SR9238 is the first selective synthetic LXR (Liver X receptor) inverse antagonist with IC50s of 214 nM and 43 nM for LXRα and LXRβ, respectively. SR9238 can effectively suppresses hepatic lipogenesis, inflammation, and hepatic lipid accumulation in a mouse model of non-alcoholic hepatosteatosis. SR9238 displays high potency for both LXRα and LXRβ (40-200 nM IC50) and was designed to display liver specificity so as to avoid potential side effects due to suppression of LXR in the periphery. Unexpectedly, treatment of diet-induced obese mice with SR9238 suppressed plasma cholesterol levels. These data indicate that liver-selective LXR inverse agonists such as SR9238 may hold utility in the treatment of liver disease. Fatty liver, which often accompanies obesity and type 2 diabetes, frequently leads to a much more debilitating hepatic disease including non-alcoholic steatohepatitis, cirrhosis, and hepatocellular carcinoma. Current pharmacological therapies lack conclusive efficacy and thus treatment options are limited. Novel therapeutics that suppress either hepatic lipogenesis and/or hepatic inflammation may be useful.
Physicochemical Properties
| Molecular Formula | C31H33NO7S2 | |
| Molecular Weight | 595373 | |
| Exact Mass | 595.169 | |
| Elemental Analysis | C, 62.50; H, 5.58; N, 2.35; O, 18.80; S, 10.76 | |
| CAS # | 1416153-62-2 | |
| Related CAS # |
|
|
| PubChem CID | 71478195 | |
| Appearance | White to off-white solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 787.7±70.0 °C at 760 mmHg | |
| Flash Point | 430.2±35.7 °C | |
| Vapour Pressure | 0.0±2.7 mmHg at 25°C | |
| Index of Refraction | 1.590 | |
| LogP | 5.79 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 8 | |
| Rotatable Bond Count | 11 | |
| Heavy Atom Count | 41 | |
| Complexity | 1060 | |
| Defined Atom Stereocenter Count | 0 | |
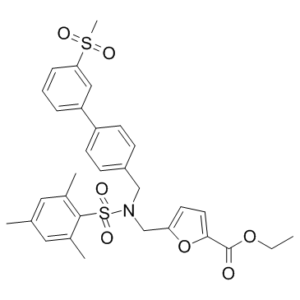
| SMILES | S(C1C(C)=CC(C)=CC=1C)(N(CC1=CC=C(C(=O)OCC)O1)CC1C=CC(C2C=CC=C(C=2)S(C)(=O)=O)=CC=1)(=O)=O |
|
| InChi Key | HDZWHJYZJWLTAG-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C31H33NO7S2/c1-6-38-31(33)29-15-14-27(39-29)20-32(41(36,37)30-22(3)16-21(2)17-23(30)4)19-24-10-12-25(13-11-24)26-8-7-9-28(18-26)40(5,34)35/h7-18H,6,19-20H2,1-5H3 | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | The results of the cell-based co-transfection assay demonstrated that SR9238 is a synthetic LXR inverse agonist, with an IC50 for LXRα of 214 nM and LXRβ of 43 nM. Additionally, SR9238 successfully suppresses the luciferase reporter gene's transcription, which is triggered by the fatty acid synthase (Fasn) promoter. The results showed that SR9238 increased the interaction between LXRα and LXRβ and the CoRNR box peptides obtained from NCoR (NCoR ID1 and NCoR ID2), but decreased the interaction with the coactivator NR box peptide produced from TRAP220. For both LXRα and LXRβ, SR9238-induced recruitment of CoRNR box peptides was dose-dependent. Fasn and Srebp1c mRNA expression was significantly reduced in HepG2 cells treated with SR9238 [1]. |
| ln Vitro |
The results of the cell-based co-transfection assay demonstrated that SR9238 is a synthetic LXR inverse agonist, with an IC50 for LXRα of 214 nM and LXRβ of 43 nM. Additionally, SR9238 successfully suppresses the luciferase reporter gene's transcription, which is triggered by the fatty acid synthase (Fasn) promoter. The results showed that SR9238 increased the interaction between LXRα and LXRβ and the CoRNR box peptides obtained from NCoR (NCoR ID1 and NCoR ID2), but decreased the interaction with the coactivator NR box peptide produced from TRAP220. For both LXRα and LXRβ, SR9238-induced recruitment of CoRNR box peptides was dose-dependent. Fasn and Srebp1c mRNA expression was significantly reduced in HepG2 cells treated with SR9238 [1]. In a cell-based cotransfection assay using a LXR-responsive luciferase reporter, SR9238 displayed potent inverse agonist activity, suppressing the basal transcriptional activity of both LXRα and LXRβ in HEK293 cells [1]. SR9238 (10 µM) effectively suppressed transcription from a FAS promoter-driven luciferase reporter in HEK293 cells [1]. In a biochemical time-resolved fluorescence resonance energy transfer (TR-FRET) cofactor interaction assay, SR9238 (10 µM) induced increased interaction of LXRα and LXRβ with CoRNR box peptides (NCoR ID1 and ID2) derived from the corepressor NCoR, while decreasing interaction with a coactivator NR box peptide (TRAP220/DRIP-2), confirming