Physicochemical Properties
| Molecular Formula | C16H19F3N4O2S |
| Molecular Weight | 388.41 |
| Exact Mass | 388.12 |
| Elemental Analysis | C, 49.48; H, 4.93; F, 14.67; N, 14.42; O, 8.24; S, 8.25 |
| CAS # | 2322245-42-9 |
| Related CAS # | SRI-37330 hydrochloride;2322245-49-6 |
| PubChem CID | 142582737 |
| Appearance | White to off-white solid powder |
| LogP | 2.7 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 26 |
| Complexity | 581 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | WRSNFEVXRKOMLL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H19F3N4O2S/c1-26(24,25)22-8-11-3-2-6-23(9-11)15-13-7-12(16(17,18)19)4-5-14(13)20-10-21-15/h4-5,7,10-11,22H,2-3,6,8-9H2,1H3 |
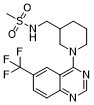
| Chemical Name | N-[[1-[6-(trifluoromethyl)quinazolin-4-yl]piperidin-3-yl]methyl]methanesulfonamide |
| Synonyms | SRI-37330 free base; SRI37330 free base, SRI 37330 free base |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | In INS-1 cells, SRI-37330 (1 μM, 24 hours) suppresses the activity of the human TXNIP promoter [1]. In INS-1 cells, TXNIP's mRNA and protein levels are inhibited by SRI-37330 (1 μM, 24 hours) [1]. The binding of polymerase II (Pol II) to the TXNIP promoter's E-box motif region is inhibited by SRI-37330 (5 μM, 24 hours) [1]. In TC1-6 cells, SRI-37330 (5 μM, 24 hours) decreases glucagon secretion [1]. In primary hepatocytes, glucagon-induced glucose production is inhibited by SRI-37330 (0-5 μM, 24 hours) [1]. |
| ln Vitro |
In INS-1 cells, SRI-37330 (1 μM, 24 hours) suppresses the activity of the human TXNIP promoter [1]. In INS-1 cells, TXNIP's mRNA and protein levels are inhibited by SRI-37330 (1 μM, 24 hours) [1]. The binding of polymerase II (Pol II) to the TXNIP promoter's E-box motif region is inhibited by SRI-37330 (5 μM, 24 hours) [1]. In TC1-6 cells, SRI-37330 (5 μM, 24 hours) decreases glucagon secretion [1]. In primary hepatocytes, glucagon-induced glucose production is inhibited by SRI-37330 (0-5 μM, 24 hours) [1]. SRI-37330 inhibited the activity of the human TXNIP promoter in INS-1 cells by approximately 70% in luciferase reporter assays. SRI-37330 inhibited endogenous TXNIP mRNA expression in INS-1 cells with an IC50 of 0.64 µM in a 7-point dose-response qRT-PCR assay, and no cytotoxicity was observed. SRI-37330 inhibited TXNIP protein levels in a dose-dependent manner in INS-1 cells. SRI-37330 significantly inhibited TXNIP expression in rat INS-1 cells, primary mouse islets, and isolated human islets under high glucose (25 mM) conditions. RNA sequencing of human islets treated with SRI-37330 showed differential expression of genes involved in TXNIP regulation and signaling pathways, including downregulation of TXNIP and MLXIPL, and upregulation of IGF-1R, MAFA, BCL2L1, and CTGF. SRI-37330 did not affect the expression of other arrestin genes (ARRB1, ARRDC3) or thioredoxin, indicating specificity. SRI-37330 inhibited glucagon secretion from mouse alpha TC1-6 cells without affecting proglucagon gene expression or glucagon content. This inhibition was mimicked by TXNIP knockdown and reversed by TXNIP overexpression. SRI-37330 did not inhibit glucagon secretion under low glucose conditions or when secretion was maximally stimulated by arginine and norepinephrine. In primary mouse hepatocytes, SRI-37330 did not affect basal or lactate-stimulated glucose output in the absence of glucagon. However, in the presence of glucagon, it dose-dependently decreased glucose output and inhibited glucagon-induced cAMP production and expression of gluconeogenic genes (Pck1, G6pc). The inhibitory effect of SRI-37330 on hepatocyte glucose output and cAMP production was absent in hepatocytes from liver-specific glucagon receptor knockout mice but remained intact in hepatocytes from TXNIP-deficient mice, indicating the hepatic effect is glucagon receptor-dependent but TXNIP-independent. Chromatin immunoprecipitation showed SRI-37330 inhibited RNA polymerase II binding to the E-box motif region of the TXNIP promoter. Mutation of this E-box motif abolished the inhibitory effect of SRI-37330 on promoter activity, and the motif could confer SRI-37330 responsiveness to a heterologous promoter. |
