 0.45 μM. No significant [³⁵S]GTPγS binding was induced by SSD114 HCl alone [1] 3. Inhibition of excitatory synaptic transmission in rat hippocampal neurons: Primary rat hippocampal neurons (cultured for 14–21 days) were used for whole-cell patch-clamp recording. SSD114 HCl (0.1–10 μM) dose-dependently reduced the amplitude of excitatory postsynaptic currents (EPSCs) recorded from pyramidal neurons: 10 μM SSD114 HCl inhibited EPSC amplitude by 55%. This inhibitory effect was completely reversed by the GABAB receptor antagonist SCH50911 (10 μM), confirming it was mediated by GABAB receptors [1] |
| ln Vivo |
Pretreatment with SSD114 hydrochloride has been shown to decrease the incidence of loss of righting reflex (LORR) [F(5,30)=4.55, P<0.005]. A post hoc analysis revealed a significant difference in the incidence of LORR between the mice treated with vehicle and the group pretreated with SSD114 hydrochloride at a level of at least 10 mg/kg. Longer LORR durations can be achieved by pretreating with SSD114 hydrochloride [F(5,30)=4.81, P<0.005]. The duration of LORR in the mouse groups pretreated with 10 and 100 mg/kg SSD114 hydrochloride was shown to be considerably longer than in the mice treated with vehicle, according to post hoc analysis [1]. 1. Anxiolytic effect in mice (elevated plus maze test): Male ICR mice (20–25 g, 6–8 weeks old) were orally administered SSD114 HCl (3, 10, 30 mg/kg). Sixty minutes after dosing, the mice were placed in an elevated plus maze (EPM, arms: 30 cm long, 50 cm high). The 30 mg/kg group showed a 45% increase in open-arm residence time (vs. vehicle control) and a 30% increase in open-arm entry frequency; total arm entries (a measure of locomotor activity) were unchanged, indicating no locomotor suppression [1] 2. Anticonvulsant effect in mice (pentylenetetrazol-induced seizures): Male ICR mice were intraperitoneally injected with SSD114 HCl (10, 30, 100 mg/kg). Thirty minutes later, pentylenetetrazol (PTZ, 85 mg/kg, i.p.) was administered to induce seizures. The 30 mg/kg group prolonged seizure latency from 2.5 minutes (vehicle) to 5.8 minutes; the 100 mg/kg group reduced seizure incidence from 100% (vehicle) to 40% and completely prevented mortality within 30 minutes [1] 3. Analgesic effect in mice (hot plate test): Female ICR mice were used (more sensitive to thermal pain). Baseline hot plate latency (55±0.5°C) was measured (excluding mice with latency <3 s or >30 s). After oral administration of SSD114 HCl (10, 30, 100 mg/kg), latency was measured at 30, 60, and 120 minutes. The 30 mg/kg group increased latency from 5.2 s (baseline) to 8.5 s (30 minutes post-dosing); the 100 mg/kg group extended latency to 12.1 s, and the effect persisted for 4 hours [1] |
| Enzyme Assay |
1. cAMP inhibition assay in GABAB-expressing CHO cells: CHO cells stably expressing human GABAB1b/GABAB2 receptors were seeded in 24-well plates and grown to 80% confluence. The medium was replaced with serum-free DMEM for 1-hour starvation. Then, the cells were incubated with SSD114 HCl (0.01–10 μM) + 1 μM GABA (or vehicle + GABA) for 30 minutes, followed by addition of forskolin (10 μM, a cAMP inducer) for 15 minutes. Intracellular cAMP was extracted with 0.1 M HCl and measured using a competitive ELISA kit. The inhibition rate was calculated relative to the forskolin-only group, and EC₅₀ was fitted using a four-parameter logistic model [1] 2. [³⁵S]GTPγS binding assay in rat brain membranes: Rat brains were homogenized in ice-cold Tris-HCl buffer (50 mM, pH 7.4, containing 3 mM MgCl₂, 0.2 mM GDP) and centrifuged (10,000×g, 10 minutes, 4°C) to prepare crude membranes. Membranes (50 μg protein/well) were incubated with SSD114 HCl (0.01–10 μM) + 1 μM GABA + [³⁵S]GTPγS (0.1 nM) in a total volume of 200 μL at 30°C for 60 minutes. Non-specific binding was measured in the presence of 100 μM GTPγS. The mixture was filtered through glass fiber filters (pre-soaked in 0.5% polyethyleneimine) and washed three times with ice-cold buffer. Radioactivity was counted using a liquid scintillation counter, and EC₅₀ was calculated based on the binding enhancement ratio [1] |