its inverse agonist mechanism [1]. The recruitment of NCoR ID1 peptide was dose-dependent with EC₅₀ values of 33 nM for LXRα and 13 nM for LXRβ [1]. In HepG2 cells cultured in the presence of insulin to induce lipogenesis, treatment with SR9238 (10 µM) for 1 day substantially suppressed the mRNA expression of FASN and SREBP1c [1]. Treatment of HepG2 cells with SR9238 (10 µM) for 48 hours blocked the constitutive nuclear localization of SREBP2, suggesting interference with its proteolytic processing [1]. In a nuclear receptor specificity panel, SR9238 displayed no activity at any of the other nuclear receptors tested, confirming its specificity for LXR [1]. |
| ln Vivo |
Two hours after SR9238 injection, the liver contained about 6 μM of the drug, whereas the plasma contained no SR9238 at all. SR9238 can alternatively be taken orally or administered intraperitoneally to detect its presence in the gut. The livers of the mice treated with SR9238 had a markedly lower lipid content. The findings demonstrated that, in comparison to mice treated with vehicle, the expression of Tnfa and Il1b was much lower in SR9238-treated mice (about 80% and >95%, respectively). In line with SR9238's positive effects on nonalcoholic steatohepatitis (NASH), F4/80 staining intensity was considerably lower in DIO mice treated with SR9238 than in DIO mice treated with vehicle. In comparison to rats treated with a vehicle, SR9238 therapy had no effect on body weight or body fat composition during the experiment. Hepatic steatosis, liver inflammation, and hepatocellular damage brought on by diet are all inhibited by SR9238 treatment [1]. In diet-induced obese (DIO) mice with hepatic steatosis, treatment with SR9238 (30 mg/kg/day, i.p., for 30 days) resulted in substantial repression of hepatic lipogenic gene expression: Fasn by ~60%, Srebp1c by ~80%, and Scd1 by almost 90% [1]. The same treatment significantly reduced hepatic lipid accumulation, as visualized and quantified in liver sections stained with Bodipy 493/503 [1]. SR9238 treatment also substantially reduced the expression of pro-inflammatory cytokines Tnfa (~80%) and Il1b (>95%) in the livers of DIO mice [1]. Immunohistochemistry (F4/80 staining) showed that SR9238-treated DIO mice had considerably lower Kupffer cell infiltration in the liver compared to vehicle-treated mice [1]. Plasma markers of hepatocellular injury (alkaline phosphatase, alanine transaminase, and aspartate aminotransferase) were substantially reduced in SR9238-treated DIO mice [1]. Unexpectedly, treatment of DIO mice with SR9238 reduced total plasma cholesterol, LDL, and HDL levels [1]. This was associated with an 80% suppression of hepatic Hmgcr (encoding HMG CoA reductase) mRNA expression and a massive reduction in Cyp7a1 expression, despite no change in Srebf2 expression [1]. In mice fed a normal chow diet, short-term treatment (30 mg/kg/day, i.p., for 7 days) with SR9238 increased hepatic expression of the pro-inflammatory cytokine Il1b, while concomitantly decreasing Fasn and Srebp1c expression [1]. |
| Enzyme Assay |
A biochemical nuclear receptor-cofactor peptide interaction assay was performed using time-resolved fluorescence resonance energy transfer (TR-FRET). Purified human LXR ligand-binding domains (LBDs) fused to a GST tag were used. Test compounds were serially diluted in DMSO and then diluted in assay buffer. The LXR-LBDs were added to assay plates, followed by a mixture of a fluorescently labeled cofactor peptide and a terbium-labeled anti-GST antibody. After incubation, the TR-FRET ratio (520 nm / 492 nm emission) was measured. This assay assessed the ability of compounds to induce or inhibit the interaction between LXR and peptides derived from corepressors (e.g., NCoR ID1, NCoR ID2) or coactivators (e.g., TRAP220) [1]. |
| Cell Assay |
For the LXR cotransfection assay, HEK293 cells were seeded in 96-well plates and transfected using a lipid-based transfection reagent. The transfection mixture included expression plasmids for full-length LXRα or LXRβ, a firefly luciferase reporter plasmid containing LXR response elements (or a specific promoter like FAS), and a renilla luciferase control plasmid. Twenty-four hours post-transfection, cells were treated with vehicle or compound for another 24 hours. Luciferase activity was measured using a dual-luciferase assay system, and firefly luciferase activity was normalized to renilla luciferase activity [1]. To induce lipogenesis in HepG2 cells, cells were seeded and grown to confluence in medium containing a high concentration of insulin (10 µg/ml) for 8 days, with media changes every two days [1]. For the SREBP2 translocation assay, HepG2 cells were seeded in chamber slides. The next day, cells were treated with DMSO or SR9238 (10 µM) in antibiotic-free media for 48 hours. Cells were then washed, fixed, and fluorescently stained for SREBP2 according to a standard immunocytochemistry protocol to assess its cellular localization (nuclear vs. cytoplasmic) [1]. |