| ln Vivo |
SRI-37330 inhibits hepatic glucose production and decreases glucagon secretion when taken orally for three weeks at a dose of 100 mg/kg in drinking water [1]. Male C57BL/6J mice responded favorably to SRI-37330 (100 mg/kg, orally, in drinking water) when given for three weeks [1]. Mice treated with SRI-37330 (100 mg/kg, orally, in drinking water) for three weeks show improvement in hepatic steatosis, hyperglycemia caused by STZ, and obesity [1]. Oral administration of SRI-37330 (100 mg/kg/day in drinking water) to male C57BL/6J mice for 3 weeks was well tolerated, caused no change in body weight, and no gross organ abnormalities. SRI-37330 treatment significantly decreased non-fasting and fasting blood glucose levels in C57BL/6J mice within 3 days, an effect that persisted for 3 weeks. It also improved glucose tolerance in intraperitoneal glucose tolerance tests (GTTs) but did not alter insulin tolerance tests (ITTs). Fasting serum glucagon levels were significantly lower in SRI-37330-treated C57BL/6J mice, while fasting serum insulin levels were unchanged. Hyperinsulinemic-euglycemic clamp studies in C57BL/6J mice revealed that SRI-37330 significantly downregulated basal hepatic glucose production but did not affect insulin sensitivity, insulin-stimulated glucose uptake in skeletal muscle or white adipose tissue, glucose infusion rates, or hepatic insulin action. In SRI-37330-treated C57BL/6J mice, liver expression of gluconeogenic enzymes (Pck1, G6pc) was lower, and liver glycogen content was higher compared to controls. Liver and serum triglyceride levels were lower, and no elevation in liver transaminases was observed. In obese, diabetic db/db mice, oral SRI-37330 (100 mg/kg/day for 4 weeks) did not affect body weight but significantly decreased non-fasting and fasting blood glucose levels, returning them to euglycemic levels. Fasting serum glucagon was lowered, while fasting serum insulin was unchanged. Hyperinsulinemic-euglycemic clamp studies in db/db mice confirmed significantly lower basal hepatic glucose production with SRI-37330 treatment, while insulin-stimulated glucose uptake in muscle and adipose tissue remained low and unchanged. SRI-37330 treatment did not cause alpha cell hyperplasia in db/db mice; instead, it led to a small but significant decline in alpha cell mass and number, while beta cell mass and number were unchanged. SRI-37330 dramatically improved the severe hepatic steatosis observed in db/db mice. In C57BL/6J mice with streptozotocin (STZ)-induced diabetes, oral SRI-37330 (starting after STZ regimen) maintained significantly lower blood glucose levels compared to untreated mice. This effect was superior to metformin and empagliflozin in this model. SRI-37330 treatment in STZ-diabetic mice resulted in lower urine glucose (unlike empagliflozin), lower serum triglycerides, higher body weight, higher serum insulin, and dramatically lower (3.6-fold) fasting serum glucagon levels compared to untreated controls. SRI-37330 was also effective in improving glucose homeostasis when treatment was started two weeks after the onset of overt STZ-induced diabetes. Oral gavage administration of SRI-37330 twice daily also demonstrated effectiveness in obesity- and STZ-induced diabetes, showing dose-dependent glucose-lowering effects. In TXNIP-deficient mice, SRI-37330 treatment no longer improved glucose homeostasis or lowered serum glucagon levels, underscoring the role of TXNIP inhibition in its mechanism of action. Mice treated with SRI-37330 maintained blood glucose levels indistinguishable from untreated mice even during insulin-induced hypoglycemia, indicating a low hypoglycemic risk. |
| Cell Assay |
