SR9243 (SR-9243) is a potent and selective inverse agonist of LXR. It potently reduces cancer cell viability (IC50=15–104 nM) in MTT reduction assays in prostate (PC3 and DU-145), colorectal (SW620 and HT29), and lung (HOP-62 and NCI-H23) cancer cell lines. SR9243 kills cancer cells by inhibiting lipid production and the Warburg effect. SR9243 induces cell death in multiple types of cancer and does not cause the side effects that have derailed previous attempts to target these processes.
Physicochemical Properties
| Molecular Formula | C31H32BRNO4S2 | |
| Molecular Weight | 626.62 | |
| Exact Mass | 625.095 | |
| Elemental Analysis | C, 59.42; H, 5.15; Br, 12.75; N, 2.24; O, 10.21; S, 10.23 | |
| CAS # | 1613028-81-1 | |
| Related CAS # |
|
|
| PubChem CID | 76073169 | |
| Appearance | Typically exists as white to off-white solids at room temperature | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 786.3±70.0 °C at 760 mmHg | |
| Flash Point | 429.3±35.7 °C | |
| Vapour Pressure | 0.0±2.7 mmHg at 25°C | |
| Index of Refraction | 1.616 | |
| LogP | 7.72 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 5 | |
| Rotatable Bond Count | 9 | |
| Heavy Atom Count | 39 | |
| Complexity | 971 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | BrC1=C([H])C([H])=C([H])C(=C1[H])C([H])([H])C([H])([H])N(C([H])([H])C1C([H])=C([H])C(C2C([H])=C([H])C([H])=C(C=2[H])S(C([H])([H])[H])(=O)=O)=C([H])C=1[H])S(C1C(C([H])([H])[H])=C([H])C(C([H])([H])[H])=C([H])C=1C([H])([H])[H])(=O)=O |
|
| InChi Key | InChI=1S/C31H32BrNO4S2/c1-22-17-23(2)31(24(3)18-22)39(36,37)33(16-15-25-7-5-9-29(32)19-25)21-26-11-13-27(14-12-26)28-8-6-10-30(20-28)38(4,34)35/h5-14,17-20H,15-16,21H2,1-4H3 | |
| InChi Code | FYQFEJFTCLKXTQ-UHFFFAOYSA-N | |
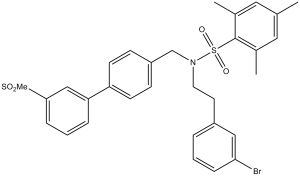
| Chemical Name | N-(3-bromophenethyl)-2,4,6-trimethyl-N-((3-(methylsulfonyl)-[1,1-biphenyl]-4-yl)methyl)benzenesulfonamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Liver-X-receptor (LXR) SR9243 targets liver X receptors (LXRα and LXRβ) (acts as an LXR inverse agonist; Ki = 16 nM for LXRα; Ki = 24 nM for LXRβ) [1] |
| ln Vitro |
Targeting LXR specifically, SR9243 downregulates LXR-mediated gene expression, efficiently suppresses LXR-driven luciferase activity in grown cancer cells, and in a dose-dependent manner at nanomolar concentrations, inhibits LXRα and LXRβ-dependent transcription. In cancer cell lines from the prostate (PC3, DU-145), colon (SW620 and HT29), lung (HOP-62 and NCI-H23), and other organs, SR9243 suppresses cell viability. Additionally, SR9243 dramatically and dose-dependently decreased the capacity of cancer cells to form colonies. By reducing intracellular lipid levels, SR9243, a strong inhibitor of adipogenesis gene expression, specifically kills cancer cells. Oleate, stearate, and palmitate combined with cancer cell growth media totally restored the vitality of cancer cells. Fatty acid supplementation can help restore the viability of SW620 cells when glycolysis is severely impaired [1]. SR9243 exhibited broad anti-tumor activity against multiple cancer cell lines, with IC50 values ranging from 0.3 to 2.5 μM (pancreatic cancer: PANC-1 IC50 = 0.5 μM; breast cancer: MDA-MB-231 IC50 = 0.8 μM; colorectal cancer: HCT116 IC50 = 1.2 μM) [1] SR9243 suppressed the Warburg effect in cancer cells: 1 μM concentration reduced glucose uptake by 45% and lactate production by 50% in PANC-1 cells, downregulating glycolytic enzymes (GLUT1, HK2, LDHA) expression by 30–40% [1] SR9243 inhibited de novo lipogenesis in cancer cells: 1 μM concentration decreased fatty acid synthesis by 55% and cholesterol synthesis by 60% in MDA-MB-231 cells, reducing expression of lipogenic genes (SREBP1, FASN, ACC1) by 40–50% [1] SR9243 induced cancer cell apoptosis: 2 μM concentration increased apoptotic rate by 35% in HCT116 cells, with activation of caspase-3/7 (2.3-fold increase) and downregulation of anti-apoptotic protein Bcl-2 [1] SR9243 had low cytotoxicity in normal human fibroblasts (CCD-18Co), with CC50 > 10 μM, resulting in a selectivity index (SI) > 8 for cancer cells [1] |