| Animal Protocol |
For the primary efficacy study, 21-week-old male C57BL/6 diet-induced obese (DIO) mice, previously maintained on a high-fat diet (60% kcal from fat) for 14 weeks, were treated with SR9238. The compound was administered via intraperitoneal injection at a dose of 30 mg/kg, once daily, for 30 days. Body weight and food intake were monitored daily. Body composition was analyzed before and after the experiment. At the end of the treatment period, blood was collected for plasma analysis, and livers were harvested for gene expression analysis and histology [1]. For pharmacokinetic and tissue distribution studies, mice were injected intraperitoneally with SR9238 (30 mg/kg). In a single-dose experiment, liver and plasma were collected 2 hours post-injection for compound level analysis. In a multiple-dose experiment, mice were injected once daily for 3 consecutive days, and liver, intestine, skeletal muscle, brain, and plasma were collected for analysis [1]. For the short-term effect study on normal mice, chow-fed mice were treated with SR9238 (30 mg/kg/day, i.p.) for 7 days before tissue collection [1]. |
| ADME/Pharmacokinetics |
SR9238 is designed as a prodrug containing an ester group, which is predicted to be rapidly hydrolyzed by plasma lipases to its inactive acid analogue, SR10389 [1]. Following a single intraperitoneal injection (30 mg/kg) in mice, SR9238 was detected at approximately 6 µM concentration in the liver 2 hours post-dose, but was not detectable in the plasma [1]. After 3 consecutive days of intraperitoneal dosing (30 mg/kg/day), SR9238 was found at high levels in the liver and intestine, but was not detectable in plasma, skeletal muscle, or brain, indicating liver-specific exposure due to rapid first-pass metabolism [1]. The acid metabolite SR10389 displayed no activity in the LXR cotransfection assay [1]. |
| Toxicity/Toxicokinetics |
In DIO mice treated with SR9238 for 30 days, no alterations in body weight or percent body fat composition were observed compared to vehicle-treated animals [1]. Plasma glucose, insulin, and triglyceride levels were also unchanged [1]. Short-term treatment (7 days) of chow-fed mice with SR9238 induced a significant increase in the hepatic expression of the pro-inflammatory cytokine Il1b, suggesting a potential for an initial pro-inflammatory effect [1]. However, in the context of the high-inflammation NASH model (DIO mice), the long-term net effect of SR9238 treatment was anti-inflammatory [1]. |
| References |
[1]. A liver-selective LXR inverse agonist that suppresses hepatic steatosis. ACS Chem Biol. 2013 Mar 15;8(3):559-67. |
| Additional Infomation |
SR9238 is described as the first selective synthetic LXR inverse agonist [1]. It was developed based on a tertiary sulfonamide LXR antagonist scaffold and optimized to effectively recruit transcriptional corepressor proteins [1]. The compound was specifically designed with an ester moiety to confer liver selectivity, aiming to avoid potential side effects from suppressing LXR activity in peripheral tissues, such as impairing reverse cholesterol transport via ABCA1 [1]. Its efficacy in suppressing hepatic steatosis, inflammation, and injury in a mouse model suggests potential utility for treating non-alcoholic fatty liver disease (NAFLD) and non-alcoholic steatohepatitis (NASH) [1]. The unexpected reduction in plasma cholesterol levels is hypothesized to be due to suppressed cholesterol synthesis, possibly via an indirect mechanism involving inhibition of SREBP2 nuclear translocation, rather than direct LXR regulation of Hmgcr [1]. |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (4.20 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.20 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.0017 mL | 0.0084 mL | 0.0168 mL | |
| 5 mM | 335.9239 nL | 0.0017 mL | 0.0034 mL | |
| 10 mM | 167.9619 nL | 839.8097 nL | 0.0017 mL |