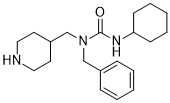
SRI-011381 (SRI011381) is a novel and potent agonist of the TGF-beta signaling pathway with the potential for treatment of Alzheimer's disease. SRI-011381 reversed or prevented neuronal damage consistently by at least 30% for all neuronal markers.
Physicochemical Properties
| Molecular Formula | C20H31N3O | |
| Molecular Weight | 329.479645013809 | |
| Exact Mass | 329.246 | |
| CAS # | 1629138-41-5 | |
| Related CAS # | SRI-011381 hydrochloride;2070014-88-7 | |
| PubChem CID | 77050694 | |
| Appearance | White to off-white solid powder | |
| LogP | 3.2 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 2 | |
| Rotatable Bond Count | 5 | |
| Heavy Atom Count | 24 | |
| Complexity | 369 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O=C(NC1CCCCC1)N(CC1C=CC=CC=1)CC1CCNCC1 |
|
| InChi Key | LNOPAJNGRAPFKZ-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C20H31N3O/c24-20(22-19-9-5-2-6-10-19)23(15-17-7-3-1-4-8-17)16-18-11-13-21-14-12-18/h1,3-4,7-8,18-19,21H,2,5-6,9-16H2,(H,22,24) | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | In mouse lung blasts, SRI-011381 (10 μM) enhances the production of TGF-β1, NALP3, collagen-1, and α-SMA substantially and promotes fibroblast swelling [1]. |
| ln Vitro |
In mouse lung blasts, SRI-011381 (10 μM) enhances the production of TGF-β1, NALP3, collagen-1, and α-SMA substantially and promotes fibroblast swelling [1]. In primary cultured mouse lung fibroblasts, treatment with a 10 μM solution of SRI-011381 was used to activate the TGF-β1 signaling pathway. [1] SRI-011381 promoted the proliferation of lung fibroblasts, as evidenced by a significantly increased cell survival rate compared to the control group in a CCK-8 assay. [1] Treatment with SRI-011381 significantly increased the protein expression levels of fibrosis-related markers, including TGF-β1, neutrophil alkaline phosphatase 3 (NALP3), collagen-1, and α-smooth muscle actin (α-SMA) in lung fibroblasts, as detected by western blot. [1] SRI-011381 treatment also increased the levels of caspase-1 and interleukin-1β (IL-1β) in the culture supernatant of lung fibroblasts, as measured by ELISA. [1] SRI-011381 partially reversed the inhibitory effects of sodium ferulate (SF) on fibroblast proliferation and the expression of the aforementioned fibrosis-related proteins (TGF-β1, NALP3, collagen-1, α-SMA) and inflammatory mediators (caspase-1, IL-1β). [1] |
| ln Vivo |
In YAPGFAP-CKO EAE mice, SRI-011381 (30 mg/kg; i.p.; every 2 days; for 22 days) partially restores optic nerve and drive deficits [2]. In an experimental autoimmune encephalomyelitis (EAE) mouse model, intraperitoneal administration of SRI-011381 (30 mg/kg, every 2 days) significantly reduced clinical scores in YAPGFAP-CKO EAE mice compared to control-treated mice. [2] SRI-011381 treatment alleviated demyelination in the optic nerve of both YAPf/f EAE and YAPGFAP-CKO EAE mice. [2] SRI-011381 treatment ameliorated inflammatory infiltration (reduced Iba1+ microglia, GFAP+ astrocytes, and CD45+ T cells) in the optic nerve of EAE mice. [2] In the retina, SRI-011381 treatment suppressed inflammatory infiltration and rescued retinal ganglion cell (RGC) loss in YAPGFAP-CKO EAE mice. [2] |
| Cell Assay |
For cell viability assay: Mouse lung fibroblasts were seeded in 96-well plates at a density of 5×10^4/mL. After cells adhered and reached 70% confluence, they were starved overnight in serum-free medium. Cells were then treated with the test compound (SRI-011381 at 10 µM) in fresh serum-free medium and cultured for 48 hours. Subsequently, 10 µL of CCK-8 solution was added to each well, followed by incubation at 37°C for 1 hour. Absorbance was measured at 450 nm using a microplate reader. For protein expression analysis: After treatment, total protein was extracted from cells using RIPA lysis buffer containing protease inhibitors. Protein concentration was determined using a BCA assay kit. Equal amounts of protein were separated by SDS-PAGE and transferred to PVDF membranes. The membranes were blocked with 5% skim milk, then incubated with primary antibodies against target proteins (e.g., TGF-β1, NALP3, collagen-1, α-SMA) overnight at 4°C. After washing, membranes were incubated with HRP-conjugated secondary antibodies at 37°C for 1 hour. Protein bands were visualized using an ECL chemiluminescence reagent and a gel imaging system.[1] |
| Animal Protocol |
Animal/Disease Models: YAPGFAP-CKO mice experimental autoimmune encephalomyelitis (EAE) [2] Doses: 30 mg/kg Route of Administration: intraperitoneal (ip) injection; once every 2 days; for 22 days Experimental Results: Dramatically inhibited YAPGFAP- Inflammatory infiltration in CKO EAE mice reduces neuronal loss. After EAE induction, SRI-011381 was dissolved in saline containing 10% DMSO and 40% PEG300. It was administered intraperitoneally at a dose of 30 mg/kg every 2 days. [2] |
| References |
[1]. The Improvement Effect of Sodium Ferulate on the Formation of Pulmonary Fibrosis in Silicosis Mice Through the Neutrophil Alkaline Phosphatase 3 (NALP3)/Transforming Growth Factor-β1 (TGF-β1)/α-Smooth Muscle Actin (α-SMA) Pathway. Med Sci Monit. 2021 Jun 15;27:e927978. [2]. Astrocytic YAP protects the optic nerve and retina in an experimental autoimmune encephalomyelitis model through TGF-β signaling. Theranostics. 2021 Jul 25;11(17):8480-8499. |
| Additional Infomation |
SRI-011381 was used as a pharmacological tool to activate the TGF-β signaling pathway in vivo to investigate its role in neuroinflammation and demyelination in EAE-induced optic neuritis. [2] Activation of TGF-β signaling by SRI-011381 partially rescued the deficits in optic nerve and retina caused by astrocytic YAP knockout, suggesting that YAP exerts its protective effects at least partially through TGF-β signaling. [2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.59 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.59 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (7.59 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0351 mL | 15.1754 mL | 30.3509 mL | |
| 5 mM | 0.6070 mL | 3.0351 mL | 6.0702 mL | |
| 10 mM | 0.3035 mL | 1.5175 mL | 3.0351 mL |