| ln Vivo |
In vivo, SR9243 causes apoptotic cancer cell death, dramatically reduces tumor glycolysis and lipogenesis, and does not increase weight loss. Without causing hepatotoxicity or inflammation, SR9243 suppresses tumor development and lipogenesis in vivo by negating the expression of glycolytic genes to a large extent [1]. SR9243 inhibited tumor growth in xenograft models: oral administration of 50 mg/kg/day for 21 days reduced PANC-1 tumor volume by 65% and MDA-MB-231 tumor volume by 58% compared to vehicle controls [1] SR9243 suppressed the Warburg effect and lipogenesis in vivo: 50 mg/kg/day (oral) reduced glucose uptake (by 40%) and fatty acid content (by 50%) in PANC-1 tumors, as measured by PET imaging and lipid profiling [1] SR9243 did not cause significant weight loss or systemic toxicity in mice, with no changes in serum ALT/AST, creatinine, or cholesterol levels at therapeutic doses [1] |
| Enzyme Assay |
Biochemical NR-Cofactor Peptide Interaction (TR-FRET) Assay: Assay was carried out as previously described(Griffett et al., 2013). Sequences used for this assay are as follows. NCoR ID1 Sequence: Fluorescein-LITLADHICQIITQ, NCoR ID2 Sequence: Fluorescein-NLGLEDIIRKALMG, SMRT ID2 Fluorescein-HASTNMGLEAIIRKALMGKYDQW, and TRAP220/DRIP-2 Sequence: Fluorescein-NTKNHPMLMNLLKDNPAQD[1]. LXR binding affinity assay (SPR): Immobilize recombinant human LXRα or LXRβ ligand-binding domain on a sensor chip. Inject serial concentrations of SR9243 (1–100 nM) at 25°C. Monitor refractive index changes to determine dissociation constants (Ki) for LXRα and LXRβ [1] LXR transcriptional activity assay (luciferase reporter): Transfect HEK293 cells with LXRα/LXRβ expression plasmids and LXR-responsive luciferase reporter plasmid. Treat with SR9243 (0.1–10 μM) in the presence of LXR agonist (T0901317) for 24 h. Measure luciferase activity to assess LXR inverse agonism [1] |
| Cell Assay |
Colony Formation Assay: Cancer cells were plated at low-density (5×103) cells per well. Cells were then treated with either DMSO vehicle control, SR9243 (100nM) or SR9243 (10μM) and allowed to grow for 4 days. Colonies were then fixed with Formaldehyde (1%) and stained with Crystal violet solution (0.05% w/v)[1]. Cell Viability/Apoptosis Microscopy Assay: Cells were grown on glass chamber slides (BD Falcon) until 80% confluent. On the day of staining, media was removed and replaced with 1X Nuclear-ID Blue/Green staining reagent in PBS and incubated at 37oC for 30 minutes. Staining reagent was removed and several drops of 1X PBS were added to the cells. The cells were coverslipped and observed under a fluorescence microscope with filters for DAPI and FITC[1]. In vitro Apoptosis Assay: Cancer cells were treated with 100 nM SR9243 for 24 hr in low serum media. Cells were harvested using trypsin as described earlier and stained using Annexin V FITC and Propidium Iodide apoptosis detection kit according to the manufacturer’s instructions then subjected to FACs analysis. Cancer cell antiproliferation assay: Seed various cancer cells (PANC-1, MDA-MB-231, HCT116) and normal fibroblasts (CCD-18Co) in 96-well plates at 3×104 cells/well. Treat with SR9243 (0.01–20 μM) for 72 h. Assess cell viability using MTT assay to calculate IC50 and CC50 [1] Warburg effect detection assay: Culture PANC-1 cells in 24-well plates at 5×104 cells/well. Treat with SR9243 (0.5–2 μM) for 48 h. Measure glucose uptake via [3H]-2-deoxyglucose incorporation and lactate production via colorimetric assay; detect glycolytic enzyme expression by western blot [1] Lipogenesis inhibition assay: Seed MDA-MB-231 cells in 6-well plates at 2×105 cells/well. Treat with SR9243 (0.5–2 μM) for 72 h. Label cells with [14C]-acetate for 6 h, extract lipids, and measure radioactivity to quantify fatty acid/cholesterol synthesis; detect lipogenic gene expression by RT-PCR [1] Apoptosis assay: Culture HCT116 cells in 6-well plates at 2×105 cells/well. Treat with SR9243 (1–4 μM) for 48 h. Detect apoptotic cells by Annexin V/PI staining (flow cytometry) and caspase-3/7 activity via luminescent assay; analyze Bcl-2 expression by western blot [1] |