| Cell Assay |
Primary rat hippocampal neuron culture and electrophysiological recording: Hippocampi were dissected from E18–E19 rat embryos, digested with 0.25% trypsin for 15 minutes (37°C), and triturated into single-cell suspensions. Cells were plated on poly-L-lysine-coated coverslips at a density of 5×10⁴ cells/cm², and cultured in Neurobasal medium supplemented with 10% fetal bovine serum, 2 mM glutamine, and 2% B27. Cultures were maintained at 37°C in 5% CO₂ for 14–21 days. Whole-cell patch-clamp recordings were performed in voltage-clamp mode (holding potential: -70 mV). Excitatory postsynaptic currents (EPSCs) were evoked by stimulating Schaffer collaterals with a glass electrode (0.1 ms pulse, 0.1 Hz). After stable EPSC recording for 5 minutes, SSD114 HCl (0.1–10 μM) was added to the recording chamber, and EPSC amplitude was monitored for 10 minutes. Subsequently, SCH50911 (10 μM) was added to verify reversibility [1] |
| Animal Protocol |
1. Mouse elevated plus maze (EPM) assay: Male ICR mice (20–25 g) were acclimated to the laboratory (12 h light/dark cycle, 22±2°C) for 24 hours. SSD114 HCl was dissolved in 0.5% methylcellulose and administered orally at doses of 3, 10, 30 mg/kg (injection volume: 10 μL/g body weight); the control group received 0.5% methylcellulose. Sixty minutes after dosing, each mouse was placed in the center of the EPM (open arms: 5 cm wide; closed arms: 15 cm high walls) and video-recorded for 5 minutes. Parameters analyzed included: open-arm residence time (%), open-arm entry frequency (%), and total arm entries [1] 2. Mouse PTZ-induced seizure assay: Male ICR mice were randomly divided into 4 groups (n=8/group). SSD114 HCl was dissolved in physiological saline and administered intraperitoneally (10, 30, 100 mg/kg); the control group received saline. Thirty minutes later, PTZ (85 mg/kg, dissolved in saline) was injected intraperitoneally. Mice were observed for 30 minutes, and parameters recorded included: seizure latency (time to first generalized clonic seizure with hindlimb extension), seizure incidence (%), and mortality (%) [1] 3. Mouse hot plate analgesic assay: Female ICR mice were screened for baseline hot plate latency (55±0.5°C) (cut-off time: 30 s). Mice with baseline latency <3 s or >30 s were excluded. SSD114 HCl (10, 30, 100 mg/kg) was administered orally; the control group received 0.5% methylcellulose. Hot plate latency was measured at 30, 60, 120, and 240 minutes post-dosing (cut-off time: 60 s to avoid tissue damage). The latency extension percentage was calculated as [(post-dosing latency - baseline latency)/baseline latency] × 100% [1] 4. Mouse rotarod assay (motor coordination test): Male ICR mice were orally administered SSD114 HCl (30, 100, 300 mg/kg) or vehicle. Sixty minutes later, mice were placed on a rotarod (10 rpm, diameter: 3 cm). The latency to fall from the rotarod was recorded (cut-off time: 180 s) to evaluate motor coordination [1] |
| ADME/Pharmacokinetics |
1. Oral pharmacokinetics in mice: Male ICR mice (n=3 per time point) were orally administered SSD114 HCl (30 mg/kg). Blood samples (0.2 mL) were collected from the tail vein at 0.25, 0.5, 1, 2, 4, 6, 8, and 12 hours post-dosing. Plasma was separated by centrifugation (3000×g, 10 minutes, 4°C) and stored at -80°C. SSD114 HCl concentration was measured by LC-MS/MS. Pharmacokinetic parameters were calculated via non-compartmental analysis: oral bioavailability (F) = 42%, Cmax = 2.8 μM, Tmax = 1 hour, terminal half-life (t₁/₂) = 3.5 hours [1] 2. Plasma protein binding rate: Human plasma (500 μL) was mixed with SSD114 HCl (0.1–10 μM) and incubated at 37°C for 4 hours using a dialysis membrane (molecular weight cutoff: 12–14 kDa). Free drug concentration in the dialysate was measured by LC-MS/MS. The plasma protein binding rate was calculated as [(total drug concentration - free drug concentration)/total drug concentration] × 100% = 92% [1] 3. Brain penetration: Male ICR mice were orally administered SSD114 HCl (30 mg/kg). One hour post-dosing, mice were euthanized, and brain tissues were homogenized in saline (1:1, w/v). Plasma and brain homogenate concentrations were measured by LC-MS/MS. The brain/plasma concentration ratio was 0.8, indicating effective blood-brain barrier penetration [1] |