RT-PCR[1] Cell Types: INS-1 Cell Tested Concentrations: 1 μM Incubation Duration: 24 h Experimental Results: Inhibits endogenous TXNIP mRNA expression with an IC50 of 0.64 μM. For TXNIP promoter activity, INS-1 rat beta-like cells were stably transfected with a human TXNIP promoter luciferase reporter plasmid. Cells were maintained overnight in 5 mM glucose media, then incubated for 24 hours in 25 mM glucose media with or without test compounds. Luciferase activity was measured to identify inhibitors. For TXNIP mRNA expression, INS-1 cells cultured in 96-well plates were treated with compounds. RNA was extracted, and one-step qRT-PCR was performed using specific primers. Gene expression was corrected using the 18S ribosomal subunit as an internal standard. For assessing effects on human islet gene expression, isolated human islets were incubated overnight at 5 mM glucose, then at 25 mM glucose in the presence or absence of SRI-37330 for 24 hours prior to RNA extraction and RNA sequencing analysis. For alpha cell studies, mouse alpha TC1-6 cells were incubated with SRI-37330 or vehicle. Proglucagon mRNA was assessed by qRT-PCR after 24 hours. Glucagon content and secretion were measured after 1 hour of incubation using a glucagon ELISA kit, normalized to total protein. For TXNIP modulation in alpha cells, cells were transfected with TXNIP-specific siRNA or scrambled siRNA using a transfection reagent, or with a TXNIP expression plasmid or control plasmid. Glucagon secretion was assessed after transfection. For hepatocyte studies, primary mouse hepatocytes were isolated and cultured in collagen-coated plates. After attachment, cells were incubated with different concentrations of SRI-37330 for 24 hours. Glucose output was assessed during the last 6 hours of incubation in substrate-free or lactate-containing media, with or without glucagon. Glucose concentration in the media was measured and normalized to total protein. For hepatocyte cAMP production, cells were washed and harvested after treatment. Intracellular cAMP levels were measured using a direct immunoassay kit. For chromatin immunoprecipitation (ChIP), INS-1 cells were cross-linked with formaldehyde. Chromatin was sonicated and immunoprecipitated with an anti-RNA polymerase II antibody or control IgG. Precipitated DNA was purified and quantified by real-time PCR using primers specific for the TXNIP promoter E-box region and control regions. |
| Animal Protocol |
Animal/Disease Models: C57BL/6J mice[1] Doses: 100 mg/kg Route of Administration: Orally (po), dissolved in drinking water, for 3 weeks. Experimental Results: diminished serum glucagon levels, inhibited hepatic glucose production and improved glucose homeostasis in mice. For studies in lean C57BL/6J mice and STZ-induced diabetic C57BL/6J mice, SRI-37330 was administered orally in the drinking water at a dose of 100 mg/kg/day. Control mice received untreated water. The dose was chosen based on previous studies with verapamil in similar models. Blood glucose was monitored using a glucometer. For studies in obese, diabetic db/db mice, SRI-37330 was similarly administered in drinking water at 100 mg/kg/day for 4 weeks. For comparative drug studies in STZ-diabetic mice, metformin (250 mg/kg/day) and empagliflozin (10 mg/kg/day) were also administered in the drinking water. For oral gavage experiments, mice were dosed with SRI-37330 twice a day, 12 hours apart. For STZ-induced diabetes, C57BL/6J mice received multiple low-dose streptozotocin injections (40 mg/kg/day for 5 days, freshly prepared in sodium citrate buffer, pH 4.5). For glucose tolerance tests (GTTs), mice were fasted for 4 hours and then injected intraperitoneally with glucose (3 g/kg). For insulin tolerance tests (ITTs), fasted mice were injected intraperitoneally with insulin (1 U/kg). For hyperinsulinemic-euglycemic clamp studies, lean C57BL/6J or db/db mice treated with SRI-37330 underwent survival surgery to implant a jugular vein catheter. After