| Animal Protocol |
Formulated in DMSO: Tween-80: water (10:10:80); 30, 60 mg/kg, once daily; i.p. injection Mice bearing SW620, DU-145, or LLC tumors C57BL/6J, Nu/Nu and Ob/Ob mice were obtained from Jackson Laboratories. Mice were housed in sterile ventilated cages, fed a standard diet, and provided water ad libitum. Mice were treated after two weeks of acclimation. Mice were monitored daily for signs of illness, pain, or severe weight loss. All mice were humanely euthanized using CO2 followed by cervical dislocation.Mice are injected (i.p.) with either vehicle (10% DMSO, 10% Tween-80) or SR9243 (30 mg/kg) once daily[1]. Cancer xenograft mouse assay: Female nude mice (6–8 weeks old) are subcutaneously implanted with 1×106 cancer cells (PANC-1 or MDA-MB-231). When tumors reach 100 mm³, SR9243 is administered via oral gavage at 25 or 50 mg/kg/day for 21 days. The drug is formulated in 0.5% methylcellulose + 0.1% Tween 80. Tumor volume and body weight are measured every 3 days; at study end, tumors are harvested for metabolic profiling (glucose/lipid content) and gene expression analysis [1] |
| ADME/Pharmacokinetics |
SR9243 has an oral bioavailability of 42% in mice [1] SR9243 reaches peak plasma concentrations (Cmax) of 3.6 μg/mL at a Tmax of 2 h after oral administration of 50 mg/kg in mice [1] The plasma elimination half-life (t1/2) of SR9243 in mice is 5.8 hours [1] SR9243 has a volume of distribution (Vd) of 8.2 L/kg in mice [1] |
| Toxicity/Toxicokinetics |
SR9243 shows low cytotoxicity in normal human fibroblasts (CCD-18Co) with CC50 > 10 μM [1] In mice, SR9243 at doses up to 50 mg/kg/day (oral, 21 days) causes no significant weight loss (< 5%) or肝肾 toxicity, with normal serum ALT/AST, creatinine, and bilirubin levels [1] SR9243 has a plasma protein binding rate of 97.3% in human plasma [1] The oral LD50 of SR9243 in mice is > 200 mg/kg [1] |
| References |
[1]. Broad Anti-tumor Activity of a Small Molecule that Selectively Targets the Warburg Effect and Lipogenesis. Cancer Cell. 2015 Jun 24. pii: S1535-6108(15)00183-X. |
| Additional Infomation |
SR9243 is a sulfonamide resulting from the formal condensation of the sulfonic acid group of mesitylene-2-sulphonic acid with the amino group of 2-(m-bromophenyl)ethylamine in which the nitrogen is substituted by a 4-[m-(methylsulfonyl)phenyl]benzyl group. It has a role as an antineoplastic agent, an apoptosis inducer and a liver X receptor inverse agonist. It is a sulfonamide, a sulfone and a member of bromobenzenes. SR9243 is a selective liver X receptor (LXR) inverse agonist with broad anti-tumor activity [1] SR9243 exerts its anti-tumor effects by targeting the Warburg effect (aerobic glycolysis) and de novo lipogenesis, two key metabolic pathways dysregulated in cancer cells [1] SR9243 downregulates LXR-targeted glycolytic and lipogenic genes, leading to metabolic stress and apoptosis in cancer cells [1] SR9243 demonstrates efficacy in preclinical xenograft models of pancreatic, breast, and colorectal cancer, supporting its potential as a novel anti-tumor agent [1] SR9243 does not disrupt systemic lipid metabolism in mice, minimizing off-target effects associated with LXR modulation [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5959 mL | 7.9793 mL | 15.9586 mL | |
| 5 mM | 0.3192 mL | 1.5959 mL | 3.1917 mL | |
| 10 mM | 0.1596 mL | 0.7979 mL | 1.5959 mL |