| Toxicity/Toxicokinetics |
1. Acute toxicity in mice: Male ICR mice (n=6) were orally administered a single dose of SSD114 HCl (300 mg/kg, the maximum tested dose). Mice were observed for 7 days for mortality, abnormal behavior (e.g., sedation, ataxia), and body weight changes. No mortality or abnormal behavior was observed; body weight increased normally (similar to the vehicle group). Rotarod test showed no significant difference in fall latency between the 300 mg/kg group and the vehicle group (P > 0.05) [1] 2. Hepatic and renal function: Male ICR mice were orally administered SSD114 HCl (30, 100 mg/kg) or vehicle once daily for 7 days. On day 8, serum was collected to measure alanine transaminase (ALT), aspartate transaminase (AST) (liver function markers), blood urea nitrogen (BUN), and creatinine (renal function markers). No significant differences in these parameters were observed between SSD114 HCl groups and the vehicle group (P > 0.05) [1] 3. CYP450 enzyme inhibition: Human liver microsomes were incubated with SSD114 HCl (0.1–100 μM) and specific substrates for CYP1A2, 2C9, 2C19, 2D6, and 3A4. The metabolic rate of substrates was measured by LC-MS/MS. IC₅₀ > 100 μM for all tested CYP enzymes, indicating no significant drug-drug interaction risk [1] |
| References |
[1]. In vitro and in vivo pharmacological characterization of SSD114, a novel GABAB positive allosteric modulator. Eur J Pharmacol. 2016 Nov 15;791:115-123. |
| Additional Infomation |
1. Background: SSD114 HCl is a novel, selective positive allosteric modulator (PAM) of the GABAB receptor. Unlike traditional GABAB agonists (e.g., baclofen), which directly activate the receptor, PAMs such as SSD114 HCl only enhance receptor activation in the presence of endogenous GABA, reducing the risk of overstimulation and side effects [1] 2. Mechanism of action: SSD114 HCl binds to an allosteric site on the GABAB receptor (distinct from the agonist binding site). This binding induces a conformational change in the receptor, increasing its affinity for GABA and enhancing GABA-mediated G protein activation (e.g., inhibition of adenylyl cyclase, activation of GIRK channels). The result is enhanced inhibitory neurotransmission in the central nervous system [1] 3. Potential indications: Preclinical data suggest SSD114 HCl has potential for treating anxiety disorders, epilepsy (seizures), and chronic pain. Its anxiolytic effect is not accompanied by locomotor suppression (evidenced by unchanged total arm entries in EPM and normal rotarod performance), a key advantage over benzodiazepines (GABA-A modulators) [1] 4. Receptor selectivity: SSD114 HCl exhibits high selectivity for the GABAB receptor. In vitro binding assays showed no significant affinity for other neurotransmitter receptors (GABA-A, 5-HT₁A, D₂, M₁) or ion channels, minimizing off-target effects [1] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~130 mg/mL (~335.20 mM) H2O : ~2 mg/mL (~5.16 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 3.25 mg/mL (8.38 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 32.5 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 3.25 mg/mL (8.38 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 32.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 3.25 mg/mL (8.38 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 32.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5784 mL | 12.8922 mL | 25.7845 mL | |
| 5 mM | 0.5157 mL | 2.5784 mL | 5.1569 mL | |
| 10 mM | 0.2578 mL | 1.2892 mL | 2.5784 mL |