an overnight fast, a 2-hour clamp was conducted in awake mice with a primed and continuous infusion of human insulin. Euglycemia was maintained by variable rate glucose infusion. Whole-body glucose turnover was assessed with a continuous infusion of [3-³H]glucose, and tissue-specific glucose uptake was measured using a bolus of 2-deoxy-D-[1-¹⁴C]glucose administered during the clamp. At the end, mice were anesthetized and tissues collected for analysis. For assessment of islet cell morphometry, pancreata from treated and untreated mice were sectioned and immunofluorescently stained for insulin and glucagon. Images were analyzed to quantify alpha and beta cell area, mass, and number. For liver histology and glycogen assessment, liver tissues were collected, sectioned, and stained (e.g., for steatosis) or homogenized for biochemical glycogen measurement. |
| ADME/Pharmacokinetics |
SRI-37330 is described as orally bioavailable. In mice receiving SRI-37330 at 100 mg/kg/day in drinking water, the average plasma concentration achieved was 5 µmol/L. No further detailed pharmacokinetic parameters (e.g., half-life, AUC, oral bioavailability percentage) are provided in the manuscript. |
| Toxicity/Toxicokinetics |
SRI-37330 showed no cytotoxicity in vitro in a cellular cytotoxicity assay (CC50/72h > 5 µM). In vivo, oral administration of SRI-37330 at 100 mg/kg/day for 3 weeks to C57BL/6J mice was well tolerated, caused no change in body weight, and no gross abnormalities in any organs were observed at sacrifice. SRI-37330 treatment did not cause any elevation in liver transaminase levels in mice. No issues with hypoglycemia were observed under any circumstances, including during insulin-induced hypoglycemia challenges. The manuscript states that SRI-37330 has a favorable safety profile and lack of off-target liabilities in the available screening panel. |
| References |
[1]. Identification of an Anti-diabetic, Orally Available Small Molecule that Regulates TXNIP Expression and Glucagon Action. Cell Metab. 2020 Sep 1;32(3):353-365.e8. |
| Additional Infomation |
SRI-37330 is a substituted quinazoline sulfonamide small molecule identified through high-throughput screening of 300,000 compounds and extensive medicinal chemistry optimization. It represents a novel approach to diabetes treatment by targeting TXNIP expression and glucagon action, addressing underlying processes of the disease characterized by hyperglycemia, beta-cell loss, and persistent glucagon secretion. Its mechanism involves TXNIP-dependent inhibition of alpha cell glucagon secretion and glucagon receptor-dependent (but TXNIP-independent) inhibition of hepatic glucagon sensitivity and action. Compared to existing oral anti-diabetic drugs like metformin and SGLT2 inhibitors (e.g., empagliflozin), SRI-37330 showed superior or comparable glucose-lowering effects in mouse models without increasing urinary glucose excretion. SRI-37330 improved hepatic steatosis in diabetic mice, suggesting potential therapeutic benefit for non-alcoholic fatty liver disease (NAFLD), a common diabetes comorbidity. Unlike verapamil (a known TXNIP-lowering L-type calcium channel blocker), SRI-37330 is not a calcium channel blocker, potentially avoiding limitations like hypotension. The studies suggest SRI-37330 may be a promising candidate for treating both type 1 and type 2 diabetes, potentially as an oral therapy. A patent is related to this work: WO 2019/089693 A1. Limitations of the study include that the precise molecular target of SRI-37330 (especially for its TXNIP-independent hepatic effects) remains unidentified, and the drug has not been tested in high-fat-fed mice or spontaneous autoimmune type 1 diabetes models. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~257.46 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5746 mL | 12.8730 mL | 25.7460 mL | |
| 5 mM | 0.5149 mL | 2.5746 mL | 5.1492 mL | |
| 10 mM | 0.2575 mL | 1.2873 mL | 2.5746 mL |